Abstract
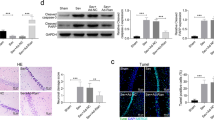
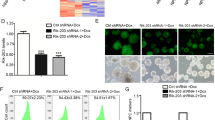
Postoperative cognitive dysfunction in elderly patients has been related to neurodegenerative disorders and mortality. Sevoflurane anesthesia has been implicated in both postoperative cognitive dysfunction and neurotoxicity. Given the advantages of using inhaled anesthetics like sevoflurane, it is important to understand how their usage results in neurotoxicity and subsequently devise ways to circumvent or attenuate the anesthetic-mediated induction in neurotoxicity. Long noncoding RNAs (LncRNAs) are a group of > 200 bp long RNAs and show specific spatiotemporal expression profiles. Several recent reports suggest that lncRNAs are involved in responses of the central nervous system (CNS) following acute injuries. However, their role in sevoflurane anesthesia-mediated cognitive dysfunction has not been studied. RNA immunoprecipitation (RIP) combined with qRT-PCR detection of six different lncRNAs showed that the HOTAIR lncRNAs were significantly more bound to both Sin3A and coREST, both corepressors of the RE-1 silencing transcription factor, within rat hippocampus following sevoflurane anesthesia compared with sham. Sevoflurane inhalation resulted in significant inhibition of brain-derived neurotrophic factor (BDNF) and cognitive impairment. Treatment with a combination of siRNAs targeting HOTAIR rescued BDNF expression and improved cognitive responses. Taken together, our results suggest that sevoflurane-mediated brain function impairment is at least in part mediated by the HOTAIR lncRNA.



Similar content being viewed by others
References
Abe Y, Kikuchi A, Kobayashi S, Wakusawa K, Tanaka S, Inui T, Kunishima S, Kure S, Haginoya K (2014) Xq26.1-26.2 gain identified on array comparative genomic hybridization in bilateral periventricular nodular heterotopia with overlying polymicrogyria. Dev Med Child Neurol 56(12):1221–1224. https://doi.org/10.1111/dmcn.12553
Barbash S, Simchovitz A, Buchman AS, Bennett DA, Shifman S, Soreq HL (2017) Neuronal-expressed microRNA-targeted pseudogenes compete with coding genes in the human brain. Transl Psychiatry 7(8):e1199. https://doi.org/10.1038/tp.2017.163
Bekker AY, Weeks EJ (2003) Cognitive function after anaesthesia in the elderly. Best Pract Res Clin Anaesthesiol 17(2):259–272. https://doi.org/10.1016/S1521-6896(03)00005-3
Bittner EA, Yue Y, Xie Z (2011) Brief review: anesthetic neurotoxicity in the elderly, cognitive dysfunction and Alzheimer’s disease. Can J Anaesth = J canadien d'anesthésie 58(2):216–223. https://doi.org/10.1007/s12630-010-9418-x
Bond AM, Vangompel MJ, Sametsky EA, Clark MF, Savage JC, Disterhoft JF, Kohtz JD (2009) Balanced gene regulation by an embryonic brain ncRNA is critical for adult hippocampal GABA circuitry. Nat Neurosci 12(8):1020–1027. https://doi.org/10.1038/nn.2371
Brosnan H, Bickler PE (2013) Xenon neurotoxicity in rat hippocampal slice cultures is similar to isoflurane and sevoflurane. Anesthesiology 119(2):335–344. https://doi.org/10.1097/ALN.0b013e31829417f0
Bussemakers MJ, Van BA, Verhaegh GW, Smit FP, Karthaus HF, Schalken JA, Debruyne FM, Ru N, Isaacs WB (1999) DD3: a new prostate-specific gene, highly overexpressed in prostate cancer. Cancer Res 59:5975
Cabili MN, Trapnell C, Goff L, Koziol M, Tazon-Vega B, Regev A, Rinn JL (2011) Integrative annotation of human large intergenic noncoding RNAs reveals global properties and specific subclasses. Genes Dev 25(18):1915–1927. https://doi.org/10.1101/gad.17446611
Castrén E (2004) Neurotrophic effects of antidepressant drugs. Curr Opin Pharmacol 4(1):58–64. https://doi.org/10.1016/j.coph.2003.10.004
Erden V, Erkalp K, Yangin Z, Delatioglu H, Kiroglu S, Ortakuz S, Ozdemir B (2011) The effect of labor on sevoflurane requirements during cesarean delivery. Int J Obstet Anesth 20(1):17–21. https://doi.org/10.1016/j.ijoa.2010.08.008
Ernfors P, Lee KF, Jaenisch R (1994) Mice lacking brain-derived neurotrophic factor develop with sensory deficits. Nature 368(6467):147–150. https://doi.org/10.1038/368147a0
Gupta RA, Shah N, Wang KC, Kim J, Horlings HM, Wong DJ, Tsai MC, Hung T, Argani P, Rinn JL (2010) Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 464(7291):1071–1076. https://doi.org/10.1038/nature08975
Guttman M, Donaghey J, Carey BW, Garber M, Grenier JK, Munson G, Young G, Lucas AB, Ach R, Bruhn L (2011) lincRNAs act in the circuitry controlling pluripotency and differentiation. Nature 477(7364):295–300. https://doi.org/10.1038/nature10398
Hall J, Thomas KL, Everitt BJ (2000) Rapid and selective induction of BDNF expression in the hippocampus during contextual learning. Nat Neurosci 3(6):533–535. https://doi.org/10.1038/75698
Huang EJ, Reichardt LF (2001) NEUROTROPHINS: roles in neuronal development and Function1. Annu Rev Neurosci 24(1):677–736. https://doi.org/10.1146/annurev.neuro.24.1.677
Huarte M, Guttman M, Feldser D, Garber M, Koziol MJ, Kenzelmannbroz D, Khalil AM, Zuk O, Amit I, Rabani M (2010) A large intergenic noncoding RNA induced by p53 mediates global gene repression in the p53 response. Cell 142(3):409–419. https://doi.org/10.1016/j.cell.2010.06.040
Iacoangeli A, Lin Y, Morley EJ, Muslimov IA, Bianchi R, Reilly J, Weedon J, Diallo R, Böcker W, Tiedge H (2004) BC200 RNA in invasive and preinvasive breast cancer. Carcinogenesis 25(11):2125–2133. https://doi.org/10.1093/carcin/bgh228
Iversen LL, Glowinski J (1966) Regional differences in the rate of turnover of norepinephrine in the rat brain. Nature 210(5040):1006–1008. https://doi.org/10.1038/2101006a0
Johnson R (2012) Long non-coding RNAs in Huntington’s disease neurodegeneration. Neurobiol Dis 46(2):245–254. https://doi.org/10.1016/j.nbd.2011.12.006
Kesslak JP, So V, Choi J, Cotman CW, Gomezpinilla F (1998) Learning upregulates brain-derived neurotrophic factor messenger ribonucleic acid: a mechanism to facilitate encoding and circuit maintenance? Behav Neurosci 112(4):1012–1019. https://doi.org/10.1037/0735-7044.112.4.1012
Kobayashi K, Takemori K, Sakamoto A (2007) Circadian gene expression is suppressed during sevoflurane anesthesia and the suppression persists after awakening. Brain Res 1185:1–7. https://doi.org/10.1016/j.brainres.2007.09.011
Korte M, Carroll P, Wolf E, Brem G, Thoenen H, Bonhoeffer T (1995) Hippocampal long-term potentiation is impaired in mice lacking brain-derived neurotrophic factor. Proc Natl Acad Sci U S A 92(19):8856–8860. https://doi.org/10.1073/pnas.92.19.8856
Liang G, Ward C, Peng J, Zhao Y, Huang B, Wei H (2010) Isoflurane causes greater neurodegeneration than an equivalent exposure of sevoflurane in the developing brain of neonatal mice. Anesthesiology 112(6):1325–1334. https://doi.org/10.1097/ALN.0b013e3181d94da5
Lopez Gil ML, Brimacombe J, Clar B (1999) Sevoflurane versus propofol for induction and maintenance of anaesthesia with the laryngeal mask airway in children. Paediatr Anaesth 9(6):485–490. https://doi.org/10.1046/j.1460-9592.1999.00404.x
Maisonpierre PC, Belluscio L, Friedman B, Alderson RF, Wiegand SJ, Furth ME, Lindsay RM, Yancopoulos GD (1990) NT-3, BDNF, and NGF in the developing rat nervous system: parallel as well as reciprocal patterns of expression. Neuron 5(4):501–509. https://doi.org/10.1016/0896-6273(90)90089-X
Mizuno M, Yamada K, Olariu A, Nawa H, Nabeshima T (2000) Involvement of brain-derived neurotrophic factor in spatial memory formation and maintenance in a radial arm maze test in rats. J Neurosci 20:7116
Moller JT, Cluitmans P, Rasmussen LS, Houx P, Rasmussen H, Canet J, Rabbitt P, Jolles J, Larsen K, Hanning CD (1998) Long-term postoperative cognitive dysfunction in the elderly ISPOCD1 study. ISPOCD investigators. International Study of Post-Operative Cognitive Dysfunction. Lancet 351:857
Nagano T, Fraser P (2011) No-nonsense functions for long noncoding RNAs. Cell 145(2):178–181. https://doi.org/10.1016/j.cell.2011.03.014
Nagano T, Mitchell JA, Sanz LA, Pauler FM, Fergusonsmith AC, Feil R, Fraser P (2008) The air noncoding RNA epigenetically silences transcription by targeting G9a to chromatin. Science 322(5908):1717–1720. https://doi.org/10.1126/science.1163802
Pandey RR, Kanduri C (2011) Transcriptional and posttranscriptional programming by long noncoding RNAs. Springer, Berlin Heidelberg
Perez DS, Hoage TR, Pritchett JR, Ducharmesmith AL, Halling ML, Ganapathiraju SC, Streng PS, Smith DI (2008) Long, abundantly expressed non-coding transcripts are altered in cancer. Hum Mol Genet 17(5):642–655. https://doi.org/10.1093/hmg/ddm336
Petrovics G, Zhang W, Makarem M, Street JP, Connelly R, Sun L, Sesterhenn IA, Srikantan V, Moul JW, Srivastava S (2004) Elevated expression of PCGEM1, a prostate-specific gene with cell growth-promoting function, is associated with high-risk prostate cancer patients. Oncogene 23(2):605–611. https://doi.org/10.1038/sj.onc.1207069
Qureshi IA, Mehler MF (2011) Non-coding RNA networks underlying cognitive disorders across the lifespan. Trends Mol Med 17(6):337–346. https://doi.org/10.1016/j.molmed.2011.02.002
Rasmussen LS, Johnson T, Kuipers HM, Kristensen D, Siersma VD, Vila P, Jolles J, Papaioannou A, Abildstrom H, Silverstein JH (2003) Does anaesthesia cause postoperative cognitive dysfunction? A randomised study of regional versus general anaesthesia in 438 elderly patients. Acta Anaesthesiol Scand 47(3):260–266. https://doi.org/10.1034/j.1399-6576.2003.00057.x
Ray PS, Wang J, Qu Y, Sim MS, Shamonki J, Bagaria SP, Ye X, Liu B, Elashoff D, Hoon DS (2010) FOXC1 is a potential prognostic biomarker with functional significance in basal-like breast cancer. Cancer Res 70(10):3870–3876. https://doi.org/10.1158/0008-5472.CAN-09-4120
Salta E (2012) Non-coding RNAs with essential roles in neurodegenerative disorders. Lancet Neurol 11(2):189–200. https://doi.org/10.1016/S1474-4422(11)70286-1
Satomoto M, Satoh Y, Terui K, Miyao H, Takishima K, Ito M, Imaki J (2009) Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in fear conditioning in mice. Anesthesiology 110(3):628–637. https://doi.org/10.1097/ALN.0b013e3181974fa2
Sauvageau M, Goff LA, Lodato S, Bonev B, Groff AF, Gerhardinger C, Sanchez-Gomez DB, Hacisuleyman E, Li E, Spence M, Liapis SC, Mallard W, Morse M, Swerdel MR, D'Ecclessis MF, Moore JC, Lai V, Gong G, Yancopoulos GD, Frendewey D, Kellis M, Hart RP, Valenzuela DM, Arlotta P, Rinn JL (2013) Multiple knockout mouse models reveal lincRNAs are required for life and brain development. elife 2:e01749
Silbert B, Evered L, Scott DA, Maruff P (2011) Anesthesiology must play a greater role in patients with Alzheimer’s disease. Anesth Analg 112(5):1242–1245. https://doi.org/10.1213/ANE.0b013e3182147f5b
Srikantan V, Zou Z, Petrovics G, Xu L, Augustus M, Davis L, Livezey JR, Connell T, Sesterhenn IA, Yoshino K (2000) PCGEM1, a prostate-specific gene, is overexpressed in prostate cancer. Proc Natl Acad Sci U S A 97(22):12216–12221. https://doi.org/10.1073/pnas.97.22.12216
Steinmetz J, Christensen KB, Lund T, Lohse N, Rasmussen LS, Group, I (2009) Long-term consequences of postoperative cognitive dysfunction. Anesthesiology 110(3):548–555. https://doi.org/10.1097/ALN.0b013e318195b569
Stiegler MP, Tung A (2014) Cognitive processes in anesthesiology decision making. Anesthesiology 120(1):204–217. https://doi.org/10.1097/ALN.0000000000000073
Xiao H, Liu B, Chen Y, Zhang J (2016) Learning, memory and synaptic plasticity in hippocampus in rats exposed to sevoflurane. Int J Dev Neurosci 48:38. https://doi.org/10.1016/j.ijdevneu.2015.11.001
Xiong WX, Zhou GX, Wang B, Xue ZG, Wang L, Sun HC, Ge SJ (2013) Impaired spatial learning and memory after sevoflurane-nitrous oxide anesthesia in aged rats is associated with down-regulated cAMP/CREB signaling. PLoS One 8(11):e79408. https://doi.org/10.1371/journal.pone.0079408
Yee BK, Zhu SW, Mohammed AH, Feldon J (2007) Levels of neurotrophic factors in the hippocampus and amygdala correlate with anxiety- and fear-related behaviour in C57BL6 mice. J Neural Transm 114(4):431–444. https://doi.org/10.1007/s00702-006-0548-9
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures were approved by the Local Ethics Committee at Binzhou People’s Hospital.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, Jy., Feng, Y., Fu, Yh. et al. Effect of Sevoflurane Anesthesia on Brain Is Mediated by lncRNA HOTAIR. J Mol Neurosci 64, 346–351 (2018). https://doi.org/10.1007/s12031-018-1029-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1029-y