Abstract
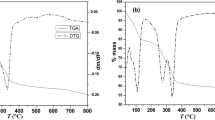
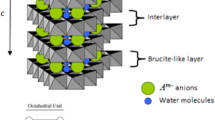
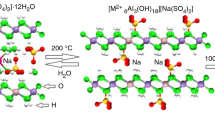
Imbibition of lithium sulphate into aluminium hydroxide is known to result in a sulphate-intercalated layered double hydroxide (LDH) of Li and Al. The perchlorate ion has the same size and molecular symmetry as the sulphate ion, but only half its charge. Consequently, twice the number of \(\hbox {ClO}_{4}^{-}\) ions is needed to balance LDHs the charge on the metal hydroxide layer, compared to the \(\hbox {SO}_{4}^{2-}\) ions. In this work, the \(\hbox {ClO}_{4}^{-}\)-intercalated LDHs were obtained from both the bayerite and gibbsite precursors. Inclusion of the hydration sphere along with the \(\hbox {ClO}_{4}^{-}\) anion, induced turbostratic disorder in the stacking of the metal hydroxide layers. Temperature-induced dehydration (\(T \sim 100\)–\(140{^{\circ }}\hbox {C}\)) brought about a partial ordering in the interlayer region and the \(\hbox {ClO}_{{4}}^{{-}}\) ion oriented itself with one of its \(C_{{2}}\)-axes parallel to the metal hydroxide layer. The close packing of \(\hbox {ClO}_{4}^{-}\) ions could be realized by the complete dehydration of LDH and the distribution of the \(\hbox {ClO}_{4}^{-}\) ions in all the available interlayer sites. In contrast, within the crystal of the sulphate analogue, the sulphate ions occupy only half the number of interlayer sites. The other half is occupied by the residual water molecules, as the sulphate analogue does not fully dehydrate even at elevated temperatures. This difference in the behaviour of the two LDHs has its origin in the large difference in the hydration enthalpies of the two anions.






Similar content being viewed by others
References
Megaw H D 1934 Z. Kristallogr. 87 185
Rothbauer R, Zigan F and O’Daniel H 1967 Z. Kristallogr. 125 317
Lipin V A 2001 Russ. J. Appl. Chem. 74 184
Clark G R, Rodgers K A and Henderson G S 1998 Z. Kristallogr. 213 96
Williams G R, Norquist A J and O’Hare D 2004 Chem. Mater. 16 975
Fogg A M and O’Hare D 1999 Chem. Mater. 11 1771
Besserguenev A V, Fogg A M, Francis R J, Price S J, O’Hare D, Isupov V P et al 1997 Chem. Mater. 9 241
Serna C J, Rendon J L and Iglesias J E 1982 Clays Clay Miner. 30 180
Britto S and Kamath P V 2009 Inorg. Chem. 48 11646
Nagendran S, Periyasamy G and Kamath P V 2016 Dalton Trans. 45 18324
Nagendran S and Kamath P V 2017 Inorg. Chem. 56 5026
Nagendran S, Periyasamy G and Kamath P V 2018 J. Solid State Chem. 266 226
Pachayappan L and Kamath P V 2019 Clays Clay Miner. 67 154
Britto S and Kamath P V 2011 Inorg. Chem. 50 5619
Hou X, Bish D L, Wang S L, Johnston C T and Kirkpatrick R J 2003 Am. Miner. 88 167
Poeppelmeier K R and Hwu S J 1987 Inorg. Chem. 26 3297
Hou X and Kirkpatrick R J 2002 Chem. Mater. 14 1195
Treacy M M J, Newsam J M and Deem M W 1991 Proc. Math. Phys. Eng. Sci. p 499
Treacy M M J, Deem M W and Newsam J M 2005 DIFFaX version 1.812. http://www.public.asu.edu/~mtreacy/DIFFaX.html
Nagendran S and Kamath P V 2013 Eur. J. Inorg. Chem. 2013 4686
Favre-Nicolin V and Černý R 2002 J. Appl. Crystallogr. 35 734
Marcus Y 1988 J. Solution Chem. 88 1475
Smith D W 1977 J. Chem. Educ. 54 540
Eklund L 2014 Hydration of oxo anions. A combined computational and experimental structure and dynamics study in aqueous solutions, Swedish University of Agricultural Sciences: Uppsala
Acknowledgements
We thank the Department of Science and Technology (DST), Government of India (GOI), for financial support. LP is a recipient of support under the Women Scientists (WOS-A) Scheme of the DST.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pachayappan, L., Kamath, P.V. Reversible hydration of the perchlorate-intercalated layered double hydroxides of Li and Al: structure models for the dehydrated phases. Bull Mater Sci 43, 141 (2020). https://doi.org/10.1007/s12034-020-02126-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-020-02126-2