Abstract
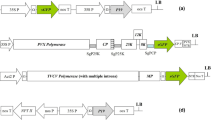
The aim of this study is to assess the effect of methyl jasmonate (MeJA) and temperature on the valuable pharmaceuticals expression in a virus-mediated transient expression system, and so the Zuchini Yellow Mosaic Virus (ZYMV) based vector was used for transferring the GFP reporter gene and recombinant tissue plasminogen activator (rtPA) gene (K2S) to cucurbit (Cucurbita pepo L.). MeJA, temperature and time (days after inoculation), were evaluated as a factorial experiment in a completely randomized design (CRD). At first, the effect of all treatment combinations on GFP expression was assessed. At this step, the ELISA test was used to select the optimum treatment combination. ELISA method revealed the significant difference between applied treatments. The optimized treatment significantly increased the expression of rtPA compared to the control. The Real-Time PCR reaction for both GFP and rtPA genes showed no significant differences between optimum and control treatments, however, transcripts of the small subunit of RuBisCO were extremely down-regulated in optimum treatment condition. Reduction in RuBisCO expression at protein level was tangible under treatment condition based on the ELISA test. Therefore, it can be inferred that suppressing the expression of RuBisCO, probably resulted in higher access of expression system to free amino acids inside the cell. In this study, MeJA has been shown to be a positive factor, but the low temperature (17 °C), unlike previous studies, suppressed the expression of recombinant protein unexpectedly, probably due to the incompatibility of the viral construct with low temperature. In conclusion, the use of a suitable gene construct, which is not sensitive to temperature, is likely to result in a more favorable outcome.








Similar content being viewed by others
References
Spiegel, H., Stöger, E., Twyman, R. M., & Buyel, J. F. (2018). Current status and perspectives of the molecular farming landscape. Molecular Pharming: Applications, Challenges, and Emerging Areas. https://doi.org/10.1002/9781118801512.ch1
Fischer, R., Schillberg, S., Buyel, F., J., and Twyman, M., R (2013). Commercial aspects of pharmaceutical protein production in plants. Current Pharmaceutical Design, 19(31), 5471–5477.
Gomord, V., Chamberlain, P., Jefferis, R., & Faye, L. (2005). Biopharmaceutical production in plants: Problems, solutions and opportunities. Trends. Biotechnol., 23(11), 559–565.
Sainsbury, F., & Lomonossoff, G. P. (2014). Transient expressions of synthetic biology in plants. Current Opinion in Plant Biology, 19, 1–7.
Weathers, P. J., Towler, M. J., & Xu, J. (2010). Bench to batch: advances in plant cell culture for producing useful products. Applied Microbiology and Biotechnology, 85(5), 1339–1351.
Ellis, R. J. (1979). The most abundant protein in the world. Trends in Biochemical Science, 4(11), 241–244.
Cavalcante, A. P. R., Jacinto, T., & Machado, O. L. T. (1999). Methyl jasmonate changes the levels of rubisco and other leaf proteins in Ricinus communis. Acta Physiology Plant, 21(2), 161–166.
Attaran, E., Major, I. T., Cruz, J. A., Rosa, B. A., Koo, A. J., Chen, J., Kramer, D. M., He, S. Y., & Howe, G. A. (2014). Temporal dynamics of growth and photosynthesis suppression in response to jasmonate signaling. Plant Physiology, 165(3), 1302–1314.
Thines, B., Katsir, L., Melotto, M., Niu, Y., Mandaokar, A., Liu, G., Nomura, K., He, S. Y., & Howe, G. A. (2007). JAZ repressor proteins are targets of the SCF COI1 complex during jasmonate signalling. Nature, 448(7154), 661.
Hou, X., Lee, L. Y. C., Xia, K., Yan, Y., & Yu, H. (2010). DELLAs modulate jasmonate signaling via competitive binding to JAZs. Development Cell, 19(6), 884–894.
Chini, A., Fonseca, S., Fernandez, G., Adie, B., Chico, J. M., Lorenzo, O., Garcia-Casado, G., López-Vidriero, I., Lozano, F. M., Ponce, M. R., & Micol, J. L. (2007). The JAZ family of repressors is the missing link in jasmonate signalling. Nature., 448(7154), 666.
Desai, P. N., Shrivastava, N., & Padh, H. (2010). Production of heterologous proteins in plants: Strategies for optimal expression. Biotechnology Advances, 28(4), 427–435.
Kim, S. T., Cho, K. S., Yu, S., Kim, S. G., Hong, J. C., Han, C. D., Bae, D. W., Nam, M. H., & Kang, K. Y. (2003). Proteomic analysis of differentially expressed proteins induced by rice blast fungus and elicitor in suspension-cultured rice cells. Proteomics, 3(12), 2368–2378.
Robert, S., Goulet, M. C., D’Aoust, M. A., Sainsbury, F., & Michaud, D. (2015). Leaf proteome rebalancing in Nicotiana benthamiana for upstream enrichment of a transiently expressed recombinant protein. Plant Biotechnology Journal, 13(8), 1169–1179.
Fraissinet-Tachet, L., Baltz, R., Chong, J., Kauffmann, S., Fritig, B., & Saindrenan, P. (1998). Two tobacco genes induced by infection, elicitor and salicylic acid encode glucosyltransferases acting on phenylpropanoids and benzoic acid derivatives, including salicylic acid. FEBS Letters, 437(3), 319–323.
Okada, K., Abe, H., & Arimura, G. I. (2014). Jasmonates induce both defense responses and communication in monocotyledonous and dicotyledonous plants. Plant Cell Physiology, 56(1), 16–27.
Faye, L., Boulaflous, A., Benchabane, M., Gomord, V., & Michaud, D. (2005). Protein modifications in the plant secretory pathway: current status and practical implications in molecular pharming. Vaccine, 23(15), 1770–1778.
Doran, P. M. (2006). Foreign protein degradation and instability in plants and plant tissue cultures. Trends Biotechnology, 24(9), 426–432.
De Neve, M., De Loose, M., Jacobs, A., Van Houdt, H., Kaluza, B., Weidle, U., Van Montagu, M., & Depicker, A. (1993). Assembly of an antibody and its derived antibody fragment in Nicotiana and Arabidopsis. Transgenic Research, 2(4), 227–237.
Outchkourov, N. S., Rogelj, B., Strukelj, B., & Jongsma, M. A. (2003). Expression of sea anemone equistatin in potato. Effects of plant proteases on heterologous protein production. Plant Physiology, 133(1), 379–390.
Khoudi, H., Laberge, S., Ferullo, J. M., Bazin, R., Darveau, A., Castonguay, Y., Allard, G., Lemieux, R., & Vézina, L. P. (1999). Production of a diagnostic monoclonal antibody in perennial alfalfa plants. Biotechnology and Bioengineering, 64(2), 135–143.
Sharp, J. M., & Doran, P. M. (2001). Characterization of monoclonal antibody fragments produced by plant cells. Biotechnology and Bioengineering, 73(5), 338–346.
Hehle, V. K., Paul, M. J., Drake, P. M., Ma, J. K., & van Dolleweerd, C.J. (2011) Antibody degradation in tobacco plants: a predominantly apoplastic process. BMC Biotechnology. 11(1), 128.
Weaver, L. M., Himelblau, E., & Amasino, R. M. (1997). Leaf senescence: gene expression and regulation, in Genetic engineering (pp. 215–234). Boston: Springer.
Smart, C. M. (1994). Gene expression during leaf senescence. New Phytology, 126(3), 419–448.
Zhao, Y., Ge, W., Kong, Y., & Zhang, C. (2003). Cloning, expression, and renaturation studies of reteplase. Journal of Microbiology and Biotechnology, 13(6), 989–992.
Hafizi, A., Malboobi, M. A., Jalali-Javaran, M., Maliga, P., & Alizadeh, H. (2017). Covalent-display of an active chimeric-recombinant tissue plasminogen activator on polyhydroxybutyrate granules surface. Biotechnology Letters, 39(11), 1683–1688.
Abdoli-Nasab, M., Jalali-Javaran, M., Cusidó, R. M., Palazón, J., Baghizadeh, A., & Alizadeh, H. (2013). Expression of the truncated tissue plasminogen activator (K2S) gene in tobacco chloroplast. Molecular Biology Reports, 40(10), 5749–5758.
Javaran, V. J., Shafeinia, A., Javaran, M. J., Gojani, E. G., & Mirzaee, M. (2017). Transient expression of recombinant tissue plasminogen activator (rt-PA) gene in cucurbit plants using viral vector. Biotechnology Letters, 39(4), 607–612.
Hidalgo, D., Abdoli-Nasab, M., Jalali-Javaran, M., Bru-Martínez, R., Cusidó, R. M., Corchete, P., & Palazon, J. (2017). Biotechnological production of recombinant tissue plasminogen activator protein (reteplase) from transplastomic tobacco cell cultures. Plant Physiology and Biochemistry, 118, 130–137.
Hsu, C. H., Lin, S. S., Liu, F. L., Su, W. C., & Yeh, S. D. (2004). Oral administration of a mite allergen expressed by zucchini yellow mosaic virus in cucurbit species downregulates allergen-induced airway inflammation and IgE synthesis. The Journal of Allergy and Clinical Immunology, 113(6), 1079–1085.
Obrero, A., Die, J. V., Román, B., Gómez, P., Nadal, S., & González-Verdejo, C. I. (2011). Selection of reference genes for gene expression studies in zucchini (Cucurbita pepo) using qPCR. Journal of Agriculture and Food Chemistry, 59(10), 5402–5411.
Goossens, A., Van Montagu, M., & Angenon, G. (1999). Co-introduction of an antisense gene for an endogenous seed storage protein can increase expression of a transgene in Arabidopsis thaliana seeds. FEBS Letters, 456(1), 160–164.
Stanton, M. A., Ullmann-Zeunert, L., Wielsch, N., Bartram, S., Svatoš, A., Baldwin, I. T., & Groten, K. (2013). Silencing ribulose-1, 5-bisphosphate carboxylase/oxygenase expression does not disrupt nitrogen allocation to defense after simulated herbivory in Nicotiana attenuata. Plant Signal Behavavior, 8(12), 27570.
Murby, M., Uhlén, M., & Ståhl, S. (1996). Upstream strategies to minimize proteolytic degradation upon recombinant production in Escherichia coli. Protein Expression and Purification, 7(2), 129–136.
Stevens, L. H., Stoopen, G. M., Elbers, I. J., Molthoff, J. W., Bakker, H. A., Lommen, A., … and Jordi, W. (2000). Effect of climate conditions and plant developmental stage on the stability of antibodies expressed in transgenic tobacco. Plant Physiology, 124(1), 173–182.
Kosaka, Y., Ryang, B. S., Kobori, T., Shiomi, H., Yasuhara, H., & Kataoka, M. (2006). Effectiveness of an attenuated Zucchini yellow mosaic virus isolate for cross-protecting cucumber. Plant Disease, 90(1), 67–72.
Mason, H. S., DeWald, D. B., & Mullet, J. E. (1993). Identification of a methyl jasmonate-responsive domain in the soybean vspB promoter. The Plant Cell, 5(3), 241–251.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karimzadegan, V., Jalali Javaran, V., Shams Bakhsh, M. et al. The Effect of Methyl Jasmonate and Temperature on the Transient Expression of Recombinant Proteins in Cucurbita pepo L.. Mol Biotechnol 61, 84–92 (2019). https://doi.org/10.1007/s12033-018-0138-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-018-0138-8