Abstract
Purpose
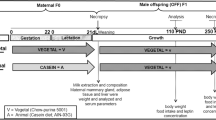
To evaluate the effects of maternal dietary soybean during lactation on the milk composition, body composition, lipid profile and glucose homeostasis of dams and offspring at weaning (21 days) and adulthood (150 days).
Methods
Lactating rats were divided into: casein control (C): casein diet; soy (S): soybean diet; soy oil control (SOC): casein diet, but with fat content similar to the S group.
Results
At 21 days, S mothers showed lower estradiol, total cholesterol (TC), low-density lipoprotein cholesterol (LDL) and triglycerides (TG) in serum; and lower TC and TG in milk. The S offspring had lower body weight, body fat mass, TC, LDL, hyperleptinemia and hypertriglyceridemia. At 150 days, S offspring presented higher total mineral content and lower TC (v. SOC) and LDL (v. C and SOC), and hyperinsulinemia with lower glycemia v. SOC group, which had lower insulinemia with higher glycemia, TC and LDL.
Conclusions
Maternal intake of soybeans in lactation changes the lipid content of breast milk and programmed offspring for phenotype of the lower metabolic risk, with lower serum TC and LDL, and seems to protect the progeny of alterations in glucose metabolism despite the higher lipid content. The difference in fat content of breast milk and the higher isoflavones content of soy diet are possible imprinting factors that could program the offspring.




Similar content being viewed by others
References
C.M. Grieshop, G.C. Fahey Jr, Comparison of quality characteristics of soybeans from Brazil, China and the United States. J. Agric. Food Chem. 49, 2669–2673 (2001)
M. Ding, A. Pan, J.E. Manson et al., Consumption of soy foods and isoflavones and risk of type 2 diabetes: a pooled analysis of three US cohorts. Eur. J. Clin. Nutr. 70, 1381–1387 (2016)
L. Noriega-López, A.R. Tovar, M. Gonzalez-Granillo et al., Pancreatic insulin secretion in rats fed a soy protein high fat diet depends on the interaction between the amino acid pattern and isoflavones. J. Biol. Chem. 282, 20657–20666 (2007)
C.R. Cederroth, M. Vinciguerra, A. Gjinovci et al., Dietary phytoestrogens activate AMP-activated protein kinase with improvement in lipid and glucose metabolism. Diabetes 57, 1176–1185 (2008)
A. Jahan-Mihan, C.E. Smith, A. Hamedani, H. Anderson, Soy protein–based compared with casein-based diets fed during pregnancy and lactation increase food intake and characteristics of metabolic syndrome less in female than male rat offspring. Nutr. Res. 31, 644–651 (2011)
R.L. Ruhlen, K.L. Howdeshell, J. Mao et al., Low phytoestrogen levels in feed increase fetal serum estradiol resulting in the “fetal estrogenization syndrome” and obesity in CD-1 mice. Environ. Health Perspect. 116, 322–328 (2008)
C.R. Cederroth, C. Zimmermann, J.L. Beny et al., Potential detrimental effects of a phytoestrogen-rich diet on male fertility in mice. Mol. Cell Endocrinol. 321, 152–160 (2010)
A.A. Troina, M.S. Figueiredo, E.G. Moura et al., Maternal flaxseed diet during lactation alters milk composition and programs the offspring body composition, lipid profile and sexual function. Food Chem Toxicol. 48, 697–703 (2010)
M.S. Figueiredo, E.G. Moura, P.C. Lisboa et al., Maternal flaxseed diet during lactation programs thyroid hormones metabolism and action in the male adult offspring in rats. Horm. Metab. Res. 43, 410–416 (2011)
D.J. Barker, In útero programming of chronic disease. Clin. Sci. (Lond.). 95, 115–128 (1998)
C.S. Rosenfeld, Homage to the ‘H’ in developmental origins of health and disease. JDOHaD 8(1), 8–29 (2017)
C.R. Cederroth, S. Nef, Fetal programming of adult glucose homeostasis in mice. PLoS One 4, e7281 (2009)
A. Jahan –Mihan, C.E. Smith, H. Anderson, Effect of protein source in diets fed during gestation and lactation on food intake regulation in male offspring of Wistar rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 300, R1175–R1184 (2011)
A. Jahan-mihan, I.M.Y. Szeto, B.L. Luhovyy, P.S.P. Huot, G.H. Anderson, Soya protein-and casein-based nutritionally complete diets fed during gestation and lactation differ in effects on characteristics of the metabolic syndrome in male offspring of Wistar rats. Br. J. Nutr. 107, 284–294 (2012)
K.L. Fischbeck, K.M. Rasmussen, Effect of repeated reproductive cycles on maternal nutritional status, lactational performance and litter growth in ad libitum-fed and chronically food-restricted rats. J. Nutr. 117, 1967–1975 (1987)
P.G. Reeves, F.H. Nielsen, G.C.J. Fahey, AIN_93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76 rodent diet. J. Nutr. 123, 1939–1951 (1993)
L.L. Soares, A.M. Lucas, G.T. Boaventura, Can organic and transgenic soy be used as a substitute for animal protein by rats? Braz. J. Med. Biol. Res. 38, 583–586 (2005)
Association of Official Analytical Chemist (AOAC) Official Methods of Analysis (Washington, 1984)
A.C.P. Barbosa, N.M.A. Hassimotto, F.M. Lajolo, M.I. Genovese, Teores de isoflavonas e capacidade antioxidanteda soja e produtos derivados. Ciênc. Tecnol. Aliment. 26, 921–926 (2006)
M.M. Almeida, S.C.P.D. Luquetti, C.M. Sabarense et al., Butter naturally enriched in cis-9, trans-11 CLA prevents hyperinsulinemia and increases both serum HDL cholesterol and triacylglycerol levels in rats. Lipids Health Dis. 13, 1–12 (2014)
M.C.F. Passos, C.F. Ramos, S.C.P. Dutra, E.G. Moura, Transfer of iodine through the milk in protein restricted lactating rats. J. Nutr. Bioch. 12, 300–303 (2001)
G.L.A. Peterson, Simplification of the protein assay method of Lowry et al. Which is more generally applicabl. Anal. Biochem. 83, 346–356 (1977)
W.T. Friedewald, Determination and calculation of serum low density lipoprotein. Clin. Chem. 18, 499 (1972)
Food and Drug Administration, Food labeling: health claims; soy protein and coronary heart disease. Food and Drug Administration, HHS. Final rule. Fed. Regist. 64, 57700–57733 (1999)
ANVISA. Alimentos com alegações de propriedades funcionais e ou de saúde, novos alimentos/ingredientes, substâncias bioativas e probióticos. http://www.anvisa.gov.br/alimentos/comissoes/tecno_lista_alega.htm. Accessed 28 Oct 2016
M.I. Genovese, F.M. Lajolo, Isoflavones in soy-based foods consumed in Brazil: levels, distribution, and estimated intake. J. Agric. Food Chem. 20, 5987–5993 (2002)
K.D. Setchell, N.M. Brown, P. Desai et al., Bioavailability of pure isoflavones in healthy humans and analysis of commercial soy isoflavone supplements. J. Nutr. 131, 1362S–1375S (2001)
M.G. Glazier, M.A. Bowman, A review of the evidence for the use of phytoestrogens as a replacement for traditional estrogen replacement therapy. Arch. Intern. Med. 161, 1161–1172 (2001)
R. Clapauch, R.M.R. Meirelles, M.A.S.G.J. Julião et al., Fitoestrogênios: Posicionamento do Departamento de Endocrinologia Feminina da Sociedade Brasileira de Endocrinologia e Metabologia (SBEM). Arq. Bras. Endocrinol. Metab. 46, 679–695 (2002)
P.A. Murphy, T. Song, G. Buseman et al., Isoflavones in retail and institutional soy foods. J. Agric. Food Chem. 47, 2697–2704 (1999)
N. Petrakis, S. Barnes, E.B. King, J. Lowenstein et al., Stimulatory influence of soy protein isolate on breast secretion in pre- and postmenopausal women. Cancer Epidemiol. Biomark. Prev. 5, 785–794 (1996)
C. Nagata, N. Takatsuka, S. Inaba, N. Kawakami, H. Shimizu, Effect of soymilk consumption on serum estrogen concentrations in premenopausal japanese women. J. Natl. Cancer Inst. 90, 1830–1835 (1998)
L.J. Lu, K.E. Anderson, J.J. Grady, M. Nagamani, Effects of soya consumption for one month on steroid hormones in premenopausal women: implications for breast cancer risk reduction. Cancer Epidemiol. Biomark. Prev. 5, 63–70 (1996)
B.K. Gangrade, J.S. Davis, J.V. May, A novel mechanism for the induction of aromatase in ovarian cells in vitro: role of transforming growth factor a-induced protein tyrosine kinase. Endocrinology 129, 2790–2792 (1991)
S. Makela, V.L. Davis, W.C. Tally, J. Korkman, L. Salo, R. Vihko, Dietary estrogens act through estrogen receptor-mediated processes and show no antiestrogenicity in culture breast cancer cells. Environ. Health Perspect. 102, 572–578 (1994)
S.C.P. Dutra, M.C.F. Passos et al., Liver deiodinase activity is increased in adult rats whose mothers were submitted to malnutrition during lactation. Horm. Metab. Res. 35, 268–270 (2003)
D.S. Guarda, P.C. Lisboa, E. Oliveira et al., Flaxseed oil during lactation changes milk and body composition in male and female suckling pups rats. Food Chem. Toxicol. 69, 69–75 (2014)
B. Palacios-González, A. Zarain-Herberg, I. Flores-Galicia et al., Genistein stimulates fatty acid oxidation in a leptin receptor-independent manner through the JAK2-mediated phosphorylation and activation of AMPK in skeletal muscle. Biochim. Biophys. Acta 1841, 132–140 (2014)
H. Yang, F. Li, X. Xiong et al., Soy isoflavones modulate adipokines and myokines to regulate lipid metabolism in adipose tissue, skeletal muscle and liver of male Huanjiang mini-pigs. Mol. Cell Endocrinol. 365, 44–51 (2013)
A.A. Franke, L.J. Custer, Daidzein and genistein concentrations in human milk after soy consumption. Clin. Chem. 42, 955–964 (1996)
S.S. Juma, Z. Ezzat-Zadeh, D.A. Khalil et al., Soy protein with or without isoflavones failed to preserve bone density in gonadal hormone deficient male rat model of osteoporosis. Nutr. Res. 32, 694–700 (2012)
M.L. Baglia, K. GU, X. Zhang et al., Soy Isoflavone intake and bone mineral density in breast cancer survivors. Cancer Causes. Control. 26, 571–580 (2015)
F. Cumin, H.P. Baum, N. Levens, Mechanism of leptina removal from the circulation by the kidney. J. Endocrinol. 155, 391–407 (1997)
M. Tanaka, S. Nakaya, T. Kumai et al., Effects of estrogen on serum leptin levels and leptin mRNA expression in adipose tissue in rats. Horm. Res. 56, 98–104 (2001)
J.J. Anderson, Soy phytoestrogens, lipid reductions, and atherosclerosisdelay in ovariectomized primates. J. Clin. Endocrinol. Metab. 86, 39–40 (2001)
A. Sewell, C.B. Hollenbeck, B. Bruce, The effects of soy-derived phytoestrogens on serum lipids and lipoproteins in moderately hypercholesterolemic postmenopausal women. J. Clin. Endocrinol. Metab. 87, 118–121 (2002)
N. Torres, I. Torre-Villalvazo, A.R. Tovar, Regulation of lipid metabolism by soy protein and its implication in diseases mediated by lipid disorders. J. Nutr. Biochem. 17, 365–373 (2006)
Acknowledgements
The authors are grateful to Ms Leandra Natália de Oliveira and Mr João Pablo Fortes Pereira for technical assistance.
Funding
This research was supported by National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq) (Process number: 476867/2011-8), the Federal University of Juiz de Fora (UFJF) and Federal University of Ouro Preto (UFOP) (A. M. V. was a recipient of a UFOP fellowship, and P. G. B. and M. S. were recipient of a UFJF fellowship).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national guides on the care and use of laboratory animals (Wistar rats) and have been approved by the institutional committee [Ethics Committee of the Federal University of Juiz de Fora at Minas Gerais, Brazil (protocol 018/2014)].
Rights and permissions
About this article
Cite this article
Vieira, A.M., de Almeida Brasiel, P.G., Ferreira, M.S. et al. Maternal soybean diet during lactation alters breast milk composition and programs the lipid profile in adult male rat offspring. Endocrine 60, 272–281 (2018). https://doi.org/10.1007/s12020-018-1572-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1572-x