Abstract
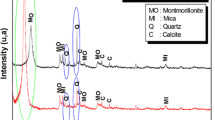
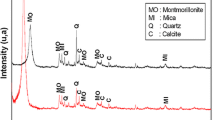
In this study, we propose Maghnite-\(\hbox {H}^{+}\), as an ecological, cost-effective and easily renewable catalyst, for the polymerization of decamethylcyclopentasiloxane (D5). The Maghnite is a clay consisting primarily of smectite minerals (montmorillonite group), which can be activated/reactivated through a simple process, by replacing interlayer cations by protons. Linear polymers with higher molecular mass and narrower molecular mass distribution (MMD) can be obtained with less cyclic by-products, using Maghnite-\(\hbox {H}^{+}\). The reaction was investigated in bulk, taking into account the effect of time, temperature and amount of catalyst on the monomer conversion and MMD. When the yield rises to 96%, the MMD is broadened, suggesting the formation of cyclic oligomers and/or crosslinking bonds, this was confirmed experimentally by infra-red spectroscopy, proton nuclear magnetic resonance (\(^{1}\hbox {H}\) NMR) and \(^{13}\hbox {C}\) NMR analyses. The kinetics have been also studied; the reaction is first order in the monomer. The proposed reaction mechanism shows clearly the role of Maghnite-\(\hbox {H}^{+}\) for D5 polymerization.











Similar content being viewed by others
References
Clark J H and Rhodes C N 2000 in: J H Clark (ed) Clean synthesis using porous inorganic solid catalysts and supported reagents (Cambridge: Royal Society of Chemistry) p 10
Chen B, Zhan X, Yi L and Chen F 2007Chin. J. Chem. Eng. 15 661
Friedel C and Crafts J M 1877 Compt. Rend. 84 1392
Friedel C and Crafts J M 1877 Bull. Soc. Chim. Fr. 27 530
Rodriquez F 1989 in Principles of polymer systems (New York: Hemisphere Publishing Corp) p 589
DeGroot J V, Norris A M, Shedric O and Clappe T V 2004 in: R Norwood, M Eich and M Kuzyk (eds) Highly transparent silicone materials, Proceedings of SPIE, Midland, p 116
Narins R S and Beer K 2006 Plast. Reconstr. Surg. 118 77
Kendrick T C, Parbhoo B and White J W 1991 in: D A Armitage, R J P Corriu, T C Kendrick, B Parbhoo, T D Tilley and J C Young (eds) The silicon–heteroatom bond (Chichester: J. Wiley & Sons) p 67
Chojnowski J and Cypryk M 2000 in: R G Jones, W Ando and J Chojnowski (eds) Silicon-containing polymers (Dordrecht: Kluwer) p 3
Chojnowski J, Cypryk M and Kazmierski K 2002 Macromolecules 36 9904
Sigwalt P 1987 Polym. J. 19 567
Molenberg A and Möller M 1995 Macromol. Rapid Commun. 16 449
Pibre G, Chaumont P, Fleury E and Cassagnau P 2008 Polymer 49 234
Gee R P 2015 Colloids Surf. A Physicochem. Eng. Asp. 481 297
Sun C N, Shen M M, Deng L L, Mo J Q and Zhou B W 2014 Chin. Chem. Lett. 25 621
Sijiu J, Teng Q and Xiaoyu L 2010 Polymer 51 4087
Conan J T, William P W and Guoping C 2003 Polymer 44 4149
Chojnowski J, Rubinsztajn S, Fortuniak W and Kurjata J 2007 J. Inorg. Organomet. Polym. Mater. 17 173
Wilczek L, Rubinsztajn S and Chojnowski J 1986 Makromol. Chem. 187 39
Hardman B and Torkelson A 1989 in: Encyclopedia of polymer science and engineering (New York: John Wiley & Sons) p 204
Kherroub D E, Belbachir M and Lamouri S 2014 Orient. J. Chem. 30 1647
Belbachir M and Bensaoula A 2001 US Patent 6,274,527B1
Kherroub D E, Belbachir M, Lamouri S, Bouhadjar L and Chikh K 2013 Orient. J. Chem. 29 1429
Kherroub D E, Belbachir M and Lamouri S 2015 Res. Chem. Intermed. 41 5217
Kherroub D E, Belbachir M and Lamouri S 2015 Bull. Mater. Sci. 38 1
Meghabar R, Megherbi A and Belbachir M 2003 Polymer 44 4097
Bouchama A, Ferrahi M I and Belbachir M 2015 J. Mater. Environ. Sci. 6 977
Ya-Qing Z, Xiang K, Xiao-Li Z and Zheng-Hong L 2010 Powder Technol. 201 146
Jian W, Xueming C, Panjin J, Qing H and Mingtao R 2015 Mater. Chem. Phys. 149 216
Ramli M R, Othman M B H, Arifin A and Ahmad Z 2011 Polym. Degrad. Stab. 96 2064
Namrata S T, Florence D J, Lawrence F and Jacques L 2012 Open J. Org. Polym. Mater. 2 13
Dollase T, Spiess H W, Gottlieb M and Yerushalmi-Rozen R 2002 Europhys. Lett. 60 390
Kherroub D E, Belbachir M and Lamouri S 2014 Bull. Chem. React. Eng. Catal. 9 74
Kherroub D E, Belbachir M and Lamouri S 2015 Arab. J. Sci. Eng. 40 143
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kherroub, D.E., Belbachir, M., Lamouri, S. et al. Acid-activated bentonite (Maghnite-\(\hbox {H}^{+})\) as a novel catalyst for the polymerization of decamethylcyclopentasiloxane. Bull Mater Sci 41, 36 (2018). https://doi.org/10.1007/s12034-018-1563-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-018-1563-9