Abstract
Purpose
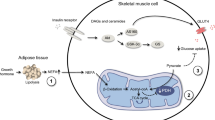
Growth hormone (GH) replacement decreases insulin sensitivity in healthy individuals. However, the effects of GH on organ-specific insulin sensitivity and glucose effectiveness are not well characterized. The purpose of this study was to evaluate the effects of GH administration for 26 weeks on muscle and hepatic insulin sensitivity and glucose effectiveness in healthy older individuals.
Methods
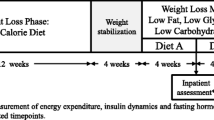
This report is from a 26-week randomized, double-blind, placebo-controlled parallel-group trial in healthy, ambulatory, community-dwelling older women and men. We compared surrogate indices of insulin sensitivity [quantitative insulin-sensitivity check index (QUICKI), muscle insulin sensitivity index (MISI), hepatic insulin resistance index (HIRI)] and glucose effectiveness [oral glucose effectiveness index (oGE)] derived from oral glucose tolerance tests (OGTTs) in subjects before and after 26 weeks of administration of GH (n = 17) or placebo (n = 15) as an exploratory outcome.
Results
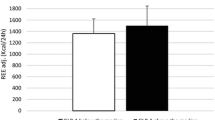
GH administration for 26 weeks significantly increased fasting insulin concentrations and HIRI but did not significantly change MISI or oGE compared to placebo.
Conclusions
GH administration for 26 weeks in healthy older subjects impairs insulin sensitivity in the liver but not skeletal muscle and does not alter glucose effectiveness.

Similar content being viewed by others
References
S. Fieffe, I. Morange, P. Petrossians, P. Chanson, V. Rohmer, C. Cortet, F. Borson-Chazot, T. Brue, B. Delemer; French Acromegaly, R, Diabetes in acromegaly, prevalence, risk factors, and evolution: data from the French Acromegaly Registry. Eur. J. Endocrinol. 164(6), 877–884 (2011). https://doi.org/10.1530/EJE-10-1050
O. Alexopoulou, M. Bex, P. Kamenicky, A.B. Mvoula, P. Chanson, D. Maiter, Prevalence and risk factors of impaired glucose tolerance and diabetes mellitus at diagnosis of acromegaly: a study in 148 patients. Pituitary 17(1), 81–89 (2014). https://doi.org/10.1007/s11102-013-0471-7
N. Moller, O. Schmitz, J.O. Joorgensen, J. Astrup, J.F. Bak, S.E. Christensen, K.G. Alberti, J. Weeke, Basal- and insulin-stimulated substrate metabolism in patients with active acromegaly before and after adenomectomy. J. Clin. Endocrinol. Metab. 74(5), 1012–1019 (1992). https://doi.org/10.1210/jcem.74.5.1569148
N. Moller, J.O. Jorgensen, Effects of growth hormone on glucose, lipid, and protein metabolism in human subjects. Endocr. Rev. 30(2), 152–177 (2009). https://doi.org/10.1210/er.2008-0027
I. Hansen, E. Tsalikian, B. Beaufrere, J. Gerich, M. Haymond, R. Rizza, Insulin resistance in acromegaly: defects in both hepatic and extrahepatic insulin action. Am. J. Physiol. 250(3 Pt 1), E269–E273 (1986). https://doi.org/10.1152/ajpendo.1986.250.3.E269
S. Kasayama, M. Otsuki, M. Takagi, H. Saito, S. Sumitani, H. Kouhara, M. Koga, Y. Saitoh, T. Ohnishi, N. Arita, Impaired beta-cell function in the presence of reduced insulin sensitivity determines glucose tolerance status in acromegalic patients. Clin. Endocrinol. 52(5), 549–555 (2000)
Y. Kinoshita, H. Fujii, A. Takeshita, M. Taguchi, M. Miyakawa, K. Oyama, S. Yamada, Y. Takeuchi, Impaired glucose metabolism in Japanese patients with acromegaly is restored after successful pituitary surgery if pancreatic {beta}-cell function is preserved. Eur. J. Endocrinol. 164(4), 467–473 (2011). https://doi.org/10.1530/EJE-10-1096
C.E. Higham, S. Rowles, D. Russell-Jones, A.M. Umpleby, P.J. Trainer, Pegvisomant improves insulin sensitivity and reduces overnight free fatty acid concentrations in patients with acromegaly. J. Clin. Endocrinol. Metab. 94(7), 2459–2463 (2009). https://doi.org/10.1210/jc.2008-2086
E.O. List, L. Sackmann-Sala, D.E. Berryman, K. Funk, B. Kelder, E.S. Gosney, S. Okada, J. Ding, D. Cruz-Topete, J.J. Kopchick, Endocrine parameters and phenotypes of the growth hormone receptor gene disrupted (GHR-/-) mouse. Endocr. Rev. 32(3), 356–386 (2011). https://doi.org/10.1210/er.2010-0009
J. Guevara-Aguirre, P. Balasubramanian, M. Guevara-Aguirre, M. Wei, F. Madia, C.W. Cheng, D. Hwang, A. Martin-Montalvo, J. Saavedra, S. Ingles, R. de Cabo, P. Cohen, V.D. Longo, Growth hormone receptor deficiency is associated with a major reduction in pro-aging signaling, cancer, and diabetes in humans. Sci. Transl. Med. 3(70), 70ra13 (2011). https://doi.org/10.1126/scitranslmed.3001845
R.A. Rizza, L.J. Mandarino, J.E. Gerich, Effects of growth hormone on insulin action in man. Mechanisms of insulin resistance, impaired suppression of glucose production, and impaired stimulation of glucose utilization. Diabetes 31(8 Pt 1), 663–669 (1982)
K.C. Corbit, J.P. Camporez, J.L. Tran, C.G. Wilson, D.A. Lowe, S.M. Nordstrom, K. Ganeshan, R.J. Perry, G.I. Shulman, M.J. Jurczak, E.J. Weiss, Adipocyte JAK2 mediates growth hormone-induced hepatic insulin resistance. JCI Insight 2(3), e91001 (2017). https://doi.org/10.1172/jci.insight.91001
F.P. Dominici, D.P. Argentino, M.C. Munoz, J.G. Miquet, A.I. Sotelo, D. Turyn, Influence of the crosstalk between growth hormone and insulin signalling on the modulation of insulin sensitivity. Growth Horm. IGF Res. 15(5), 324–336 (2005). https://doi.org/10.1016/j.ghir.2005.07.001
N. Moller, P.C. Butler, M.A. Antsiferov, K.G. Alberti, Effects of growth hormone on insulin sensitivity and forearm metabolism in normal man. Diabetologia 32(2), 105–110 (1989)
P.M. Piatti, L.D. Monti, A. Caumo, M. Conti, F. Magni, M. Galli-Kienle, E. Fochesato, A. Pizzini, L. Baldi, G. Valsecchi, A.E. Pontiroli, Mediation of the hepatic effects of growth hormone by its lipolytic activity. J. Clin. Endocrinol. Metab. 84(5), 1658–1663 (1999). https://doi.org/10.1210/jcem.84.5.5685
L. Orskov, O. Schmitz, J.O. Jorgensen, J. Arnfred, N. Abildgaard, J.S. Christiansen, K.G. Alberti, H. Orskov, Influence of growth hormone on glucose-induced glucose uptake in normal men as assessed by the hyperglycemic clamp technique. J. Clin. Endocrinol. Metab. 68(2), 276–282 (1989). https://doi.org/10.1210/jcem-68-2-276
A. Thankamony, P.H. Tossavainen, A. Sleigh, C. Acerini, D. Elleri, R.N. Dalton, N.C. Jackson, A.M. Umpleby, R.M. Williams, D.B. Dunger, Short-term administration of pegvisomant improves hepatic insulin sensitivity and reduces soleus muscle intramyocellular lipid content in young adults with type 1 diabetes. J. Clin. Endocrinol. Metab. 99(2), 639–647 (2014). https://doi.org/10.1210/jc.2013-3264
J. Svensson, J. Fowelin, K. Landin, B.A. Bengtsson, J.O. Johansson, Effects of seven years of GH-replacement therapy on insulin sensitivity in GH-deficient adults. J. Clin. Endocrinol. Metab. 87(5), 2121–2127 (2002). https://doi.org/10.1210/jcem.87.5.8482
A.M. Rosenfalck, S. Maghsoudi, S. Fisker, J.O. Jorgensen, J.S. Christiansen, J. Hilsted, A.A. Volund, S. Madsbad, The effect of 30 months of low-dose replacement therapy with recombinant human growth hormone (rhGH) on insulin and C-peptide kinetics, insulin secretion, insulin sensitivity, glucose effectiveness, and body composition in GH-deficient adults. J. Clin. Endocrinol. Metab. 85(11), 4173–4181 (2000). https://doi.org/10.1210/jcem.85.11.6930
T. Munzer, S.M. Harman, J.D. Sorkin, M.R. Blackman, Growth hormone and sex steroid effects on serum glucose, insulin, and lipid concentrations in healthy older women and men. J. Clin. Endocrinol. Metab. 94(10), 3833–3841 (2009). https://doi.org/10.1210/jc.2009-1275
M.R. Blackman, J.D. Sorkin, T. Munzer, M.F. Bellantoni, J. Busby-Whitehead, T.E. Stevens, J. Jayme, K.G. O’Connor, C. Christmas, J.D. Tobin, K.J. Stewart, E. Cottrell, C. St Clair, K.M. Pabst, S.M. Harman, Growth hormone and sex steroid administration in healthy aged women and men: a randomized controlled trial. JAMA 288(18), 2282–2292 (2002)
T. Munzer, S.M. Harman, P. Hees, E. Shapiro, C. Christmas, M.F. Bellantoni, T.E. Stevens, K.G. O’Connor, K.M. Pabst, C. St Clair, J.D. Sorkin, M.R. Blackman, Effects of GH and/or sex steroid administration on abdominal subcutaneous and visceral fat in healthy aged women and men. J. Clin. Endocrinol. Metab. 86(8), 3604–3610 (2001). https://doi.org/10.1210/jcem.86.8.7773
American Diabetes Associations, Standards of medical care in diabetes-2017 abridged for primary care providers. Clin. Diabetes. 35(1), 5–26 (2017). https://doi.org/10.2337/cd16-0067
T. Munzer, C.J. Rosen, S.M. Harman, K.M. Pabst, C. St Clair, J.D. Sorkin, M.R. Blackman, Effects of GH and/or sex steroids on circulating IGF-I and IGFBPs in healthy, aged women and men. Am. J. Physiol. Endocrinol. Metab. 290(5), E1006–E1013 (2006). https://doi.org/10.1152/ajpendo.00166.2005
M.A. Abdul-Ghani, M. Matsuda, B. Balas, R.A. DeFronzo, Muscle and liver insulin resistance indexes derived from the oral glucose tolerance test. Diabetes Care 30(1), 89–94 (2007). https://doi.org/10.2337/dc06-1519
S. Nagasaka, I. Kusaka, K. Yamashita, Y. Funase, K. Yamauchi, M. Katakura, S. Ishibashi, T. Aizawa, Index of glucose effectiveness derived from oral glucose tolerance test. Acta Diabetol. 49(Suppl 1), S195–S204 (2012). https://doi.org/10.1007/s00592-012-0417-y
R. Weiss, S.N. Magge, N. Santoro, C. Giannini, R. Boston, T. Holder, M. Shaw, E. Duran, K.J. Hershkop, S. Caprio, Glucose effectiveness in obese children: relation to degree of obesity and dysglycemia. Diabetes Care 38(4), 689–695 (2015). https://doi.org/10.2337/dc14-2183
R. Muniyappa, S. Lee, H. Chen, M.J. Quon, Current approaches for assessing insulin sensitivity and resistance in vivo: advantages, limitations, and appropriate usage. Am. J. Physiol. Endocrinol. Metab. 294(1), E15–E26 (2008). https://doi.org/10.1152/ajpendo.00645.2007
A. Kotronen, M. Lewitt, K. Hall, K. Brismar, H. Yki-Jarvinen, Insulin-like growth factor binding protein 1 as a novel specific marker of hepatic insulin sensitivity. J. Clin. Endocrinol. Metab. 93(12), 4867–4872 (2008). https://doi.org/10.1210/jc.2008-1245
J.L. Kuk, T.J. Saunders, L.E. Davidson, R. Ross, Age-related changes in total and regional fat distribution. Ageing Res. Rev. 8(4), 339–348 (2009). https://doi.org/10.1016/j.arr.2009.06.001
E.B. Geer, W. Shen, Gender differences in insulin resistance, body composition, and energy balance. Gend. Med. 6(Suppl 1), 60–75 (2009). https://doi.org/10.1016/j.genm.2009.02.002
M. Krotkiewski, P. Bjorntorp, L. Sjostrom, U. Smith, Impact of obesity on metabolism in men and women. Importance of regional adipose tissue distribution. J. Clin. Invest. 72(3), 1150–1162 (1983). https://doi.org/10.1172/JCI111040
R. Basu, C. Dalla Man, M. Campioni, A. Basu, G. Klee, G. Toffolo, C. Cobelli, R.A. Rizza, Effects of age and sex on postprandial glucose metabolism: differences in glucose turnover, insulin secretion, insulin action, and hepatic insulin extraction. Diabetes 55(7), 2001–2014 (2006). https://doi.org/10.2337/db05-1692
J.O. Clausen, K. Borch-Johnsen, H. Ibsen, R.N. Bergman, P. Hougaard, K. Winther, O. Pedersen, Insulin sensitivity index, acute insulin response, and glucose effectiveness in a population-based sample of 380 young healthy Caucasians. Analysis of the impact of gender, body fat, physical fitness, and life-style factors. J. Clin. Invest. 98(5), 1195–1209 (1996). https://doi.org/10.1172/JCI118903
J.W. Kolaczynski, J.F. Caro, Insulin-like growth factor-1 therapy in diabetes: physiologic basis, clinical benefits, and risks. Ann. Intern. Med. 120(1), 47–55 (1994)
T. Pratipanawatr, W. Pratipanawatr, C. Rosen, R. Berria, M. Bajaj, K. Cusi, L. Mandarino, S. Kashyap, R. Belfort, R.A. DeFronzo, Effect of IGF-I on FFA and glucose metabolism in control and type 2 diabetic subjects. Am. J. Physiol. Endocrinol. Metab. 282(6), E1360–E1368 (2002). https://doi.org/10.1152/ajpendo.00335.2001
G. Paolisso, M.R. Tagliamonte, M.R. Rizzo, C. Carella, A. Gambardella, M. Barbieri, M. Varricchio, Low plasma insulin-like growth factor-1 concentrations predict worsening of insulin-mediated glucose uptake in older people. J. Am. Geriatr. Soc. 47(11), 1312–1318 (1999)
J.E. Henriksen, K. Levin, P. Thye-Ronn, F. Alford, O. Hother-Nielsen, J.J. Holst, H. Beck-Nielsen, Glucose-mediated glucose disposal in insulin-resistant normoglycemic relatives of type 2 diabetic patients. Diabetes 49(7), 1209–1218 (2000)
S. Kehlenbrink, S. Koppaka, M. Martin, R. Relwani, M.H. Cui, J.H. Hwang, Y. Li, R. Basu, M. Hawkins, P. Kishore, Elevated NEFA levels impair glucose effectiveness by increasing net hepatic glycogenolysis. Diabetologia 55(11), 3021–3028 (2012). https://doi.org/10.1007/s00125-012-2662-6
F. Giorgino, L. Laviola, J.W. Eriksson, Regional differences of insulin action in adipose tissue: insights from in vivo and in vitro studies. Acta Physiol. Scand. 183(1), 13–30 (2005). https://doi.org/10.1111/j.1365-201X.2004.01385.x
A. Garg, A. Misra, Hepatic steatosis, insulin resistance, and adipose tissue disorders. J. Clin. Endocrinol. Metab. 87(7), 3019–3022 (2002). https://doi.org/10.1210/jcem.87.7.8736
T. McLaughlin, C. Lamendola, A. Liu, F. Abbasi, Preferential fat deposition in subcutaneous versus visceral depots is associated with insulin sensitivity. J. Clin. Endocrinol. Metab. 96(11), E1756–E1760 (2011). https://doi.org/10.1210/jc.2011-0615
N. Jessen, C.B. Djurhuus, J.O. Jorgensen, L.S. Jensen, N. Moller, S. Lund, O. Schmitz, Evidence against a role for insulin-signaling proteins PI 3-kinase and Akt in insulin resistance in human skeletal muscle induced by short-term GH infusion. Am. J. Physiol. Endocrinol. Metab. 288(1), E194–E199 (2005). https://doi.org/10.1152/ajpendo.00149.2004
M.S. Raben, Growth hormone. 1. Physiologic aspects. N. Engl. J. Med. 266, 31–35 (1962). https://doi.org/10.1056/NEJM196201042660109
S. Nielsen, N. Moller, J.S. Christiansen, J.O. Jorgensen, Pharmacological antilipolysis restores insulin sensitivity during growth hormone exposure. Diabetes 50(10), 2301–2308 (2001)
M. Segerlantz, M. Bramnert, P. Manhem, E. Laurila, L.C. Groop, Inhibition of the rise in FFA by Acipimox partially prevents GH-induced insulin resistance in GH-deficient adults. J. Clin. Endocrinol. Metab. 86(12), 5813–5818 (2001). https://doi.org/10.1210/jcem.86.12.8096
M.H. Pedersen, M.V. Svart, J. Lebeck, M. Bidlingmaier, H. Stodkilde-Jorgensen, S.B. Pedersen, N. Moller, N. Jessen, J.O.L. Jorgensen, Substrate metabolism and insulin sensitivity during fasting in obese human subjects: impact of GH blockade. J. Clin. Endocrinol. Metab. 102(4), 1340–1349 (2017). https://doi.org/10.1210/jc.2016-3835
R.J. Perry, J.P. Camporez, R. Kursawe, P.M. Titchenell, D. Zhang, C.J. Perry, M.J. Jurczak, A. Abudukadier, M.S. Han, X.M. Zhang, H.B. Ruan, X. Yang, S. Caprio, S.M. Kaech, H.S. Sul, M.J. Birnbaum, R.J. Davis, G.W. Cline, K.F. Petersen, G.I. Shulman, Hepatic acetyl CoA links adipose tissue inflammation to hepatic insulin resistance and type 2 diabetes. Cell 160(4), 745–758 (2015). https://doi.org/10.1016/j.cell.2015.01.012
S.B. Wheatcroft, M.T. Kearney, IGF-dependent and IGF-independent actions of IGF-binding protein-1 and -2: implications for metabolic homeostasis. Trends Endocrinol. Metab.: TEM 20(4), 153–162 (2009). https://doi.org/10.1016/j.tem.2009.01.002
J.H. Goedecke, L.K. Micklesfield, The effect of exercise on obesity, body fat distribution and risk for type 2 diabetes. Med. Sport. Sci. 60, 82–93 (2014). https://doi.org/10.1159/000357338
M.C. Moore, A.D. Cherrington, D.H. Wasserman, Regulation of hepatic and peripheral glucose disposal. Best. Pract. Res. Clin. Endocrinol. Metab. 17(3), 343–364 (2003)
T. Laursen, C.H. Gravholt, L. Heickendorff, J. Drustrup, A.M. Kappelgaard, J.O. Jorgensen, J.S. Christiansen, Long-term effects of continuous subcutaneous infusion versus daily subcutaneous injections of growth hormone (GH) on the insulin-like growth factor system, insulin sensitivity, body composition, and bone and lipoprotein metabolism in GH-deficient adults. J. Clin. Endocrinol. Metab. 86(3), 1222–1228 (2001). https://doi.org/10.1210/jcem.86.3.7323
J. Vangipurapu, A. Stancakova, T. Kuulasmaa, J. Paananen, J. Kuusisto, E.-R.S. Group, E. Ferrannini, M. Laakso, A novel surrogate index for hepatic insulin resistance. Diabetologia 54(3), 540–543 (2011). https://doi.org/10.1007/s00125-010-1966-7
J.P. Bastard, M. Faraj, A.D. Karelis, J. Lavasseur, D. Garrel, D. Prud’homme, R. Rabasa-Lhoret, Muscle and liver insulin resistance indexes derived from the oral glucose tolerance test: response to Abdul-Ghani et al. Diabetes Care 30(7), e84 (2007). author reply e84 https://doi.org/10.2337/dc07-0622.
Funding
This work was supported in part by the Intramural Research Programs of the National Institute on Aging (NIA), Baltimore, Maryland and the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), Washington, DC, National Institutes of Health Research Grants RO-1 AG11005 (to MRB), and the Research Service, Veterans Affairs Medical Center, Washington DC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of The Institutional Review Board of the Johns Hopkins Bayview Medical Center and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Forrest, L., Sedmak, C., Sikder, S. et al. Effects of growth hormone on hepatic insulin sensitivity and glucose effectiveness in healthy older adults. Endocrine 63, 497–506 (2019). https://doi.org/10.1007/s12020-018-01834-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-01834-4