Abstract
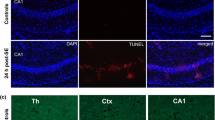
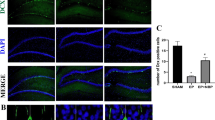
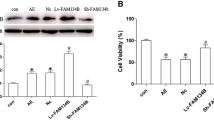
Epileptic seizures are generally associated with pathological changes in the hippocampus such as astrogliosis, mossy fiber sprouting, and neuronal damage. However, more than 30% of temporal lobe epilepsy in humans shows neither neuronal damage nor mossy fiber sprouting despite chronic epileptic seizures. A similar situation exists in certain commonly used strains of mice, specifically C57BL/6 and BALB/c, which exhibit epileptic seizures, but no neuronal damage upon kainic acid administration. This suggests that intrinsic factors may influence the pathological manifestations of epilepsy. Mechanisms which are behind the resistance of hippocampal cells to KA-induced neuronal death are unknown. Autophagy seems to be involved in the pathogenesis of many brain insults and to have a dual nature in neuroprotection and cell death. This study addresses the role of autophagy upon status epilepticus (SE) that has been induced by kainic acid (KA) in the C57BL/6 strain which is classified as seizure resistant. We analyzed the dynamics in the expression of autophagic and cell death markers in the hippocampus upon SE. Immunofluorescence data show that KA did not induce neuronal death in the hippocampal CA1–CA3 subfields; however, it leads to an exclusive activation of caspase-3 in the mossy fibers. We also found alterations in the expression of core proteins of the autophagic machinery. Levels of MAP1LC3, phospho-mTOR/mTOR, and Beclin 1 were significantly increased after induction of seizures. However, levels of Atg3, Atg14, Atg5-Atg12, Atg7, BAG3, Hsp70, and LAMP1 showed no significant alterations compared to controls. Although KA did not induce neuronal death, this study provides morphological and biochemical evidence that status epilepticus induced by KA activates caspase-3 in mossy fibers and induces autophagy in the C57BL/6 hippocampus. These data indicate that autophagic factors may modulate the sensitivity of pyramidal cells to KA and that autophagy may constitute a part of an endogenous neuroprotective arsenal which might be behind the resistance of C57BL/6-hippocampal cells to KA-induced neuronal death.







Similar content being viewed by others
References
DeLorenzo RJ, Pellock JM, Towne AR, Boggs JG (1995) Epidemiology of status epilepticus. J Clin Neurophysiol 12:316–325
Smith BN, Dudek FE (2001) Short- and long-term changes in CA1 network excitability after kainate treatment in rats. J Neurophysiol 85:1–9
Schauwecker PE (2002) Complications associated with genetic background effects in models of experimental epilepsy. Prog Brain Res 135:139–148
Schauwecker PE, Steward O (1997) Genetic determinants of susceptibility to excitotoxic cell death: implications for gene targeting approaches. Proc Natl Acad Sci U S A 94:4103–4410
Kurschner VC, Petruzzi RL, Golden GT, Berrettini WH, Ferraro TN (1998) Kainate and AMPA receptor binding in seizure-prone and seizure-resistant inbred mouse strains. Brain Res 780:1–8
Steward O, Schauwecker PE, Guth L, Zhang Z, Fujiki M, Inman D et al (1999) Genetic approaches to neurotrauma research: opportunities and potential pitfalls of murine models. Exp Neurol 157:19–42
Levine B, Klionsky DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6:463–477
Liu L, Hamre KM, Goldowitz D (2012) Kainic acid-induced neuronal degeneration in hippocampal pyramidal neurons is driven by both intrinsic and extrinsic factors: analysis of FVB/N↔C57BL/6 chimeras. J Neurosci 32:12093–12101
Kihara A, Kabeya Y, Ohsumi Y, Yoshimori T (2001) Beclinphosphatidylinositol 3-kinase complex functions at the trans-Golgi network. EMBO Rep 2:330–335
Kuma A, Hatano M, Matsui M, Yamamoto A, Nakaya H, Yoshimori T, Ohsumi Y, Tokuhisa T (2004) The role of autophagy during the early neonatal starvation period. Nature 432:1032–1036
Rami A, Kögel D (2008) Apoptosis meets autophagy-like cell death in the ischemic penumbra: two sides of the same coin? Autophagy 4:422–426
Nikoletopoulou V, Markaki M, Palikaras K, Tavernarakis N (2013) Crosstalk between apoptosis, necrosis and autophagy. Biochim Biophys Acta 1833:3448–3459
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R, Yokoyama M, Mishima K et al (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885–889
Steiger-Barraissoul S, Rami A (2009) Serum deprivation induced autophagy and predominantly an AIF-dependent apoptosis in hippocampal HT22 neurons. Apoptosis 14:1274–1288
Rami A (2009) Review: autophagy in neurodegeneration: firefighter and/or incendiarist? Neuropathol Appl Neurobiol 35:449–461
Fekadu J, Rami A (2016) Beclin-1 deficiency alters autophagosome formation, lysosome biogenesis and enhances neuronal vulnerability of HT22 hippocampal cells. Mol Neurobiol 53:5500–5509
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I, Ueno T, Koike M et al (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441:880–884
Pickford F, Masliah E, Britschgi M, Lucin K, Narasimhan R, Jaeger PA, Small S, Spencer B et al (2008) The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid beta accumulation in mice. J Clin Invest 118:2190–2199
D'Amelio M, Sheng M, Cecconi F (2012) Caspase-3 in the central nervous system: beyond apoptosis. Trends Neurosci 35:700–709
Snigdha S, Smith ED, Prieto GA, Cotman CW (2012) Caspase-3 activation as a bifurcation point between plasticity and cell death. Neurosci Bull 28:14–24
Wang JYY, Luo ZG (2014) Non-apoptotic role of caspase-3 in synapse refinement. Neurosci Bul 30:667–670
Li Z, Sheng M (2012) Caspases in synaptic plasticity. Mol Brain 5:15
Dash PK, Blum S, Moore AN (2000) Caspase activity plays an essential role in long-term memory. Neuroreport 11:2811–2816
Mattson MP, Keller JN, Begley JG (1998) Evidence for synaptic apoptosis. Exp Neurol 153:35–48
Chan SL, Mattson MP (1999) Caspase and calpain substrates: roles in synaptic plasticity and cell death. J Neurosci Res 58:167–190
Acknowledgements
This work was supported by the Adolf-Messer-Stiftung (grant to A. Rami—“Molecular mechanisms of autophagy”). We thank Alena Konoplew for the technical assistance. A.P. Benz was supported partly by the AMS and the FPF (Frankfurter Promotionsförderung). We thank Professor J. Stehle for support of our work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rami, A., Benz, A. Exclusive Activation of Caspase-3 in Mossy Fibers and Altered Dynamics of Autophagy Markers in the Mice Hippocampus upon Status Epilepticus Induced by Kainic Acid. Mol Neurobiol 55, 4492–4503 (2018). https://doi.org/10.1007/s12035-017-0665-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0665-5