Abstract
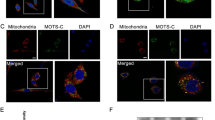
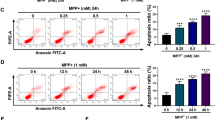
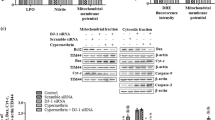
Mitochondria harbor small circular genomes (mtDNA) that encode 13 oxidative phosphorylation (OXPHOS) proteins, and types of damage to mtDNA may contribute to neuronal damage. Recent studies suggested that regulation of mtDNA repair proteins may be a potential strategy for treating neuronal damage. The mtDNA repair system contains its own repair enzymes and is independent from the nuclear DNA repair system. Endo/exonuclease G-like(EXOG) is a mitochondria-specific 5-exo/endonuclease required for repairing endogenous single-strand breaks (SSBs) in mtDNA. However, whether EXOG plays a key role in neuronal damage induced by rotenone remains unknown. Thus, in this study, we aimed to investigate the effect of EXOG on mtDNA repair and mitochondrial functional maintenance in rotenone-induced neurotoxicity. Our results indicated that rotenone influenced the expression and location of EXOG in PC12 cells. Meanwhile, after rotenone exposure, the expression was reduced for proteins responsible for mtDNA repair, including DNA polymerase γ (POLG), high-temperature requirement protease A2 (HtrA2), and the heat-shock factor 1-single-stranded DNA-binding protein 1 (HSF1-SSBP1) complex. Further analysis demonstrated that EXOG knockdown led to reduced mtDNA copy number and mtDNA transcript level and increased mtDNA deletion, which further aggravated the mtDNA damage and mitochondrial dysfunction under rotenone stress. In turn, EXOG overexpression protected PC12 cells from mtDNA damage and mitochondrial dysfunction induced by rotenone. As a result, EXOG knockdown reduced cell viability and tyrosine hydroxylase expression, while EXOG overexpression alleviated rotenone’s effect on cell viability and tyrosine hydroxylase expression in PC12 cells. Further, we observed that EXOG influenced mtDNA repair by regulating protein expression of the HSF1-SSBP1 complex and POLG. Furthermore, our study showed that PGC-1α upregulation with ZLN005 led to increased protein levels of EXOG, POLG, HSF1, and SSBP1, all of which contribute to mtDNA homeostasis. Therefore, PGC-1α may be involved in mtDNA repair through interacting with multiple mtDNA repair proteins, especially with the help of EXOG. In summary, EXOG regulation by PGC-1α plays an essential role in rotenone-induced neurotoxicity in PC12 cells. EXOG represents a protective effect strategy in PC12 cells exposed to rotenone.








Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Alencar RR, Batalha C, Freire TS, de Souza-Pinto NC (2019) Enzymology of mitochondrial DNA repair enzymes 45:257–287. https://doi.org/10.1016/bs.enz.2019.06.002
Bartz RR et al (2011) Staphylococcus aureus sepsis and mitochondrial accrual of the 8-oxoguanine DNA glycosylase DNA repair enzyme in mice. Am J Respir Crit Care Med 183:226–233. https://doi.org/10.1164/rccm.200911-1709OC
Bruni F, Polosa PL, Gadaleta MN, Cantatore P, Roberti M (2010) Nuclear respiratory factor 2 induces the expression of many but not all human proteins acting in mitochondrial DNA transcription and replication. J Biol Chem 285:3939–3948. https://doi.org/10.1074/jbc.M109.044305
Corona JC, Duchen MR (2015) PPARγ and PGC-1α as therapeutic targets in Parkinson’s. Neurochem Res 40:308–316. https://doi.org/10.1007/s11064-014-1377-0
Cymerman IA, Chung I, Beckmann BM, Bujnicki JM, Meiss G (2008) EXOG, a novel paralog of Endonuclease G in higher eukaryotes. Nuclei Acids Res 36:1369–1379. https://doi.org/10.1093/nar/gkm1169
Fujimoto M, Nakai A (2010) The heat shock factor family and adaptation to proteotoxic stress. FEBS J 277:4112–4125. https://doi.org/10.1111/j.1742-4658.2010.07827.x
George J, Jacobs HT (2019) Minimal effects of spargel (PGC-1) overexpression in a mitochondrial disease model. Biol Open 8(7):bio042135. https://doi.org/10.1242/bio.042135
Goldstein DS, Sullivan P, Cooney A, Jinsmaa Y, Kopin IJ, Sharabi Y (2015) Rotenone decreases intracellular aldehyde dehydrogenase activity: implications for the pathogenesis of Parkinson’s disease. J Neurochem 133:14–25. https://doi.org/10.1111/jnc.13042
Goo HG, Jung MK, Han SS, Rhim H, Kang S (2013) HtrA2/Omi deficiency causes damage and mutation of mitochondrial DNA. Biochim Biophys Acta 1833:1866–1875. https://doi.org/10.1016/j.bbamcr.2013.03.016
Grima B, Lamouroux A, Boni C, Julien JF, Javoy-Agid F, Mallet J (1987) A single human gene encoding multiple tyrosine hydroxylases with different predicted functional characteristics. Nature 326:707–711. https://doi.org/10.1038/326707a0
Jang JY, Blum A, Liu J, Finkel T (2018) The role of mitochondria in aging. J Clin Invest 128:3662–3670. https://doi.org/10.1172/jci120842
Kim SB, Heo JI, Kim H, Kim KS (2019) Acetylation of PGC1α by histone deacetylase 1 downregulation is implicated in radiation-induced senescence of brain endothelial cells. J Gerontol A Biol Sci Med Sci 74:787–793. https://doi.org/10.1093/gerona/gly167
Li Y et al (2018) Age-associated decline in Nrf2 signaling and associated mtDNA damage may be involved in the degeneration of the auditory cortex: implications for central presbycusis. Int J Mol Med 42:3371–3385. https://doi.org/10.3892/ijmm.2018.3907
Lu J et al (2018) Mitochondrial regulation by pyrroloquinoline quinone prevents rotenone-induced neurotoxicity in Parkinson’s disease models. Neurosci Lett 687:104–110. https://doi.org/10.1016/j.neulet.2018.09.031
Murphy MP, Hartley RC (2018) Mitochondria as a therapeutic target for common pathologies. Nat Rev Drug Discov 17:865–886. https://doi.org/10.1038/nrd.2018.174
Mursaleen L, Somavarapu S, Zariwala MG (2020) Deferoxamine and curcumin loaded nanocarriers protect against rotenone-induced neurotoxicity. J Parkinsons Dis 10:99–111. https://doi.org/10.3233/jpd-191754
Nicklas JA, Brooks EM, Hunter TC, Single R, Branda RF (2004) Development of a quantitative PCR (TaqMan) assay for relative mitochondrial DNA copy number and the common mitochondrial DNA deletion in the rat. Environ Mol Mutagen 44:313–320. https://doi.org/10.1002/em.20050
Nierenberg AA, Ghaznavi SA, Sande Mathias I, Ellard KK, Janos JA, Sylvia LG (2018) Peroxisome proliferator-activated receptor gamma coactivator-1 alpha as a novel target for bipolar disorder and other neuropsychiatric disorders. Biol Psychiatry 83:761–769. https://doi.org/10.1016/j.biopsych.2017.12.014
Ozbey G, Nemutlu-Samur D, Parlak H, Yildirim S, Aslan M, Tanriover G, Agar A (2020) Metformin protects rotenone-induced dopaminergic neurodegeneration by reducing lipid peroxidation. Pharmacol Rep 72:1397–1406. https://doi.org/10.1007/s43440-020-00095-1
Paradies G, Paradies V, Ruggiero FM, Petrosillo G (2019) Role of cardiolipin in mitochondrial function and dynamics in health and disease: molecular and pharmacological aspects. Cells 8 https://doi.org/10.3390/cells8070728
Peng K et al (2018) Mitochondrial ATP-sensitive potassium channel regulates mitochondrial dynamics to participate in neurodegeneration of Parkinson’s disease. Biochim Biophys Acta Mol Basis Dis 1864:1086–1103. https://doi.org/10.1016/j.bbadis.2018.01.013
Peng K et al (2017) The interaction of mitochondrial biogenesis and fission/fusion mediated by PGC-1α regulates rotenone-induced dopaminergic neurotoxicity. Mol Neurobiol 54:3783–3797. https://doi.org/10.1007/s12035-016-9944-9
Prieto I, Zambrano A, Laso J, Aranda A, Samper E, Monsalve M (2019) Early induction of senescence and immortalization in PGC-1α-deficient mouse embryonic fibroblasts. Free Radic Biol Med 138:23–32. https://doi.org/10.1016/j.freeradbiomed.2019.04.015
Quiros PM, Goyal A, Jha P, Auwerx J (2017) Analysis of mtDNA/nDNA ratio in mice. Curr Protoc Mouse Biol 7:47–54. https://doi.org/10.1002/cpmo.21
Rahman S, Copeland WC (2019) POLG-related disorders and their neurological manifestations. Nat Rev Neurol 15:40–52. https://doi.org/10.1038/s41582-018-0101-0
Reeve A, Meagher M, Lax N, Simcox E, Hepplewhite P, Jaros E, Turnbull D (2013) The impact of pathogenic mitochondrial DNA mutations on substantia nigra neurons. J Neurosci 33:10790–10801. https://doi.org/10.1523/jneurosci.3525-12.2013
Saki M, Prakash A (2017) DNA damage related crosstalk between the nucleus and mitochondria. Free Radic Biol Med 107:216–227. https://doi.org/10.1016/j.freeradbiomed.2016.11.050
Sanders LH et al (2014) Mitochondrial DNA damage: molecular marker of vulnerable nigral neurons in Parkinson’s disease. Neurobiol Dis 70:214–223. https://doi.org/10.1016/j.nbd.2014.06.014
Sas K, Szabó E, Vécsei L (2018) Mitochondria, oxidative stress and the kynurenine system, with a focus on ageing and neuroprotection. Molecules 23(1):191. https://doi.org/10.3390/molecules23010191
Selvakumar GP et al (2018) Glia maturation factor dependent inhibition of mitochondrial PGC-1α triggers oxidative stress-mediated apoptosis in N27 rat dopaminergic neuronal cells. Mol Neurobiol 55:7132–7152. https://doi.org/10.1007/s12035-018-0882-6
Sousa SC, Castilho RF (2005) Protective effect of melatonin on rotenone plus Ca2+-induced mitochondrial oxidative stress and PC12 cell death. Antioxid Redox Signal 7:1110–1116. https://doi.org/10.1089/ars.2005.7.1110
Suomalainen A, Battersby BJ (2018) Mitochondrial diseases: the contribution of organelle stress responses to pathology. Nat Rev Mol Cell Biol 19:77–92. https://doi.org/10.1038/nrm.2017.66
Szczesny B, Olah G, Walker DK, Volpi E, Rasmussen BB, Szabo C, Mitra S (2013) Deficiency in repair of the mitochondrial genome sensitizes proliferating myoblasts to oxidative damage. PLoS ONE 8:e75201. https://doi.org/10.1371/journal.pone.0075201
Tan K, Fujimoto M, Takii R, Takaki E, Hayashida N, Nakai A (2015) Mitochondrial SSBP1 protects cells from proteotoxic stresses by potentiating stress-induced HSF1 transcriptional activity. Nat Commun 6:6580. https://doi.org/10.1038/ncomms7580
Tann AW, Boldogh I, Meiss G, Qian W, Van Houten B, Mitra S, Szczesny B (2011) Apoptosis induced by persistent single-strand breaks in mitochondrial genome: critical role of EXOG (5’-EXO/endonuclease) in their repair. J Biol Chem 286:31975–31983. https://doi.org/10.1074/jbc.M110.215715
Tigchelaar W, De Jong AM, van Gilst WH, De Boer RA, Silljé HH (2016) In EXOG-depleted cardiomyocytes cell death is marked by a decreased mitochondrial reserve capacity of the electron transport chain. BioEssays S136–145. https://doi.org/10.1002/bies.201670914
Trifunovic A et al (2004) Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 429:417–423. https://doi.org/10.1038/nature02517
Xu SC et al (2011) Nickel exposure induces oxidative damage to mitochondrial DNA in Neuro2a cells: the neuroprotective roles of melatonin. J Pineal Res 51:426–433. https://doi.org/10.1111/j.1600-079X.2011.00906.x
Zemskov EA et al (2019) Biomechanical forces and oxidative stress: implications for pulmonary vascular disease. Antioxid Redox Signal 31:819–842. https://doi.org/10.1089/ars.2018.7720
Zhong Y et al (2011) Mitochondrial transcription factor A overexpression and base excision repair deficiency in the inner ear of rats with D-galactose-induced aging. FEBS J 278:2500–2510. https://doi.org/10.1111/j.1742-4658.2011.08176.x
Acknowledgments
The authors thank Dr. Sun Baofei and Mr. Yang Wang for technical assistance and statistics.
Funding
This work was supported by grants from the NSFC (Natural Science Foundation of China) (81973090, 81473006) to Yan Sai.
Author information
Authors and Affiliations
Contributions
Jingsong Xiao: Investigation, Writing-original draft, Visualization; Xunhu Dong and Kaige Peng: Investigation, Methodology, Software; Feng Ye, Jin Cheng and Guorong Dan: Formal analysis, Methodology, Software; Zhongmin Zou: Writing—review and editing, Supervision; Jia Cao and Yan Sai: Project administration, Conceptualization, Supervision, Validation, Writing—review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
Ethics approval was obtained from the ethics committee of Third Military Medical University of China.
Consent for Publication
I certify that this manuscript is original and has not been published and will not be submitted elsewhere for publication while being considered by Journal of Molecular Neuroscience. No data have been fabricated or manipulated to support our conclusions.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Rotenone induced mitochondrial DNA damage through reducing mitochondrial DNA repair ability.
• EXOG maintained mitochondrial DNA and mitochondrial function homeostasis in PC12 cells after rotenone exposure.
• EXOG interacted with HSF1-SSBP1complex and POLG.
• PGC-1α regulated the expression of EXOG, POLG and HSF1-SSBP1 complex.
Rights and permissions
About this article
Cite this article
Xiao, J., Dong, X., Peng, K. et al. PGC-1ɑ Mediated-EXOG, a Specific Repair Enzyme for Mitochondrial DNA, Plays an Essential Role in the Rotenone-Induced Neurotoxicity of PC12 Cells. J Mol Neurosci 71, 2336–2352 (2021). https://doi.org/10.1007/s12031-020-01775-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01775-6