Abstract
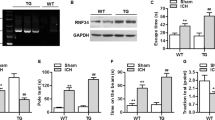
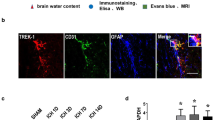
Intracranial hemorrhage (ICH) is a common subtype of stroke with high morbidity and mortality. However, few clinical therapies that can reduce ICH-induced brain injury and promote the recovery outcome in ICH patients are available to improve the recovery from ICH. Given that aquaporin 9 (AQP9) plays a critical role in brain edema after ischemic stroke and traumatic brain injury and is involved in the regulation of angiogenesis, we examined the role of AQP9 in preventing neuronal loss and in neovascularization in the dorsal hippocampus (DH) after ICH. We found that intra-DH collagenase-induced ICH increased AQP9 protein levels in the hippocampus, which was associated with behavioral deficits in wild-type mice. However, ICH robustly enhanced behavioral deficits in the AQP9-null mice, as compared with the wild-type mice. Furthermore, neovascularization and proliferation of brain microvascular endothelial cells following ICH were severely impaired in the AQP9-null mice, as compared with the wild-type mice. Finally, hippocampal neuronal loss following ICH became severer in the AQP9-null mice, relative to the wild-type mice. Taken together, our findings indicated that AQP9 in the brain may play a compensatory role in response to ICH, promote brain angiogenesis, and prevent subsequent neuronal death, thus preventing the deterioration of neurological outcome of ICH.





Similar content being viewed by others
References
Arima H, Yamamoto N, Sobue K, Umenishi F, Tada T, Katsuya H, Asai K (2003) Hyperosmolar mannitol simulates expression of aquaporins 4 and 9 through a p38 mitogen-activated protein kinase-dependent pathway in rat astrocytes. J Biol Chem 278:44525–44534. doi:10.1074/jbc.M304368200
Badaut J (2010) Aquaglyceroporin 9 in brain pathologies. Neuroscience 168:1047–1057. doi:10.1016/j.neuroscience.2009.10.030
Badaut J, Hirt L, Granziera C, Bogousslavsky J, Magistretti PJ, Regli L (2001) Astrocyte-specific expression of aquaporin-9 in mouse brain is increased after transient focal cerebral ischemia. J Cereb Blood Flow Metab 21:477–482. doi:10.1097/00004647-200105000-00001
Badaut J, Petit JM, Brunet JF, Magistretti PJ, Charriaut-Marlangue C, Regli L (2004) Distribution of aquaporin 9 in the adult rat brain: preferential expression in catecholaminergic neurons and in glial cells. Neuroscience 128:27–38. doi:10.1016/j.neuroscience.2004.05.042
Broderick J et al (2007) Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke 38:2001–2023. doi:10.1161/STROKEAHA.107.183689
Camerino GM, Nicchia GP, Dinardo MM, Ribatti D, Svelto M, Frigeri A (2006) In vivo silencing of aquaporin-1 by RNA interference inhibits angiogenesis in the chick embryo chorioallantoic membrane assay. Cell Mol Biol (Noisy-le-grand) 52:51–56
El Hindy N et al (2013) Correlation of aquaporin-1 water channel protein expression with tumor angiogenesis in human astrocytoma. Anticancer Res 33:609–613
Elkjaer M et al (2000) Immunolocalization of AQP9 in liver, epididymis, testis, spleen, and brain. Biochem Biophys Res Commun 276:1118–1128. doi:10.1006/bbrc.2000.3505
Fu X, Li Q, Feng Z, Mu D (2007) The roles of aquaporin-4 in brain edema following neonatal hypoxia ischemia and reoxygenation in a cultured rat astrocyte model. Glia 55:935–941. doi:10.1002/glia.20515
Griesdale DE, Honey CR (2004) Aquaporins and brain edema. Surg Neurol 61:418–421. doi:10.1016/j.surneu.2003.10.047
Guo Q, Sayeed I, Baronne LM, Hoffman SW, Guennoun R, Stein DG (2006) Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp Neurol 198:469–478. doi:10.1016/j.expneurol.2005.12.013
Huebert RC et al (2010) Aquaporin-1 facilitates angiogenic invasion in the pathological neovasculature that accompanies cirrhosis. Hepatology 52:238–248. doi:10.1002/hep.23628
Hwang IK, Yoo KY, Li H, Lee BH, Suh HW, Kwon YG, Won MH (2007) Aquaporin 9 changes in pyramidal cells before and is expressed in astrocytes after delayed neuronal death in the ischemic hippocampal CA1 region of the gerbil. J Neurosci Res 85:2470–2479. doi:10.1002/jnr.21381
Kaneko K et al (2008) Aquaporin 1 is required for hypoxia-inducible angiogenesis in human retinal vascular endothelial cells. Microvasc Res 75:297–301. doi:10.1016/j.mvr.2007.12.003
Kleffner I, Bungeroth M, Schiffbauer H, Schabitz WR, Ringelstein EB, Kuhlenbaumer G (2008) The role of aquaporin-4 polymorphisms in the development of brain edema after middle cerebral artery occlusion. Stroke 39:1333–1335. doi:10.1161/STROKEAHA.107.500785
Liu H, Yang M, Qiu GP, Zhuo F, Yu WH, Sun SQ, Xiu Y (2012) Aquaporin 9 in rat brain after severe traumatic brain injury. Arq Neuropsiquiatr 70:214–220
National Research Council (2010) Guide for the Care and Use of Laboratory Animals. The National Academies Press, Washington, DC 1–248
Papadopoulos MC, Krishna S, Verkman AS (2002) Aquaporin water channels and brain edema. Mt Sinai J Med 69:242–248
Papadopoulos MC, Verkman AS (2007) Aquaporin-4 and brain edema. Pediatr Nephrol 22:778–784. doi:10.1007/s00467-006-0411-0
Qureshi AI, Mendelow AD, Hanley DF (2009) Intracerebral haemorrhage. Lancet 373:1632–1644. doi:10.1016/S0140-6736(09)60371-8
Qureshi AI et al (2005) A prospective multicenter study to evaluate the feasibility and safety of aggressive antihypertensive treatment in patients with acute intracerebral hemorrhage. J Intensive Care Med 20:34–42. doi:10.1177/0885066604271619
Ribeiro Mde C, Hirt L, Bogousslavsky J, Regli L, Badaut J (2006) Time course of aquaporin expression after transient focal cerebral ischemia in mice. J Neurosci Res 83:1231–1240. doi:10.1002/jnr.20819
Rosenberg GA, Mun-Bryce S, Wesley M, Kornfeld M (1990) Collagenase-induced intracerebral hemorrhage in rats. Stroke 21:801–807
Saadoun S, Papadopoulos MC, Hara-Chikuma M, Verkman AS (2005) Impairment of angiogenesis and cell migration by targeted aquaporin-1 gene disruption. Nature 434:786–792. doi:10.1038/nature03460
Schurr A (2002) Lactate, glucose and energy metabolism in the ischemic brain (review). Int J Mol Med 10:131–136
Sobue K, Asai K, Katsuya H (2006) Aquaporin water channels in the brain and molecular mechanisms of brain edema. Nihon Rinsho 64:1181–1189
Sulyok E, Vajda Z, Doczi T, Nielsen S (2004) Aquaporins and the central nervous system. Acta Neurochir 146:955–960. doi:10.1007/s00701-004-0319-z
Unterberg AW, Stover J, Kress B, Kiening KL (2004) Edema and brain trauma. Neuroscience 129:1021–1029. doi:10.1016/j.neuroscience.2004.06.046
Vacca A et al (2001a) Microvessel overexpression of aquaporin 1 parallels bone marrow angiogenesis in patients with active multiple myeloma. Br J Haematol 113:415–421
Vacca A, Ribatti D, Roccaro AM, Frigeri A, Dammacco F (2001b) Bone marrow angiogenesis in patients with active multiple myeloma. Semin Oncol 28:543–550
Vacca A, Ribatti D, Roccaro AM, Ria R, Palermo L, Dammacco F (2001c) Bone marrow angiogenesis and plasma cell angiogenic and invasive potential in patients with active multiple myeloma. Acta Haematol 106:162–169
Venero JL, Machado A, Cano J (2004) Importance of aquaporins in the physiopathology of brain edema. Curr Pharm Des 10:2153–2161
Venero JL, Vizuete ML, Machado A, Cano J (2001) Aquaporins in the central nervous system. Prog Neurobiol 63:321–336
Wagner KR, Sharp FR, Ardizzone TD, Lu A, Clark JF (2003) Heme and iron metabolism: role in cerebral hemorrhage. J Cereb Blood Flow Metab 23:629–652. doi:10.1097/01.WCB.0000073905.87928.6D
Wang BF, Cui ZW, Zhong ZH, Sun YH, Sun QF, Yang GY, Bian LG (2015) Curcumin attenuates brain edema in mice with intracerebral hemorrhage through inhibition of AQP4 and AQP9 expression. Acta Pharmacol Sin 36:939–948. doi:10.1038/aps.2015.47
Wei X et al (2015) Phosphorylation of p38 MAPK mediates aquaporin 9 expression in rat brains during permanent focal cerebral ischaemia. J Mol Histol 46:273–281. doi:10.1007/s10735-015-9618-3
Xi G, Keep RF, Hoff JT (2006) Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol 5:53–63. doi:10.1016/S1474-4422(05)70283-0
Yang M et al (2013) Hyperosmotic induction of aquaporin expression in rat astrocytes through a different MAPK pathway. J Cell Biochem 114:111–119. doi:10.1002/jcb.24308
Yool AJ (2007) Aquaporins: multiple roles in the central nervous system. Neuroscientist 13:470–485. doi:10.1177/1073858407303081
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal experiments were approved by the Institutional Animal Care and Use Committee of Wuxi No. 2 Hospital. The housing and treatment of the mice followed the guidelines of the “Guide for the Care and Use of Laboratory Animals” (Institute of Laboratory Animal Resources, Commission on Life Sciences 2011). All efforts were made to minimize suffering.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Ji, W., Wang, J., Xu, J. et al. Lack of Aquaporin 9 Reduces Brain Angiogenesis and Exaggerates Neuronal Loss in the Hippocampus Following Intracranial Hemorrhage in Mice. J Mol Neurosci 61, 351–358 (2017). https://doi.org/10.1007/s12031-016-0862-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-016-0862-0