Abstract
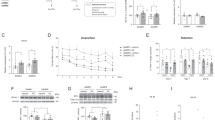
Elevated glucocorticoid (GC) exposure is widely accepted as a key factor in the age-related cognitive decline in rodents and humans. 11β-HSD1 is a key enzyme in the GCs pathway, catalyzing the conversion of 11β-dehydrocorticosterone to corticosterone in mice, with possible implications in neurodegenerative processes and cognitive impairment. Here, we determined the effect of a new 11β-HSD1 inhibitor, RL-118, administered to 12-month-old senescence-accelerated mouse-prone 8 (SAMP8) mice with neuropathological AD-like hallmarks and widely used as a rodent model of cognitive dysfunction. Behavioral tests (open field and object location) and neurodegeneration molecular markers were studied. After RL-118 treatment, increased locomotor activity and cognitive performance were found. Likewise, we found changes in hippocampal autophagy markers such as Beclin1, LC3B, AMPKα, and mTOR, indicating a progression in the autophagy process. In line with autophagy increase, a diminution in phosphorylated tau species (Ser 396 and Ser 404) jointly with an increase in ADAM10 and sAPPα indicated that an improvement in removing the abnormal proteins by autophagy might be implicated in the neuroprotective role of the 11β-HSD1 inhibitor. In addition, gene expression of oxidative stress (OS) and inflammatory markers, such as Hmox1, Aldh2, Il-1β, and Ccl3, were reduced in old treated mice in comparison to that of the control group. Consistent with this, we further demonstrate a significant correlation with autophagy markers and cognitive improvement and significant inverse correlation with autophagy, OS, and neuroinflammation markers. We concluded that inhibition of 11β-HSD1 by RL-118 prevented neurodegenerative processes and cognitive decline, acting on autophagy process, being an additional neuroprotective mechanism not described previously.








Similar content being viewed by others
References
López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153(6):1194–1217
Vílchez D, Saez I, Dillin A (2014) The role of protein clearance mechanisms in organismal ageing and age-related diseases. Nat Commun 5:5659
Boya P, Reggiori F, Codogno P (2013) Emerging regulation and functions of autophagy. Nat Cell Biol 15(7):713–720. https://doi.org/10.1038/ncb2788
Levine B (2005) Eating oneself and uninvited guests: autophagy-related pathways in cellular defense. Cell 120:159–162
Son JH, Shim JH, Kim K-H, Ha J-Y, Han JY (2012) Neuronal autophagy and neurodegenerative diseases. Exp Mol Med 44(2):89–98
Yu WH, Kumar A, Peterhoff C, Shapiro Kulnane L, Uchiyama Y, Lamb BT, Cuervo AM, Nixon RA (2004) Autophagic vacuoles are enriched in amyloid precursor protein-secretase activities: implications for beta-amyloid peptide over-production and localization in Alzheimer’s disease. Int J Biochem Cell Biol 36:2531–2540
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I, Ueno T, Koike M et al (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441:880–884
Gella A, Durany N (2009) Oxidative stress in Alzheimer disease. Cell Adhes Migr 3(1):88–93
Höhn A, Weber D, Jung T, Ott C, Hugo M, Kochlik B, Kehm R, König J et al (2016) Happily (n)ever after: aging in the context of oxidative stress, proteostasis loss and celular senescence. Redox Biol S2213-2317(16):30351–30352
Anderson RA, Qin B, Canini F, Poulet L, Roussel AM (2013) Cinnamon counteracts the negative effects of a high fat/high fructose diet on behavior, brain insulin signalling and Alzheimer-associated changes. PLoS One 8(12):e83243
Casadesús G, Shukitt-Hale B, Joseph JA (2002) Qualitative versus quantitative caloric intake: are they equivalent paths to successful aging? Neurobiol Aging 23(5):747–769
Ye SM, Johnson RW (1999) Increased interleukin-6 expression by microglia from brain of aged mice. J Neuroimmunol 93(1–2):139–148
Terao A, Apte-Deshpande A, Dousman L, Morairty S, Eynon BP, Kilduff TS, Freund YR (2002) Immune response gene expression increases in the aging murine hippocampus. J Neuroimmunol 132(1–2):99–112
Mathew R, Karp C, Beaudoin B, Vuong N, Chen G, Chen HY, Bray K, Reddy A et al (2009) Autophagy suppresses tumorigenesis through elimination of p62. Cell 137(6):1062–1075
Lara VP, Caramelli P, Teixeira AL, Barbosa MT, Carmona KC, Carvalho MG, Fernandes AP, Gomes KB (2013) High cortisol levels are associated with cognitive impairment no-dementia (CIND) and dementia. Clin Chim Acta 423:18–22
De Quervain DJ, Poirier R, Wollmer MA, Grimaldi LM, Tsolaki M, Streffer JR, Hock C, Nitsch RM et al (2004) Glucocorticoid-related genetic susceptibility for Alzheimer’s disease. Hum Mol Genet 13(1):47–52
Holmes MC, Carter RN, Noble J, Chitnis S, Dutia A, Paterson JM, Yau JLW (2010) 11β-Hydroxysteroid dehydrogenase type 1 expression is increased in the aged mouse hippocampus and parietal cortex and causes memory impairments. J Neurosci 30(20):6916–6920. https://doi.org/10.1523/JNEUROSCI.0731-10.2010
Yau JL, Noble J, Kenyon CJ, Hibberd C, Kotelevtsev Y, Mullins JJ, Seckl JR (2001) Lack of tissue glucocorticoid reactivation in 11β-hydroxysteroid dehydrogenase type 1 knockout mice ameliorates age-related learning impairments. Proc Natl Acad Sci U S A 98:4716–4721
Yau JL, McNair KM, Noble J, Brownstein D, Hibberd C, Morton N, Mullins JJ, Morris RG et al (2007) Enhanced hippocampal longterm potentiation and spatial learning in aged 11β-hydroxysteroid dehydrogenase type 1 knock-out mice. J Neurosci 27:10487–10496
Mohler EG, Browman KE, Roderwald VA, Cronin EA, Markosyan S, Scott Bitner R, Strakhova MI, Drescher KU et al (2011) Acute inhibition of 11β hydroxysteroid dehydrogenase type-1 improves memory in rodent models of cognition. J Neurosci 31(14):5406–5413
Leiva R, Griñán-Ferré C, Seira C, Valverde E, McBride A, Binnie M, Pérez B, Luque FJ et al (2017) Design, synthesis and in vivo study of novel pyrrolidine-based 11β-HSD1 inhibitors for age-related cognitive dysfunction. Eur J Med Chem 139:412–428. https://doi.org/10.1016/j.ejmech.2017.08.003
Nomura Y, Okuma Y (1999) Age-related defects in lifespan and learning ability in SAMP8 mice. Neurobiol Aging 20(2):111–115
Pallàs M, Camins A, Smith MA, Perry G, Lee HG, Casadesus G (2008) From aging to Alzheimer’s disease: unveiling “the switch” with the senescence-accelerated mouse model (SAMP8). J Alzheimers Dis 15(4):615–624
Morley JE, Farr SA, Kumar VB, Armbrecht HJ (2012) The SAMP8 mouse: a model to develop therapeutic interventions for Alzheimer’s disease. Curr Pharm 18(8):1123–1130
Murai T, Okuda S, Tanaka T, Ohta H (2007) Characteristics of object location memory in mice: behavioral and pharmacological studies. Physiol Behav 90:116–124
Griñán C, Palomera-Ávalos V, Puigoriol-Illamola D, Camins A, Porquet D, Plá V, Aguado F, Pallàs M (2016) Behavior and cognitive changes correlated with hippocampal neuroinflammaging and neuronal markers in female SAMP8, a model of accelerated senescence. Exp Gerontol 80:57–69
Sooy K, Webster SP, Noble J, Binnie M, Walker BR, Seckl JR, Yau JLW (2010) Partial deficiency or short-term inhibition of 11β-hydroxysteroid dehydrogenase type 1 improves cognitive function in ageing mice. J Neurosci 30(41):13867–13872. https://doi.org/10.1523/JNEUROSCI.2783-10.2010
Mah L, Binns MA, Steffens DC, Alzheimer’s Disease Neuroimaging Initiative (2015) Anxiety symptoms in amnestic mild cognitive impairment are associated with medial temporal atrophy and predict conversion to Alzheimer disease. Am J Geriatr Psychiatry 23:466–476. https://doi.org/10.1016/j.jagp.2014.10.005
Ramakers IHGB, Verhey FRJ, Scheltens P, Hampel H, Soininen H, Aalten P, Rikkert MO, Verbeek MM et al (2013) Anxiety is related to Alzheimer cerebrospinal fluid markers in subjects with mild cognitive impairment. Psychol Med 43:911–920. https://doi.org/10.1017/S0033291712001870
Harr MW, Distelhorst CW (2010) Apoptosis and autophagy: decoding calcium signals that mediate life or death. Cold Spring Harb Perspect Biol 2(10):a005579
El Zaoui I, Behar-Cohen F, Torriglia A (2015) Glucocorticoids exert direct toxicity on microvasculature: analysis of cell death mechanisms. Toxicol Sci 143(2):441–453
Triplett JC, Tramutola A, Swomley A, Kirk J, Grimes K, Lewis K, Orr M, Rodriguez K et al (2015) Age-related changes in the proteostasis network in the brain of the naked mole-rat: implications promoting healthy longevity. Biochim Biophys Acta 1852:2213–2224
Maiese K (2016) Targeting molecules to medicine with mTOR, autophagy and neurodegenerative disorders. Br J Clin Pharmacol 82(5):1245–1266
Guo H, Chen Y, Liao L, Wu W (2013) Resveratrol protects HUVECs from oxidized-LDL induced oxidative damage by autophagy upregulation via the AMPK7SIRT1 pathway. Cardiovasc Drugs Ther 27:189–198
Rahman MA, Rhim H (2017) Therapeutic implication of autophagy in neurodegenerative diseases. BMB Rep 50(7):345–354
Koyuncu S, Fatima A, Gutierrez-Garcia R, Vilchez D (2017) Proteostasis of huntingtin in health and disease. Int J Mol Sci 18(7):1568
Funderburk SF, Marcellino BK, Yue Z (2010) Cell “self-eating” (autophagy) mechanism in Alzheimer’s disease. Mt Sinai J Med 77(1):59–68. https://doi.org/10.1002/msj.20161
Caballero B, Coto-Montes A (2012) An insight into the role of autophagy in cell responses in the aging and neurodegenerative brain. Histol Histopathol 27(3):263–275
Friedman LG, Qureshi YH, Yu WH (2015) Promoting autophagic clearance: viable therapeutic targets in Alzheimer’s disease. Neurotherapeutics 12(1):94–108. https://doi.org/10.1007/s13311-014-0320-z
Wirawan E, Lippens S, Vanden Berghe T, Romagnoli A, Fimia GM, Piacentini M, Vandenabeele P (2012) Beclin 1: a role in membrane dynamics and beyond. Autophagy 8(1):6–17
Kang R, Zeh HJ, Lotze MT, Tang D (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18(4):571–580
Rusten TE, Stenmark H (2010) p62, an autophagy hero or culprit? Nat Cell Biol 12(3):207–209
Tanida I, Ueno T, Kominami E (2008) LC3 and autophagy. Methods Mol Biol 445:77–88
Klionsky DJ, Abeliovich H, Agostinis P, Agrawal DK, Aliev G, Askew DS, Baba M, Baehrecke EH et al (2008) Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 4(2):151–175
Ginaldi L, De Martinis M, D’Ostilio A, Marini L, Loreto F, Modesti M, Quaglino D (2001) Changes in the expression of surface receptors on lymphocyte subsets in the elderly: quantitative flow cytometric analysis. Am J Hematol 67(2):63–72
Williams AD, Brown BE, Putchim L, Sweet MJ (2015) Age-related shifts in bacterial diversity in a reef coral. PLoS One 10(12):e0144902
Acknowledgments
We thank Maggie Brunner, M.A., for revising the language and style of the manuscript.
Funding
This study was supported by the Ministerio de Economía y Competitividad of Spain SAF2016-33307 and SAF2014-57094R. D.P-I. and R.L. thank the Spanish Ministerio de Educación Cultura y Deporte for a PhD Grant (FPU program). Authors belong to 2017SGR106 from AGAUR, Catalonia.
Author information
Authors and Affiliations
Contributions
Participated in research design: Puigoriol-Illamola, Griñan-Ferré., Pallàs.
Conducted experiments: Puigoriol-Illamola, Griñan-Ferré, Vasilopoulou, Leiva.
Contributed new reagents or analytic tools: Leiva, Vazquez.
Performed data analysis: Puigoriol-Illamola, Griñan-Ferré.
Wrote or contributed to the writing of the manuscript: Puigoriol-Illamola, Griñán-Ferré, Leiva, Vazquez, Pallàs
Corresponding author
Rights and permissions
About this article
Cite this article
Puigoriol-Illamola, D., Griñán-Ferré, C., Vasilopoulou, F. et al. 11β-HSD1 Inhibition by RL-118 Promotes Autophagy and Correlates with Reduced Oxidative Stress and Inflammation, Enhancing Cognitive Performance in SAMP8 Mouse Model. Mol Neurobiol 55, 8904–8915 (2018). https://doi.org/10.1007/s12035-018-1026-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1026-8