Abstract
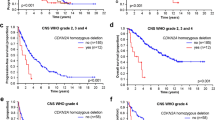
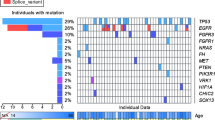
Despite tremendous progress in understanding the pathobiology of astrocytoma, major gaps remain in our knowledge of the molecular basis underlying the aggressiveness of high-grade astrocytoma (glioblastoma - GBM). Recently, we and others have shown nuclear respiratory factor 1 (NRF1) transcription factor being highly active in human cancers, but its role in astrocytoma remains unknown. Therefore, the purpose of this study was to uncover the role of NRF1 in the progression of GBM. NRF1 has higher mRNA expression and transcription factor activity in astrocytoma compared to non-tumor brain tissue. NRF1 activity also correlated with the aggressiveness of cancer. Increased NRF1 TF activity coupled with overexpression of RHOG was associated with poor survival of GBM patients. NRF1 activity was associated with transcriptomic signatures of neurogenesis, cell stemness, epithelial-mesenchymal transition and cell cycle progression. Overexpression of CDK4, AKT1, APAF1, HDAC1, NBN, TGFB1, & TNFRSF1A and downregulation of CASP3, IL7, STXBP1 and OPA1 predicted GBM malignancy in high expressors of NRF1 activity. Increased expression of the NRF1 motif containing genes, H6PD, NAT10, NBEAL2, and RNF19B predicted poor survival of IDH1 wild-type GBM patients. Poor survival outcomes and resistance to Temozolomide therapy were associated with higher NRF1 expression including its targets - LDHA, ZMAT3, NSUN2, ARMC5, NDEL1, CLPTM1L, ALKBH5, YIPF5, PPP2CA, and TFG. These findings suggest that aberrant NRF1 activity may contribute to the pathogenesis of GBM and severity of astrocytoma. Further analyses of NRF1 gene signatures will pave the way for next generation targeted therapies and drug combination strategies for GBM patients.








Similar content being viewed by others
References
Tamimi AF, Juweid M (2017) Epidemiology and Outcome of Glioblastoma. In: De Vleeschouwer S, editor. Glioblastoma [Internet]. Brisbane (AU): Codon Publications. Chapter 8. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470003 https://doi.org/10.15586/codon.glioblastoma.2017.ch8.
Weller M, Wick W, Aldape K, Brada M, Berger M, Pfister SM, Nishikawa R, Rosenthal M et al (2015) Glioma. Nature Reviews Disease Primers 1:15017. https://doi.org/10.1038/nrdp.2015.17
Koshy M, Villano JL, Dolecek TA, Howard A, Mahmood U, Chmura SJ, Weichselbaum RR, McCarthy BJ (2012) Improved survival time trends for glioblastoma using the SEER 17 population-based registries. J Neuro-Oncol 107:207–212. https://doi.org/10.1007/s11060-011-0738-7
Stupp R, Mason WP, van den Bent M, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA et al (2005) Radiotherapy plus concomitant and adjuvant Temozolomide for Glioblastoma. N Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
Pennacchio LA, Loots GG, Nobrega MA, Ovcharenko I (2007) Predicting tissue-specific enhancers in the human genome. Genome Res 17:201–211. https://doi.org/10.1101/gr.5972507
Li Z, Cogswell M, Hixson K, Brooks-Kayal AR, Russek SJ (2018) Nuclear respiratory factor 1 (NRF-1) controls the activity dependent transcription of the GABA-A receptor Beta 1 subunit gene in neurons. Front Mol Neurosci 11:285. https://doi.org/10.3389/fnmol.2018.00285
Das JK, Felty Q, Poppiti R, Jackson RM, Roy D (2018) Nuclear Respiratory Factor 1 Acting as an Oncoprotein Drives Estrogen-Induced Breast Carcinogenesis. Cells 7(12):234.https://doi.org/10.3390/cells7120234
Bhawe K, Roy D (2018) Interplay between NRF1, E2F4 and MYC transcription factors regulating common target genes contributes to cancer development and progression. Cell Oncol 41:465–484. https://doi.org/10.1007/s13402-018-0395-3
Sartor MA, Leikauf GD, Medvedovic M (2009) LRpath: A logistic regression approach for identifying enriched biological groups in gene expression data. Bioinformatics 25:211–217. https://doi.org/10.1093/bioinformatics/btn592
Falco MM, Bleda M, Carbonell-Caballero J, Dopazo J (2016) The pan-cancer pathological regulatory landscape. Sci Rep 6:39709. https://doi.org/10.1038/srep39709
Zhang J, Jiao J (2015) Molecular biomarkers for embryonic and adult neural stem cell and neurogenesis. Biomed Res Int 2015:727542–727514. https://doi.org/10.1155/2015/727542
Kunkle BW, Yoo C, Roy D (2013) Reverse engineering of modified genes by Bayesian network analysis defines molecular determinants critical to the development of glioblastoma. PLoS One 8:e64140. https://doi.org/10.1371/journal.pone.0064140
Mondal G, Stevers M, Goode B, Ashworth A, Solomon DA (2019) A requirement for STAG2 in replication fork progression creates a targetable synthetic lethality in cohesin-mutant cancers. Nat Commun 10:1686. https://doi.org/10.1038/s41467-019-09659-z
Zhang Y et al (2018) The p53 Pathway in Glioblastoma. Cancers 10:297. https://doi.org/10.3390/cancers10090297
Zuccarini M et al (2018) The Role of Wnt Signal in Glioblastoma Development and Progression: A Possible New Pharmacological Target for the Therapy of This Tumor. Genes (Basel) 9(2):105. https://doi.org/10.3390/genes9020105
Zhao M, Kong L, Liu Y, Qu H (2015) dbEMT: an epithelial-mesenchymal transition associated gene resource. Sci Rep 5:11459. https://doi.org/10.1038/srep11459
Merry TL, Ristow M (2016) Nuclear factor erythroid-derived 2-like 2 (NFE2L2, Nrf2) mediates exercise-induced mitochondrial biogenesis and the anti-oxidant response in mice. J Physiol 594:5195–5207. https://doi.org/10.1113/JP271957
Cam H, Balciunaite E, Blais A, Spektor A, Scarpulla RC, Young R, Kluger Y, Dynlacht BD (2004) A common set of gene regulatory networks links metabolism and growth inhibition. Mol Cell 16:399–411. https://doi.org/10.1016/j.molcel.2004.09.037
Demir O, Aysit N, onder Z, Turkel N, Ozturk G, Sharrocks AD, Kurnaz IA (2011) ETS-domain transcription factor Elk-1 mediates neuronal survival: SMN as a potential target. Biochim Biophys Acta 1812:652–662. https://doi.org/10.1016/j.bbadis.2011.02.012
Pantarelli C, Welch HCE (2018) Rac-GTPases and Rac-GEFs in neutrophil adhesion, migration and recruitment. Eur J Clin Investig 48(Suppl. 2):e12939. https://doi.org/10.1111/eci.12939
Kwiatkowska A, Didier S, Fortin S, Chuang Y, White T, Berens ME, Rushing E, Eschbacher J et al (2012) The small GTPase RhoG mediates glioblastoma cell invasion. Mol Cancer 11:65. https://doi.org/10.1186/1476-4598-11-65
Schug TT, Xu Q, Gao H, Peres-da-Silva A, Draper DW, Fessler MB, Purushotham A, Li X (2010) Myeloid deletion of SIRT1 induces inflammatory signaling in response to environmental stress. Mol Cell Biol 30(19):4712–4721. https://doi.org/10.1128/MCB.00657-10
Liu TF, McCall CE (2013) Deacetylation by SIRT1 reprograms inflammation and cancer. Genes Cancer 4(3–4):135–147. https://doi.org/10.1177/1947601913476948
Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W, Kos I, Batinic-Haberle I et al (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773. https://doi.org/10.1056/NEJMoa0808710
Wu H, Chen S, Yu J, Li Y, Zhang XY, Yang L, Zhang H, hou Q et al (2018) Single-cell Transcriptome analyses reveal molecular signals to intrinsic and acquired paclitaxel resistance in esophageal squamous Cancer cells. Cancer Lett 420:156–167. https://doi.org/10.1016/j.canlet.2018.01.059
Yamamoto K, Okamoto A, Isonishi S, Ochiai K, Ohtake Y (2001) A Novel Gene, CRR9, Which Was Up-Regulated in CDDP-Resistant Ovarian Tumor Cell Line, Was Associated with Apoptosis. Biochemical and Biophysical Research Communications. Biochem Arch 280(4):1148–1154. https://doi.org/10.1006/bbrc.2001.4250
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K et al (2000) Gene ontology: Tool for the unification of biology. The Gene Ontology Consortium Nat Genet 25:25–29. https://doi.org/10.1038/75556
The Gene Ontology Consortium (2019)The Gene Ontology Resource: 20 years and still GOing strong. Nucleic Acids Res 47:D330–D338. https://doi.org/10.1093/nar/gky1055
Mi H, Muruganujan A, Ebert D, Huang X, Thomas PD (2019) PANTHER version 14: More genomes, a new PANTHER GO-slim and improvements in enrichment analysis tools. Nucleic Acids Res 47:D419–D426. https://doi.org/10.1093/nar/gky1038
Acknowledgments
We are grateful to the United States-India Educational Foundation for a Senior US Fulbright Nehru Scholar Award to DR.
Availability of Data and Materials
We used public datasets in this study and details of analysis used in the current study are available from the first author or corresponding author upon request.
Author information
Authors and Affiliations
Contributions
The study was designed, analyzed and interpreted by DR and KB. KB was helped by QF and DR in writing the manuscript. SEH, NE, VS, IM and CW contributed in the preparation of the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
All authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bhawe, K., Felty, Q., Yoo, C. et al. Nuclear Respiratory Factor 1 (NRF1) Transcriptional Activity-Driven Gene Signature Association with Severity of Astrocytoma and Poor Prognosis of Glioblastoma. Mol Neurobiol 57, 3827–3845 (2020). https://doi.org/10.1007/s12035-020-01979-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-020-01979-2