Abstract
Purpose
This phase II single-institution trial of adjuvant thalidomide after cytoreductive surgery (CS) and hyperthermic intraperitoneal chemotherapy (HIPEC) for patients with appendiceal and colorectal malignancies sought to detect an improvement in progression-free survival (PFS) from 7 to 12 months.
Methods
Eligible patients received CS, HIPEC, and baseline imaging, followed by pretreatment thalidomide counseling. All participants were then started on a 28-day regimen of thalidomide, 100 mg by mouth at bedtime, followed by 200 mg for 4 weeks, followed by 300 mg as the final maintenance dose, as tolerated.
Results
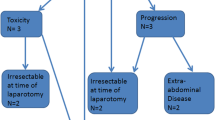
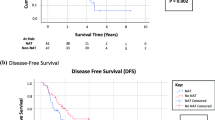
Twenty-seven eligible patients (median age 52 years; 52 % appendiceal/48 % colorectal) were enrolled on this trial and included in the analysis, and 26 were evaluable for response. Eighteen patients demonstrated stable disease on adjuvant thalidomide, while eight showed evidence of progression. Approximately 30 % of the patients withdrew due to toxicity. Grade 3/4 toxicities included neurological disorders (16 %), nausea (12 %), vomiting (8 %), and thromboembolism (8 %). Median overall survival (OS) and PFS were 43.0 and 9.3 months, respectively, and median follow-up was 40.4 months. Multivariate modeling showed significant improvements in PFS and OS for appendiceal patients and those with R0 or R1 resections. On an intent-to-treat analysis, the PFS of the study group was 9 months.
Conclusions
Based on these findings, thalidomide cannot be recommended as adjuvant therapy after CS and HIPEC for gastrointestinal malignancies. Further research is needed to identify active agents in this population.



Similar content being viewed by others
References
Dawson LE, Russell AH, Tong D, et al. Adenocarcinoma of the sigmoid colon: sites of initial dissemination and clinical patterns of recurrence following surgery alone. J Surg Oncol. 1983;22:95–9.
Chu DZ, Lang NP, Thompson C, et al. PSD in nongynecologic malignancy: a prospective study of prognostic factors. Cancer. 1989;63:3643–67.
Sugarbaker PH, Cunliffe WJ, Belliveau J, et al. Rationale for integrating early postoperative intraperitoneal chemotherapy into the surgical treatment of gastrointestinal cancer. Semin Oncol. 1989;16(4 Suppl 6):83–97.
Sugarbaker PH, Landy D, Jaffe G, et al. Histologic changes induced by intraperitoneal chemotherapy with 5-fluorouracil and mitomycin C in patients with PSD from cystadenocarcinoma of the colon or appendix. Cancer. 1990;65:1495–501.
Stewart 4th JH, Shen P, Russell GB, et al. Appendiceal neoplasms with peritoneal dissemination: outcomes after cytoreductive surgery and intraperitoneal hyperthermic chemotherapy. Ann Surg Oncol. 2006;13(5):624–34.
Shen P, Stewart 4th JH, Levine EA. The role of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for metastatic colorectal cancer with peritoneal surface disease. Curr Probl Cancer. 2009;33:154–67.
Folkman J. Clinical applications of research on angiogenesis. N Engl J Med. 1995;333(26):1757–63.
Vile R. Cancer therapy. Less blood means more sanguinity. Curr Biol. 1995;5(1):10–3.
Bikfalvi A. Significance of angiogenesis in tumour progression and metastasis. Eur J Cancer. 1995;31A(7/8):1101–4.
Or R, Feferman R, Shoshan S. Thalidomide reduces vascular density in granulation tissue of subcutaneously implanted polyvinyl alcohol sponges in guinea pigs. Exp Hematol. 1998;26:217–21.
Kenyon B, Browne F, D’Amato R. Effects of thalidomide and related metabolites in a mouse corneal model of neovascularization. Exp Eye Res. 1997;64:971–8.
Stewart 4th JH, Shen P, Levine EA. Intraperitoneal hyperthermic chemotherapy for peritoneal surface malignancy: current status and future directions. Ann Surg Oncol. 2005;12:765–77.
Dindo D, Demartines N, Calvien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Glehen O, Kwiatkowski F, Sugarbaker PH, et al. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of PSD from colorectal cancer: a multi-institutional study. J Clin Oncol. 2004;22(16):3284–92.
Sadeghi B, Arvieux C, Glehen O, et al. PSD from non-gynecologic malignancies: results of the EVOCAPE 1 multicentric prospective study. Cancer. 2000;88:358–63.
Verwaal VJ, Van Ruth S, de Bree E, et al. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with PSD of colorectal cancer. J Clin Oncol. 2003;21:3737–43.
Abramson N, Stokes PK, Luke M, et al. Ovarian and papillary-serous peritoneal carcinoma: pilot study with thalidomide. J Clin Oncol. 2002;20:1147–9.
Chan JK, Manuel MR, Ciaravino G, et al. Safety and efficacy of thalidomide in recurrent epithelial ovarian and peritoneal carcinoma. Gynecol Oncol. 2006;103:919–23.
Lago LD, Richter MF, Cancela AI, et al. Phase II trial and pharmacokinetic study of thalidomide in patients with metastatic colorectal cancer. Invest New Drugs. 2003;21:359–66.
Palumbo A, Facon T, Sonneveld P, et al. Thalidomide for the treatment of multiple myeloma: 10 years later. Blood. 2008;111:3968–77.
Kaushal V, Kaushal GP, Melkaveri SN, et al. Thalidomide protects endothelial cells from doxorubicin-induced apoptosis but alters cell morphology. J Thromb Haemost. 2004;2:327–34.
Rajkumar SV, Gertz MA, Lacy MQ, et al. Thalidomide as initial therapy for early-stage myeloma. Leukemia. 2003;17:775–9.
Ronnett BM, Yan H, Sugarbaker PH, et al. Patients with pseudomyxoma peritonei associated with disseminated peritoneal adenomucinosis have a significantly more favorable prognosis than patients with peritoneal mucinous surface disease. Cancer. 2001;92(1):85–91.
Yan TD, Black D, Savady R, et al. Systematic review on the efficacy of cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for PSD from colorectal carcinoma. J Clin Oncol. 2006;24:4011–9.
Levine EA, Stewart 4th JH, Russell GB, et al. Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy for peritoneal surface malignancy: experience with 501 procedures. J Am Coll Surg. 2007;204:943–55.
Shen P, Hawksworth J, Lovato J, et al. Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy with mitomycin C for peritoneal carcinomatosis from nonappendiceal colorectal carcinoma. Ann Surg Oncol. 2004;11:178–86.
O’Neil BH, Goldberg RM. Innovations in chemotherapy for metastatic colorectal cancer: an update of recent clinical trials. Oncologist. 2008;13:1074–83.
Franko J, Shi Q, Goldman CD, et al. Treatment of colorectal peritoneal carcinomatosis with systemic chemotherapy: a pooled analysis of north central cancer treatment group phase III trials N9741 and N9841. J Clin Oncol. 2012;30:263–7.
Allegra CJ, Yothers G, O’Connell MJ, et al. Phase III trial assessing bevacizumab in stages II and III carcinoma of the colon: results of NSABP protocol C-08. J Clin Oncol. 2011;29:11–6.
Acknowledgments
The authors would like to acknowledge the Celgene Corporation for their sponsorship of this clinical trial by providing the drug and funding for this study. The final manuscript was reviewed and approved for publication by the Celgene Corporation.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, P., Thomas, C.R., Fenstermaker, J. et al. Phase II Trial of Adjuvant Oral Thalidomide Following Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy for Peritoneal Surface Disease from Colorectal/Appendiceal Cancer. J Gastrointest Canc 45, 268–275 (2014). https://doi.org/10.1007/s12029-014-9578-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-014-9578-y