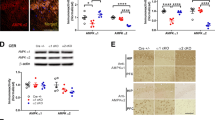
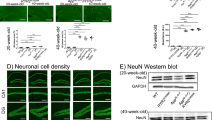
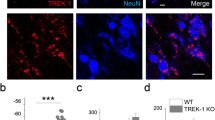
Abstract
Among five members of the K+-dependent Na+/Ca2+ exchanger (NCKX) family (NCKX1–5), only NCKX2 is highly expressed in mouse brain. NCKX2 in plasma membranes mediates cytosolic calcium excretion through electrogenic exchange of 4 Na+ for 1 Ca2+ and 1 K+. Here, we observed significantly decreased levels of NCKX2 protein and mRNA in the CA1 region of APP23 mice, a model of Alzheimer’s disease. We also found that, like APP23 mice, heterozygous NCKX2-mutant mice exhibit mildly impaired hippocampal LTP and memory acquisition, the latter based on novel object recognition and passive avoidance tasks. When we addressed underlying mechanisms, we found that both CaMKII autophosphorylation and CaMKIV phosphorylation significantly decreased in CA1 regions of NCKX2+/− relative to control mice. Likewise, phosphorylation of GluA1 (Ser-831) and CREB (Ser-133), respective downstream targets of CaMKII and CaMKIV, also significantly decreased in the CA1 region. BDNF protein and mRNA levels significantly decreased in CA1 of NCKX2+/− relative to control mice. Finally, CaN activity increased in CA1 of NCKX2+/− mice. Our findings suggest that like APP23 mice, NCKX2+/− mice may exhibit impaired learning and hippocampal LTP due to decreased CaM kinase II and CaM kinase IV activities.







Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- AMPAR:
-
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionate receptor
- BDNF:
-
Brain-derived neurotrophic factor
- CaMKII:
-
Calcium/calmodulin-dependent protein kinase II
- CaMKIV:
-
Calcium/calmodulin-dependent protein kinase IV
- CaN:
-
Calcineurin
- CREB:
-
cAMP-responsive element binding protein
- DG:
-
Dentate gyrus
- ERK:
-
Extracellular signal-regulated kinase
- fEPSPs:
-
Field excitatory post-synaptic potentials
- GFP:
-
Green fluorescent protein
- HFS:
-
High-frequency stimulation
- LTP:
-
Long-term potentiation
- NCKXs:
-
K+-dependent Na+/Ca2+ exchangers
- NCXs:
-
Na+/Ca2+ exchangers
- PP1:
-
Protein phosphatase 1
- WT:
-
Wild-type
References
Berridge MJ (1998) Neuronal calcium signaling. Neuron 21:13–26
Berrie MJ, Lipp P, Bootman MD (2000) The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol 1:11–21
Augustine GJ, Santamaria F, Tanaka K (2003) Local calcium signaling in neurons. Neuron 40:331–346
Berridge MJ, Bootman MD, Roderick HL (2003) Calcium signaling: dynamics, homeostasis and remodeling. Nat Rev Mol Cell Biol 4:517–529
Jeon D, Yang YM, Jeong MJ et al (2003) Enhanced learning and memory in mice lacking Na+/Ca2+ exchanger 2. Neuron 38:965–976
Li XF, Kiedrowski L, Tremblay F et al (2006) Importance of K+-dependent Na+/Ca2+-exchanger 2, NCKX2, in motor learning and memory. J Biol Chem 281:6273–6282
Lee SH, Park KH, Ho WK et al (2007) Postnatal developmental changes in Ca2+ homeostasis in supraoptic magnocellular neurons. Cell Calcium 41:441–450
Dong H, Light PE, French RJ et al (2001) Electrophysiological characterization and ionic stoichiometry of the rat brain K+-dependent Na+/Ca2+ exchanger, NCKX2. J Biol Chem 276:25919–25928
Cai X, Lytton J (2004) Molecular cloning of a sixth member of the K+-dependent Na+/Ca2+ exchanger gene family, NCKX6. J Biol Chem 279:5867–5876
Lytton J (2007) Na+/Ca2+ exchangers: three mammalian gene families control Ca2+ transport. Biochem J 406:365–382
Tsoi M, Rhee KH, Bungard D et al (1998) Molecular cloning of a novel potassium-dependent sodium-calcium exchanger from rat brain. J Biol Chem 273:4155–4162
Lytton J, Li XF, Dong H et al (2002) K+-dependent Na+/Ca2+ exchangers in the brain. Ann N Y Acad Sci 976:382–393
Lee KH, Ho WK, Lee SH (2013) Endocytosis of somatodendritic NCKX2 is regulated by Src family kinase-dependent tyrosine phosphorylation. Front Cell Neurosci 7:14
Bliss TV, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31–39
Malenka RC, Nicoll RA (1999) Long-term potentiation—a decade of progress? Science 285:1870–1874
Fukunaga K, Stoppini L, Miyamoto E et al (1993) Long-term potentiation is associated with a increased activity of Ca2+/calmodulin-dependent protein kinase II. J Biol Chem 268:7863–7867
Fukunaga K, Muller D, Miyamoto E (1995) Increased phosphorylation of Ca2+/calmodulin-dependent protein kinase II and its endogenous substrates in the induction of long term potentiation. J Biol Chem 270:6119–6124
Silva AJ, Paylor R, Wehner JM et al (1992) Impaired spatial learning in alpha-calcium-calmodulin kinase II mutant mice. Science 257:206–211
Lledo PM, Hjelmstad GO, Mukherji S et al (1995) Calcium/calmodulin-dependent kinase II and long-term potentiation enhance synaptic transmission by the same mechanism. Proc Natl Acad Sci U S A 92:11175–11179
Wang YJ, Chen GH, Hu XY et al (2005) The expression of calcium/calmodulin-dependent protein kinase II-alpha in the hippocampus of patients with Alzheimer’s disease and its links with AD-related pathology. Brain Res 1031:101–108
Barria A, Muller D, Derkach V et al (1997) Regulatory phosphorylation of AMPA-type glutamate receptor by CaM-KII during long-term potentiation. Science 276:2042–2045
Giese KP, Fedorov NB, Filipkowski RK et al (1998) Autophosphorylation at Thr286 of the alpha calcium-calmodulin kinase II in LTP and learning. Science 279:870–873
McGlade-McCulloh E, Yamamoto H, Tan SE et al (1993) Phosphorylation and regulation of glutamate receptors by calcium/calmodulin-dependent protein kinase II. Nature 362:640–642
Jensen KF, Ohmstede CA, Fisher RS et al (1991) Nuclear and axonal localization of Ca2+/calmodulin-dependent protein kinase type Gr in rat cerebellar cortex. Proc Natl Acad Sci U S A 88:2850–2853
Miyano O, Kameshita I, Fujisawa H (1992) Purification and characterization of a brain-specific multifunctional calmodulin-dependent protein kinase from rat cerebellum. J Biol Chem 267:1198–1203
Kasahara J, Fukunaga K, Miyamoto E (2001) Activation of calcium/calmodulin-dependent protein kinase IV in long term potentiation in the rat hippocampal CA1 region. J Biol Chem 276:24044–24050
Ho N, Liauw JA, Blaeser F et al (2000) Impaired synaptic plasticity and cAMP response element-binding protein activation in Ca2+/calmodulin-dependent protein kinase type IV/Gr-deficient mice. J Neurosci 20:6459–6472
Lee KH, Chatila TA, Ram RA et al (2009) Impaired memory of eyeblink conditioning in CaMKIV KO mice. Behav Neurosci 123:438–442
Wei F, Qiu CS, Liauw J et al (2002) Calcium calmodulin-dependent protein kinase IV is required for fear memory. Nat Neurosci 5:573–579
Takao K, Tanda K, Nakamura K et al (2010) Comprehensive behavioral analysis of calcium/calmodulin-dependent protein kinase IV knockout mice. PLoS One 5:e9460
Sturchler-Pierrat C, Abramowski D, Duke M et al (1997) Two amyloid precursor protein transgenic mouse models with Alzheimer’s disease-like pathology. Proc Natl Acad Sci U S A 94:13287–13292
Sturchler-Pierrat C, Staufenbiel M (2000) Pathogenic mechanisms of Alzheimer’s disease analyzed in the APP23 transgenic mouse model. Ann N Y Acad Sci 920:134–139
Calhoun ME, Burgermeister P, Phinney AL et al (1999) Neuronal overexpression of mutant amyloid precursor proteins results in prominent deposition of cerebrovascular amyloid. Proc Natl Acad Sci U S A 96:14088–14093
Moriguchi S, Ishizuka T, Yabuki Y et al (2016) Blockade of the KATP channel Kir6.2 by memantine represents a novel mechanism relevant to Alzheimer’s disease therapy. Mol Psychiatry. doi:10.1038/mp.2016.187
Keng VW, Yae K, Hayakawa T et al (2005) Region-specific saturation germline mutagenesis in mice using the Sleeping Beauty transposon system. Nat Methods 2:763–769
Moriguchi S, Yamamoto Y, Ikuno T et al (2011) Sigma-1 receptor stimulation by dehydroepiandrosterone ameliorates cognitive impairment through activation of CaM kinase II, protein kinase C and extracellular signal-regulated kinase in olfactory bulbectomized mice. J Neurochem 117:879–891
Moriguchi S, Sakagami H, Yabuki Y et al (2015) Stimulation of sigma-1 receptor ameliorates depressive-like behaviors in CaMKIV null mice. Mol Neurobiol 52:1210–1222
Moriguchi S, Yabuki Y, Fukunaga K (2012) Reduced calcium/calmodulin-dependent protein kinase II activity in the hippocampus is associated with impaired cognitive function in MPTP-treated mice. J Neurochem 120:541–551
Fukunaga K, Horikawa K, Shibata S et al (2002) Ca2+/calmodulin-dependent protein kinase II-dependent long-term potentiation in the rat suprachiasmatic nucleus and its inhibition by melatonin. J Neurosci Res 70:799–807
Taigen T, De Windt LJ, Lim HW et al (2000) Targeted inhibition of calcineurin prevents agonist-induced cardiomyocyte hypertrophy. Proc Natl Acad Sci U S A 97:1196–1201
Zheng F, Zhou X, Luo Y et al (2011) Regulation of brain-derived neurotrophic factor exon IV transcription through calcium responsive elements in cortical neurons. PLoS One 6:e28441
Kidane AH, Heinrich G, Dirks RP et al (2009) Differential neuroendocrine expression of multiple brain-derived neurotrophic factor transcripts. Endocrinology 150:1361–1368
Soderling TR, Derkach VA (2000) Postsynaptic protein phosphorylation and LTP. Trend Neurosci 23:75–80
Lisman J, Schulman H, Cline H (2002) The molecular basis of CaMKII function synaptic and behavioral memory. Nat Rev Neurosci 3:175–190
Blaustein MP, Lederer WJ (1999) Sodium/calcium exchange: its physiological implications. Physiol Rev 79:763–854
Kang H, Sun LD, Atkins CM et al (2001) An important role of neural activity-dependent CaMKIV signaling in the consolidation of long-term memory. Cell 106:771–783
Tao X, Finkbeiner S, Arnold DB et al (1998) Ca2+ influx regulates BDNF transcription by a CREB family transcription factor-dependent mechanism. Neuron 20:709–726
Gass P, Wolfer DP, Balschun D et al (1998) Deficits in memory tasks of mice with CREB mutations depend on gene dosage. Learn Mem 5:274–288
Montkowski A, Holsboer F (1997) Intact spatial learning and memory in transgenic mice with reduced BDNF. Neuroreport 8:779–782
Korte M, Kang H, Bonhoeffer T et al (1998) A role for BDNF in the late-phase of hippocampal long-term potentiation. Neuropharmacology 37:553–559
Sun P, Enslen H, Myung PS et al (1994) Differential activation of CREB by Ca2+/calmodulin-dependent protein kinases type II and type IV involves phosphorylation of a site that negatively regulates activity. Genes Dev 8:2527–2539
Schlman H, Lou LL (1989) Multifunctional Ca2+/calmodulin-dependent protein kinase: domain structure and regulation. Trends Biol Sci 14:62–66
Klee CB (1991) Concerted regulation of protein phosphorylation and dephosphorylation by calmodulin. Neurochem Res 16:1059–1065
Strack S, Barban MA, Wadzinski BE et al (1997) Differential inactivation of postsynaptic density-associated and soluble Ca2+/calmodulin-dependent protein kinase II by protein phosphatase 1 and 2A. J Neurochem 68:2119–2128
Hemmings HC Jr, Greengard P, Tung HY et al (1984) DARPP-32, a dopamine-regulated neuronal phosphoprotein, is a potent inhibitor of protein phosphatase-1. Nature 310:503–505
Morioka M, Nagahiro S, Fukunaga K et al (1997) Calcineurin in the adult rat hippocampus: different distribution in CA1 and CA3 subfields. Neuroscience 78:673–684
Kasahara J, Fukunaga K, Miyamoto E (1999) Differential effects of a calcineurin inhibitor on glutamate-induced phosphorylation of Ca2+/calmodulin-dependent protein kinases in cultured rat hippocampal neurons. J Biol Chem 274:9061–9067
Thayer SA, Usachev YM, Pottorf WJ (2002) Modulating Ca2+ clearance from neurons. Front Biosci 7:D1255–D1279
Lee KH, Lee JS, Lee D et al (2012) KIF21A-mediated axonal transport and selective endocytosis underlie the polarized targeting of NCKX2. J Neurosci 32:4102–4117
Wang JH, Kelly PT (1995) Postsynaptic injection of Ca2+/CaM induces synaptic potentiation requiring CaMKII and PKC activity. Neuron 15:443–452
Acknowledgements
This work was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology, and the Ministry of Health and Welfare of Japan (KAKENHI 22390109 to K.F.; 20790398 to S.M.; 23590319 to T.I.; 25460350 to S.K.), the Uehara Memorial Foundation (K.F.) and the Smoking Research Foundation (S.M.). We also thank Novartis Pharma for providing APP23 mice.
Author information
Authors and Affiliations
Contributions
S.M., Y.Y., R.I., H.I., Y.S., K.S., H.T., and T.I. performed experiments. S.K., K.H., J.T., and T.I. provided NCKX2 antibody and NCKX2 knockout mice, and critical advice. S.M. and K.F. wrote the manuscript and designed the study.
Corresponding authors
Ethics declarations
All animal protocols were approved by the Committee on Animal Experiments at Tohoku University.
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Moriguchi, S., Kita, S., Yabuki, Y. et al. Reduced CaM Kinase II and CaM Kinase IV Activities Underlie Cognitive Deficits in NCKX2 Heterozygous Mice. Mol Neurobiol 55, 3889–3900 (2018). https://doi.org/10.1007/s12035-017-0596-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0596-1