Abstract
Purpose
Whether autophagy plays a key role in thyroxine-induced cardiomyocyte hypertrophy, and whether the role of autophagy in thyroxine-induced cardiomyocyte hypertrophy is related to targeting of Beclin-1 by miR-762 remains unclear. This research focused on testing these two hypotheses. Importantly, the results of this study will help us better understand the molecular mechanisms of thyroxine-induced cardiomyocyte hypertrophy.
Methods
In vivo and in vitro, RT-PCR, western blot, and dual luciferase reporter assay were performed to understand the molecular mechanism of thyroxine-induced cardiomyocyte hypertrophy. HE staining, Masson staining, transmission electron microscopy, and immunofluorescence were used to observe intuitively changes of hearts and cardiomyocytes.
Results
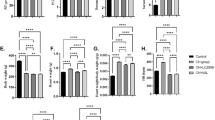
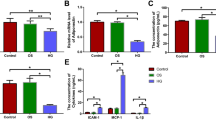
Our results showed that in vivo, serum TT3, TT4, and heart rate were significantly upregulated in the T4 group compared with the control group. Moreover, the surface area of cardiomyocytes was significantly increased in the T4 group, and the structural disorder was accompanied by obvious hyperplasia of collagen fibers. The expression of ANP, and β-MHC was significantly upregulated in the T4 group. In addition, LC3 II/LC3 I, Beclin-1 and the count of autophagic vacuoles were significantly upregulated, but miR-762 was significantly downregulated in the T4 group compared to the control group. Subsequently, a dual luciferase reporter assay suggested that Beclin-1 was the target gene of miR-762. In vitro, the results for the T3 group were consistent with the results for the T4 group. Furthermore, cardiomyocyte hypertrophy and autophagic activity were attenuated in the T3 + miR-762 mimic group compared with the T3 group. In contrast, cardiomyocyte hypertrophy and autophagic activity were aggravated in the T3 + miR-762 inhibitor group compared with the T3 group.
Conclusions
miR-762 modulates thyroxine-induced cardiomyocyte hypertrophy by inhibiting Beclin-1.





Similar content being viewed by others
Change history
14 January 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
14 January 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
R.S. Bahn, H.B. Burch, D.S. Cooper, J.R. Garber, M.C. Greenlee, I. Klein, P. Laurberg, I.R. McDougall, V.M. Montori, S.A. Rivkees, Hyperthyroidism and other causes of thyrotoxicosis: management guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Endocr. Pract. 17(3), 456–520 (2011). https://doi.org/10.14341/ket2011748-18
D. Devereaux, S.Z. Tewelde, Hyperthyroidism and thyrotoxicosis. Emerg. Med. Clin. N. Am. 32(2), 277–292 (2014). https://doi.org/10.1016/j.emc.2013.12.001
A.P. Weetman, Graves’ disease. N. Engl. J. Med. 343(17), 1236–1248 (2000). https://doi.org/10.1056/NEJM200010263431707
A. Jabbar, A. Pingitore, S.H. Pearce, A. Zaman, Iervasi G., S. Razvi, Thyroid hormones and cardiovascular disease. Nat. Rev. Cardiol. 14(1), 39–55 (2017). https://doi.org/10.1038/nrcardio.2016.174
J. Yuan, H. Liu, W. Gao, L. Zhang, Y. Ye, L. Yuan, Z. Ding, J. Wu, L. Kang, X. Zhang, MicroRNA-378 suppresses myocardial fibrosis through a paracrine mechanism at the early stage of cardiac hypertrophy following mechanical stress. Theranostics 8(9), 2565–2582 (2018). https://doi.org/10.7150/thno.22878
J.P. Wang, R.F. Chi, K. Wang, T. Ma, X.F. Guo, X.L. Zhang, B. Li, F.Z. Qin, X.B. Han, B.A. Fan, Oxidative stress impairs myocyte autophagy, resulting in myocyte hypertrophy. Exp. Physiol. 103(4), 461–472 (2018). https://doi.org/10.1113/EP086650
D. Glick, S. Barth, K.F. Macleod, Autophagy: cellular and molecular mechanisms. J. Pathol. 221(1), 3–12 (2010). https://doi.org/10.1002/path.2697
S. Kaushik, U. Bandyopadhyay, S. Sridhar, R. Kiffin, M. Martinez-Vicente, M. Kon, S.J. Orenstein, E. Wong, A.M. Cuervo, Chaperone-mediated autophagy at a glance. J. Cell Sci. 124(4), 495–499 (2011). https://doi.org/10.1242/jcs.073874
Shuhei Nakamura, Tamotsu Yoshimori, Autophagy and longevity. Mol. Cells 41(1), 65–72 (2018). https://doi.org/10.14348/molcells.2018.2333
Y.Y. Sun, S.S. Qin, Y.H. Cheng, C.Y. Wang, X.J. Liu, Y. Liu, X.L. Zhang, W. Zhang, J.X. Zhan, S. Shao, MicroRNA expression profile and functional analysis reveal their roles in contact inhibition and its disruption switch of rat vascular smooth muscle cells. Acta Pharmacol. Sin. 39(5), 885–892 (2018). https://doi.org/10.1038/aps.2018.6
K. Liu, Q. Hao, J. Wei, G.H. Li, Y. Wu, Y.F. Zhao, MicroRNA-19a/b-3p protect the heart from hypertension-induced pathological cardiac hypertrophy through PDE5A. J. Hypertens. 36(9), 1847–1857 (2018). https://doi.org/10.1097/HJH.0000000000001769
F. Tang, T.L. Yang, MicroRNA-126 alleviates endothelial cells injury in atherosclerosis by restoring autophagic flux via inhibiting of PI3K/Akt/mTOR pathway. Biochem. Biophys. Res. Commun. 495(1), 1482–1489 (2018). https://doi.org/10.1016/j.bbrc.2017.12.001
H. Liu, P. Liu, X. Shi, D. Yin, J. Zhao, NR4A2 protects cardiomyocytes against myocardial infarction injury by promoting autophagy. Cell Death Discov. 4, 27 (2018). https://doi.org/10.1038/s41420-017-0011-8
Y. Li, Y. Wang, M. Zou, C. Chen, Y. Chen, R. Xue, Y. Dong, C. Liu, AMPK blunts chronic heart failure by inhibiting autophagy. Biosci. Rep. 38(4), BSR20170982 (2018). https://doi.org/10.1042/BSR20170982
Q. Yao, X. Wang, W. He, Z. Song, B. Wang, J. Zhang, Q. Qin, Circulating microRNA-144-3p and miR-762 are novel biomarkers of Graves’ disease. Endocrine (2019). https://doi.org/10.1007/s12020-019-01884-2.
H.H. Samuels, J.S. Tsai, J. Casanova, F. Stanley, Thyroid hormone action: in vitro characterization of solubilized nuclear receptors from rat liver and cultured GH1 cells. J. Clin. Investig. 54(4), 853–865 (1974). https://doi.org/10.1172/JCI107825
P.J. Davis, F.B. Davis, S.A. Mousa, M.K. Luidens, H.Y. Lin, Membrane receptor for thyroid hormone: physiologic and pharmacologic implications. Annu. Rev. Pharmacol. Toxicol. 51, 99–115 (2011). https://doi.org/10.1146/annurev-pharmtox-010510-100512
W. Pan, Y. Zhong, C. Cheng, B. Liu, L. Wang, A. Li, L. Xiong, S. Liu, MiR-30-regulated autophagy mediates angiotensin II-induced myocardial hypertrophy. PLoS ONE 8(1), e53950 (2013). 10.1371/journal.pone.0053950
J. Huang, W. Sun, H. Huang, J. Ye, W. Pan, Y. Zhong, C. Cheng, X. You, B. Liu, L. Xiong, miR-34a modulates angiotensin II-induced myocardial hypertrophy by direct inhibition of ATG9A expression and autophagic activity. PLoS ONE 9(4), e94382 (2014). 10.1371/journal.pone.0094382
A.L. Li, J.B. Lv, L. Gao, MiR-181a mediates Ang II-induced myocardial hypertrophy by mediating autophagy. Eur. Rev. Med. Pharmacol. Sci. 21(23), 5462–5470 (2017). https://doi.org/10.26355/eurrev20171213936
Z. Li, Y. Song, L. Liu, N. Hou, X. An, D. Zhan, Y. Li, L. Zhou, P. Li, L. Yu, miR-199a impairs autophagy and induces cardiac hypertrophy through mTOR activation. Cell Death Differ. 24(7), 1205–1213 (2017). https://doi.org/10.1038/cdd.2015.95
M. Su, J. Wang, C. Wang, X. Wang, W. Dong, W. Qiu, Y. Wang, X. Zhao, Y. Zou, L. Song, MicroRNA-221 inhibits autophagy and promotes heart failure by modulating the p27/CDK2/mTOR axis. Cell Death Differ. 22(6), 986–999 (2015). https://doi.org/10.1038/cdd.2014.187
M. Su, Z. Chen, C. Wang, L. Song, Y. Zou, L. Zhang, R. Hui, J. Wang, Cardiac-specific overexpression of miR-222 induces heart failure and inhibits autophagy in mice. Cell Physiol. Biochem. 39(4), 1503–1511 (2016). https://doi.org/10.1159/00044785
M.A. Cortez, C. Bueso-Ramos, J. Ferdin, G. Lopez-Berestein, A.K. Sood, G.A. Calin, MicroRNAs in body fluids-the mix of hormones and biomarkers. Nat. Rev. Clin. Oncol. 8(8), 467–477 (2011). https://doi.org/10.1038/nrclinonc.2011.76
L. Zheng, C. Zhuang, X. Wang, L. Ming, Serum miR-146a, miR-155, and miR-210 as potential markers of Graves’ disease. J. Clin. Lab. Anal. 32(2) (2018). https://doi.org/10.1002/jcla.22266
J. Wang, Y. Xiao, H. Zhang, Role of miR-146a in the Regulation of Inflammation in an In Vitro Model of Graves’ orbitopathy. Investig. Ophthalmol. Vis. Sci. 57(15), 6795 (2016). https://doi.org/10.1167/iovs.16-20559
Funding
The work was supported by the National Natural Science Foundation of China (81660046), the Guangxi Scholarship Fund of the Guangxi Education Department, and the Natural Science Foundation of Guangxi Province (2018GXNSFAA050096).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All animal protocols were approved by the review board of the Animal Care and Ethics Committee of Guilin Medical University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Qiang, Z., Jin, B., Peng, Y. et al. miR-762 modulates thyroxine-induced cardiomyocyte hypertrophy by inhibiting Beclin-1. Endocrine 66, 585–595 (2019). https://doi.org/10.1007/s12020-019-02048-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-019-02048-y