Abstract
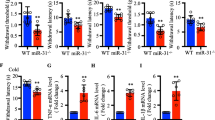
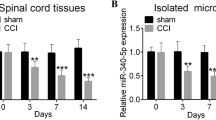
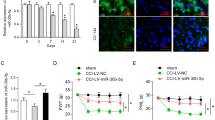
Recently, microRNAs are reported to be participated in the development of pain and persistence of neuropathic and inflammatory pain in animal models. Here, we characterized the functional role of miR-129-5p in pain processing in chronic constriction injury (CCI) rat models. Bilateral CCI operation was used to generate neuropathic pain rat model. Paw withdrawal threshold (PWT) and paw withdrawal latency (PWL) were used to assess pain-related behaviors. Gene expression was evaluated using qRT-PCR, luciferase assay, western blotting, and enzyme-linked immunosorbent assay. Compared with the control rats, expression level of miR-129-5p was downregulated significantly over time in CCI rats post operation. Interestingly, downregulation of miR-129-5p in CCI rats was correlated with increased proinflammatory cytokine expression and pain-related behaviors. Furthermore, we found that miR-129-5p alleviated neuropathic pain through downregulating high mobility group protein B1 (HMGB1) expression in CCI rats as overexpression of miR-129-5p suppressed expression of both HMGB1 and proinflammatory cytokine and alleviated pain sensation in CCI rats. In summary, our results show that alteration in miR-129-5p expression contributes to pain processing in our CCI pain rat model, suggesting miR-129-5p could be a causal factor in neuropathic pain and serve as a promising potential biomarker and therapeutic target for neuropathic pain.






Similar content being viewed by others
References
Agalave NM, Svensson CI (2015) Extracellular high-mobility group box 1 protein (HMGB1) as a mediator of persistent pain. Mol Med 20:569–578. https://doi.org/10.2119/molmed.2014.00176
Attal N (2000) Chronic neuropathic pain: mechanisms and treatment. Clin J Pain 16:S118–S130
Bai G, Ambalavanar R, Wei D, Dessem D (2007) Downregulation of selective microRNAs in trigeminal ganglion neurons following inflammatory muscle pain. Mol Pain 3:15. https://doi.org/10.1186/1744-8069-3-15
Bali KK, Hackenberg M, Lubin A, Kuner R, Devor M (2014) Sources of individual variability: miRNAs that predispose to neuropathic pain identified using genome-wide sequencing. Mol Pain 10:22. https://doi.org/10.1186/1744-8069-10-22
Bao Y, Wang S, Xie Y, Jin K, Bai Y, Shan S (2018) MiR-28-5p relieves neuropathic pain by targeting Zeb1 in CCI rat models. J Cell Biochem 119:8555–8563. https://doi.org/10.1002/jcb.27096
Blom SM, Pfister JP, Santello M, Senn W, Nevian T (2014) Nerve injury-induced neuropathic pain causes disinhibition of the anterior cingulate cortex. J Neurosci 34:5754–5764. https://doi.org/10.1523/JNEUROSCI.3667-13.2014
Bouhassira D, Lanteri-Minet M, Attal N, Laurent B, Touboul C (2008) Prevalence of chronic pain with neuropathic characteristics in the general population. Pain 136:380–387. https://doi.org/10.1016/j.pain.2007.08.013
Clark AK, Old EA, Malcangio M (2013) Neuropathic pain and cytokines: current perspectives. J Pain Res 6:803–814. https://doi.org/10.2147/JPR.S53660
Colloca L, Ludman T, Bouhassira D, Baron R, Dickenson AH, Yarnitsky D, Freeman R, Truini A, Attal N, Finnerup NB, Eccleston C, Kalso E, Bennett DL, Dworkin RH, Raja SN (2017) Neuropathic pain. Nat Rev Dis Primers 3:17002. https://doi.org/10.1038/nrdp.2017.2
Colvin LA, Dougherty PM (2015) Peripheral neuropathic pain: signs, symptoms, mechanisms, and causes: are they linked? Br J Anaesth 114:361–363. https://doi.org/10.1093/bja/aeu323
Dai Z, Chu H, Ma J, Yan Y, Zhang X, Liang Y (2018) The regulatory mechanisms and therapeutic potential of microRNAs: from chronic pain to morphine tolerance. Front Mol Neurosci 11:80. https://doi.org/10.3389/fnmol.2018.00080
DeLeo JA, Colburn RW, Nichols M, Malhotra A (1996) Interleukin-6-mediated hyperalgesia/allodynia and increased spinal IL-6 expression in a rat mononeuropathy model. J Interf Cytokine Res 16:695–700. https://doi.org/10.1089/jir.1996.16.695
Doyen PJ, Vergouts M, Pochet A, Desmet N, van Neerven S, Brook G, Hermans E (2017) Inflammation-associated regulation of RGS in astrocytes and putative implication in neuropathic pain. J Neuroinflammation 14:209. https://doi.org/10.1186/s12974-017-0971-x
Genda Y, Arai M, Ishikawa M, Tanaka S, Okabe T, Sakamoto A (2013) microRNA changes in the dorsal horn of the spinal cord of rats with chronic constriction injury: a TaqMan(R) Low Density Array study. Int J Mol Med 31:129–137. https://doi.org/10.3892/ijmm.2012.1163
Hung AL, Lim M, Doshi TL (2017) Targeting cytokines for treatment of neuropathic pain. Scand J Pain 17:287–293. https://doi.org/10.1016/j.sjpain.2017.08.002
Janssens R, Struyf S, Proost P (2018) The unique structural and functional features of CXCL12. Cell Mol Immunol 15:299–311. https://doi.org/10.1038/cmi.2017.107
Jiangpan P, Qingsheng M, Zhiwen Y, Tao Z (2016) Emerging role of microRNA in neuropathic pain. Curr Drug Metab 17:336–344
McCarberg BH, Billington R (2006) Consequences of neuropathic pain: quality-of-life issues and associated costs. Am J Manag Care 12:S263–S268
Tang S, Zhou J, Jing H, Liao M, Lin S, Huang Z, Huang T, Zhong J, HanbingWang (2019) Functional roles of lncRNAs and its potential mechanisms in neuropathic pain. Clin Epigenetics 11:78. https://doi.org/10.1186/s13148-019-0671-8
von Schack D, Agostino MJ, Murray BS, Li Y, Reddy PS, Chen J, Choe SE, Strassle BW, Li C, Bates B, Zhang L, Hu H, Kotnis S, Bingham B, Liu W, Whiteside GT, Samad TA, Kennedy JD, Ajit SK (2011) Dynamic changes in the microRNA expression profile reveal multiple regulatory mechanisms in the spinal nerve ligation model of neuropathic pain. PLoS One 6:e17670. https://doi.org/10.1371/journal.pone.0017670
Wei W, Wang H, Ji S (2017) Paradoxes of the EphB1 receptor in malignant brain tumors. Cancer Cell Int 17:21. https://doi.org/10.1186/s12935-017-0384-z
Xie X, Ma L, Xi K, Zhang W, Fan D (2017) MicroRNA-183 suppresses neuropathic pain and expression of AMPA receptors by targeting mTOR/VEGF signaling pathway. Cell Physiol Biochem 41:181–192. https://doi.org/10.1159/000455987
Yang D et al (2017) The role of miR-190a-5p contributes to diabetic neuropathic pain via targeting SLC17A6. J Pain Res 10:2395–2403. https://doi.org/10.2147/JPR.S133755
Yu SN, Liu GF, Li LY, Zhao GQ, Liu L, Li XF (2019) Analgesic effects of microRNA-129-5p against bone cancer pain through the EphB1/EphrinB2 signaling pathway in mice. J Cell Biochem 120:2876–2885. https://doi.org/10.1002/jcb.26605
Zempleni J, Baier SR, Howard KM, Cui J (2015) Gene regulation by dietary microRNAs. Can J Physiol Pharmacol 93:1097–1102. https://doi.org/10.1139/cjpp-2014-0392
Zeng J, Shen JP, Wang JT, Hu HW, Zhang CJ, Bai R, Zhang LM, He JZ (2018) Impacts of projected climate warming and wetting on soil microbial communities in Alpine grassland ecosystems of the Tibetan Plateau. Microb Ecol 75:1009–1023. https://doi.org/10.1007/s00248-017-1098-4
Zhang JM, An J (2007) Cytokines, inflammation, and pain. Int Anesthesiol Clin 45:27–37. https://doi.org/10.1097/AIA.0b013e318034194e
Zhang J, Zhang H, Zi T (2015) Overexpression of microRNA-141 relieves chronic constriction injury-induced neuropathic pain via targeting high-mobility group box 1. Int J Mol Med 36:1433–1439. https://doi.org/10.3892/ijmm.2015.2342
Zhang Y, Huang B, Wang HY, Chang A, Zheng XFS (2017) Emerging role of microRNAs in mTOR signaling. Cell Mol Life Sci 74:2613–2625. https://doi.org/10.1007/s00018-017-2485-1
Zhou Y, Ferguson J, Chang JT, Kluger Y (2007) Inter- and intra-combinatorial regulation by transcription factors and microRNAs. BMC Genomics 8:396. https://doi.org/10.1186/1471-2164-8-396
Funding
The study was supported by the Hebei Province Natural Fund Surface Project (H2017106022).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tian, J., Song, T., Wang, W. et al. miR-129-5p Alleviates Neuropathic Pain Through Regulating HMGB1 Expression in CCI Rat Models. J Mol Neurosci 70, 84–93 (2020). https://doi.org/10.1007/s12031-019-01403-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-019-01403-y