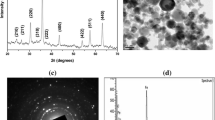
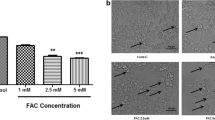
Abstract
Iron oxide (Fe2O3) nanoparticles (NPs) with its unique magnetic and paramagnetic properties are popular in biomedical applications. Some of their neurotoxic mechanisms due to repeated administration are proven. However, we speculate that the neuronal damage might be due to apoptosis resulting from unusual cell cycle entry. Moreover, iron accumulation has been shown to be closely associated with most of the neurodegenerative disorders. Thus, in the current study, mice were orally (po) treated with the Fe2O3-NPs to investigate cell cycle-associated events/components and occurrence of apoptosis. A subsequent increase in oxidant levels was observed with the iron accumulation due to Fe2O3-NPs exposure. The accumulated β-amyloid and reduced level of cdk5 seem to aid in the cell cycle entry and forcing progression towards apoptosis. Expression of Cyclin D1 and pRb (Ser 795) indicate the cell cycle re-entry of neurons. Overexpression of RNA Pol II and PARP cleavage suggests DNA damage due to Fe2O3-NPs exposure. Further, hyperphosphorylation of p38 (Thr 180/Tyr 182) confirms the activation of DNA damage-dependent checkpoint. Expression patterns of pro- and anti-apoptotic markers, TUNEL and TEM indicate the occurrences of apoptosis.








Similar content being viewed by others
References
Akashiba H, Ikegaya Y, Nishiyama N, Matsuki N (2008) Differential involvement of cell cycle reactivation between striatal and cortical neurons in cell death induced by 3-nitropropionic acid. J Biol Chem 283(10):6594–6606. https://doi.org/10.1074/jbc.M707730200
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1):276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bonda DJ, Bajic VP, Spremo-Potparevic B, Casadesus G, Zhu X, Smith MA, Lee HG (2010) Review: cell cycle aberrations and neurodegeneration. Neuropathol Appl Neurobiol 36(2):157–163. https://doi.org/10.1111/j.1365-2990.2010.01064.x
Bregman DB, Pestell RG, Kidd VJ (2000) Cell cycle regulation and RNA polymerase II. Front Biosci 5:244–257
Busca G (2006) The surface acidity and basicity of solid oxides and zeolites. In: Fierro JLG (ed) Metal oxides, chemistry and applications. CRC Press, pp 247–318
Chen MJ, Ng JM, Peng ZF, Manikandan J, Yap YW, Llanos RM, Beart PM, Cheung NS (2013) Gene profiling identifies commonalities in neuronal pathways in excitotoxicity: evidence favouring cell cycle re-activation in concert with oxidative stress. Neurochem Int 62(5):719–730. https://doi.org/10.1016/j.neuint.2012.12.015
Cuenda A, Rousseau S (2007) p38 MAP-kinases pathway regulation, function and role in human diseases. Biochim Biophys Acta 1773(8):1358–1375. https://doi.org/10.1016/j.bbamcr.2007.03.010
Czapski GA, Gąssowska M, Songin M, Radecka UD, Strosznajder JB (2011) Alterations of cyclin dependent kinase 5 expression and phosphorylation in amyloid precursor protein (APP) transfected PC12 cells. FEBS Lett 585(8):1243–1248. https://doi.org/10.1016/j.febslet.2011.03.058
Dhakshinamoorthy V, Manickam V, Perumal E (2017) Neurobehavioral toxicity of iron oxide nanoparticles in mice. Neurotox Res 32:187–203
Dissanayake NM, Current KM, Obare SO (2015) Mutagenic effects of iron oxide nanoparticles on biological cells. Int J Mol Sci 16(10):23482–23516. https://doi.org/10.3390/ijms161023482
Duguet E, Vasseur S, Mornet S, Devoisselle JM (2006) Magnetic nanoparticles and their applications in medicine. Nanomed 1(2):157–168. https://doi.org/10.2217/17435889.1.2.157
Folch J, Junyent F, Verdaguer E, Auladell C, Pizarro JG, Zarate CB, Pallas M, Camins A (2012) Role of cell cycle re-entry in neurons: a common apoptotic mechanism of neuronal cell death. Neurotox Res 22(3):195–207. https://doi.org/10.1007/s12640-011-9277-4
Frade JM, Benito MCO (2015) Neuronal cell cycle: the neuron itself and its circumstances. Cell Cycle 14(5):712–720. https://doi.org/10.1080/15384101.2015.1004937
Fu PP, Xia Q, Hwang H, Ray PC, Yu H (2014) Mechanisms of nanotoxicity: generation of reactive oxygen species. J Food Drug Anal 22(64–7):5
Genovese C, Trani D, Caputi M, Claudio Z (2006) Cell cycle control and beyond: emerging roles for the retinoblastoma gene family. Oncogene 25(38):5201–5209. https://doi.org/10.1038/sj.onc.1209652
Guo J, Sheng G, Warner BW (2005) EGF induced rapid Rb phosphorylation at Ser780 and Ser795 is mediated by ERK1/2 in small intestine epithelial cells. J Biol Chem 280(43):35992–35998. https://doi.org/10.1074/jbc.M504583200
Hagemeier J, Geurts JJ, Zivadinov R (2012) Brain iron accumulation in aging and neurodegenerative disorders. Expert Rev Neurother 12(12):1467–1480. https://doi.org/10.1586/ern.12.128
Ito A, Shinkai M, Honda H, Kobayashi T (2005) Medical application of functionalized magnetic nanoparticles. J Biosci Bioeng 100:1–11
Joaquin M, Gubern A, Posas F (2012) A novel G1 checkpoint mediated by the p57 CDK inhibitor and p38 SAPK promotes cell survival upon stress. Cell Cycle 11:3339–3350
Katsuda K, Kataoka M, Uno F, Murakami T, Kondo T, Roth JA, Tanaka N, Fujiwara T (2002) Activation of caspase-3 and cleavage of Rb are associated with p16-mediated apoptosis in human non-small cell lung cancer cells. Oncogene 21(13):2108–2123. https://doi.org/10.1038/sj.onc.1205272
Kawauchi S, Shikanai M, Kosodo Y (2013) Extra-cell cycle regulatory functions of cyclin-dependent kinases (CDK) and CDK inhibitor proteins contribute to brain development and neurological disorders. Genes Cells 18:176–194
Kruman II (2004) Why do neurons enters cell cycle. Cell Cycle 3(6):769–773
Kulkarni PV, Roney CA, Antich PP, Bonte FJ, Raghu AV, Aminabhavi TM (2010) Quinolinenbutylcyanoacrylate based nanoparticles for brain targeting for the diagnosis of Alzheimer’s disease. Wiley Interdisciplinary Rev Nanomed Nanobiotechnol 2(1):35–47. https://doi.org/10.1002/wnan.59
Lee EY, Hu N, Yuan SS, Cox LA, Bradley A, Lee WH, Herrup K (1994) Dual roles of the retinoblastoma protein in cell cycle regulation and neuron differentiation. Genes Dev 8:21–36
Li JJ, Zou L, Hartono D, Ong CN, Bay BH, Yung LY (2008) Gold nanoparticles induce oxidative damage in lung fibroblasts in vitro. Adv Mater 20(1):138–142. https://doi.org/10.1002/adma.200701853
Lopes JP, Oliveira CR, Agostinho P (2009) Cdk5 acts as a mediator of neuronal cell cycle re-entry triggered by amyloid-β and prion peptides. Cell Cycle 8(1):97–104. https://doi.org/10.4161/cc.8.1.7506
Mahmoudi M, Laurent S, Shokrgozar MA, Hosseinkhani M (2011) Toxicity evaluations of superparamagnetic iron oxide nanoparticles: cell “vision” versus physicochemical properties of nanoparticles. ACS Nano 5(9):7263–7276. https://doi.org/10.1021/nn2021088
Manickam V, Periyasamy M, Dhakshinamoorthy V, Panneerselvam L, Perumal P (2017) Recurrent exposure to ferric oxide nanoparticles alters myocardial oxidative stress, apoptosis and necrotic markers in male mice. Chem Biol Interact 278:54–64
McShea (2007) Neuronal cell cycle re-entry mediates Alzheimer disease-type changes. Biochim Biophys Acta 1772(4):467–472. https://doi.org/10.1016/j.bbadis.2006.09.010
Meikrantz W, Schlegel R (1995) Apoptosis and the cell cycle. J Cell Biochem 58(2):160–174. https://doi.org/10.1002/jcb.240580205
Modi PK, Jaiswal S, Sharma P (2016) Regulation of neuronal cell cycle and apoptosis by MicroRNA 34a. Mol Cell Biol 36:84–94
Naser R, Vandenbosch R, Omais S, Hayek D, Jaafar C, Lafi SA, Saliba A, Baghdadi M, Skaf L, Ghanem N (2016) Role of the retinoblastoma protein, Rb, during adult neurogenesis in the olfactory bulb. Sci Rep 6(1). https://doi.org/10.1038/srep20230
Park DS, Morris EJ, Bremner R, Keramaris E, Padmanabhan J, Rosenbaum M, Shelanski ML, Geller HM, Greene LA (2000) Involvement of retinoblastoma family members and E2F/DP complexes in the death of neurons evoked by DNA damage. J Neurosci 20(9):3104–3114
Patil US, Adireddy S, Jaiswal A, Mandava S, Lee BR, Chrisey DB (2015) In vitro/in vivo toxicity evaluation and quantification of iron oxide nanoparticles. Int J Mol Sci 16(10):24417–24450. https://doi.org/10.3390/ijms161024417
Pelegrí C, Duran-Vilaregut J, del Valle J, Crespo-Biel N, Ferrer I, Pallàs M, Camins A, Vilaplana J (2008) Cell cycle activation in striatal neurons from Huntington’s disease patients and rats treated with 3-nitropropionic acid. Int J Dev Neurosci 26(7):665–671. https://doi.org/10.1016/j.ijdevneu.2008.07.016
Pucci B, Kasten M, Giordano A (2000) Cell cycle and apoptosis. Neoplasia 2:291–299
Schwartz EI, Smilenov LB, Price MA, Osredkar T, Baker RA, Ghosh S, Shi F, Vollmer TL, Lencinas A, Stearns DM, Gorospe M, Kruman II (2007) Cell cycle activation in postmitotic neurons is essential for DNA repair. Cell Cycle 6(3):318–329. https://doi.org/10.4161/cc.6.3.3752
Stone JG, Siedlak SL, Tabaton M, Hirano A, Castellani RJ, Santocanale C, Perry G, Smith MA, Zhu X, Lee H (2011) The cell cycle regulator phosphorylated retinoblastoma protein is associated with tau pathology in several tauopathies. J Neuropathol Exp Neur 70(7):578–587. https://doi.org/10.1097/NEN.0b013e3182204414
Sundarraj K, Raghunath A, Panneerselvam L, Perumal E (2017) Iron oxide nanoparticles modulate heat shock proteins and organ specific markers in mice male accessory organs. Toxicol Appl Pharmacol 317:12–24. https://doi.org/10.1016/j.taap.2017.01.002
Tahirbegi IB, Pardo WA, Alvira M, Mir M, Samitier J (2016) Amyloid Aβ 42 a promoter of magnetite nanoparticle formation in Alzheimer’s disease. Nanotechnology 27(46):465102. https://doi.org/10.1088/0957-4484/27/46/465102
Thornton TM, Rincon M (2009) Non-classical P38 map kinase functions: cell cycle checkpoints and survival. Int J Biol Sci 5:44–52
Tyagi S, Herr W (2009) E2F1 mediates DNA damage and apoptosis through HCF1 and the MLL family of histone methyltransferases. EMBO J 28(20):3185–3195. https://doi.org/10.1038/emboj.2009.258
Vogel CF, Charrier JG, Wu D, McFall AS, Li W, Abid A, Kennedy IM, Anastasio C (2016) Physicochemical properties of iron oxide nanoparticles that contribute to cellular ROS-dependent signaling and acellular production of hydroxyl radical. Free Radic Res 50(11):1153–1164. https://doi.org/10.3109/10715762.2016.1152360
Wang B, Feng WY, Wang M, Shi JW, Zhang F, Ouyang H, Zhao YL, Chai ZF, Huang YY, Xie YN, Wang HF, Wang J (2007a) Transport of intranasally instilled fine Fe2O3 particles into the brain: micro-distribution, chemical states, and histopathological observation. Biol Trace Elem Res 118(3):233–243. https://doi.org/10.1007/s12011-007-0028-6
Wang J, Zhoua G, Chena C, Yu H, Wang T, Mad Y, Jia G, Gao Y, Li B, Suna J, Li Y, Jiao F, Zhao Y, Chai Z (2007b) Acute toxicity and bio distribution of different sized titanium dioxide particles in mice after oral administration. J. Toxicol Lett 168(2):176–185. https://doi.org/10.1016/j.toxlet.2006.12.001
Wang W, Bu B, Xie M, Zhang M, Yu Z, Tao D (2009) Neural cell cycle dysregulation and central nervous system diseases. Prog Neurobiol 89(1):1–17. https://doi.org/10.1016/j.pneurobio.2009.01.007
Winer JL, Liu CY, Apuzzo ML (2012) The use of nanoparticles as contrast media in neuroimaging: a statement on toxicity. World Neurosurg 78(6):709–711. https://doi.org/10.1016/j.wneu.2011.08.013
Wu J, Ding T, Sun J (2013) Neurotoxic potential of iron oxide nanoparticles in the rat brain striatum and hippocampus. Neurotoxicology 34:243–253. https://doi.org/10.1016/j.neuro.2012.09.006
Yarjanli Z, Ghaedi K, Esmaeili A, Rahgozar S, Zarrabi A (2017) Iron oxide nanoparticles may damage to the neural tissue through iron accumulation, oxidative stress, and protein aggregation. BMC Neurosci 18:51–67
Zhang J, Herrup K (2008) Cdk5 and the non-catalytic arrest of the neuronal cell cycle. Cell Cycle 7(22):3487–3490. https://doi.org/10.4161/cc.7.22.7045
Zhang J, Li H, Zhou T, Zhou J, Herrup K (2012) Cdk5 levels oscillate during the neuronal cell cycle: Cdh1 ubiquitination triggers proteosome-dependent degradation during S-phase. J Biol Chem 287:25985–25994
Zhu (2007) Oxidative imbalance in Alzheimer’s disease, apoptosis and the cell cycle. Mol Neurobiol 31:205–217
Acknowledgements
The authors would like to acknowledge Sophisticated Analytical Instrument Facility, All India Institute of Medical science (AIIMS), New Delhi, for the technical assistance in transmission electron microscopy. Vijayprakash Manickam acknowledges the UGC-BSR fellowship (UGC-BSR-No.F.7-25/2007) funded by UGC-BSR, New Delhi, India. We also thank the UGC-SAP DRS II (F-3-30/2013) and DST FIST (SR/FST/LSI-618/2014), New Delhi, India, for their partial financial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All the experiments were conducted in accordance with the ethical norms approved by Institutional Animal Ethical Committee (722/02/A/CPCSEA).
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in this study involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Manickam, V., Dhakshinamoorthy, V. & Perumal, E. Iron Oxide Nanoparticles Induces Cell Cycle-Dependent Neuronal Apoptosis in Mice. J Mol Neurosci 64, 352–362 (2018). https://doi.org/10.1007/s12031-018-1030-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1030-5