Abstract
Purpose
Several medications may interact with levothyroxine (LT4) intestinal absorption or metabolism, thus reducing its bioavailability. We investigated the variability of thyroid stimulating hormone (TSH) levels and prescribed daily dosages (PDDs) of LT4 before and during potential drug–drug interactions (DDIs) in users of tablets vs. oral liquid LT4 formulations.
Methods
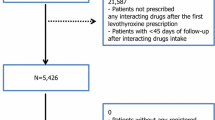
By using the Italian general practice Health Search Database (HSD), we retrospectively selected adult patients with at least one LT4 prescription from 2012 to 2015 and at least 1 year of clinical history recorded. The incident prescription of interacting medications (e.g., proton pump inhibitors, calcium or iron salts) was the index date. Analysis was carried out using a self-controlled study design.
Results
Overall, 3965 users of LT4 formed the study cohort (84.1% women, mean age 56 ± 16.5 years). TSH variability on the entry date was greater among liquid LT4 users than in those prescribed with tablets as shown by the difference between 75th and 25th centile, which were 3.01 and 3.8, respectively. The incidence rate ratio (IRR) for TSH variability did not differ between groups, before and during exposure to DDIs. In contrast, PDDs less likely increased during the exposure to DDI with oral liquid LT4 compared with tablets (IRR = 0.84; 95% CI: 0.77–0.92), especially in patients with post-surgical hypothyroidism (IRR = 0.75; 95% CI: 0.64–0.85).
Conclusions
In clinical practice, the use of oral liquid LT4 is not associated with increased PDDs, compared with tablets formulation, during exposure to DDIs. These results support the need for individualizing LT4 formulation to prescribe, especially in patients with various comorbidities and complex therapeutic regimens.
Similar content being viewed by others
References
J.R. Garber, R.H. Cobin, H. Gharib, J.V. Hennessey, I. Klein, J.I. Mechanick, R. Pessah-Pollack, P.A. Singer, K.A. Woeber, Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid 22(12), 1200–1235 (2012). https://doi.org/10.1089/thy.2012.0205
A.J. Chakera, S.H. Pearce, B. Vaidya, Treatment for primary hypothyroidism: current approaches and future possibilities. Drug. Des. Devel. Ther. 6, 1–11 (2012). https://doi.org/10.2147/DDDT.S12894
W.E. Visser, E.C. Friesema, T.J. Visser, Minireview: thyroid hormone transporters: the knowns and the unknowns. Mol. Endocrinol. 25(1), 1–14 (2011). https://doi.org/10.1210/me.2010-0095
M.T. Hays, Localization of human thyroxine absorption. Thyroid 1(3), 241–248 (1991). https://doi.org/10.1089/thy.1991.1.241
M. Centanni, L. Gargano, G. Canettieri, N. Viceconti, A. Franchi, G. Delle Fave, B. Annibale, Thyroxine in goiter, Helicobacter pylori infection, and chronic gastritis. N. Engl. J. Med. 354(17), 1787–1795 (2006)
C. Virili, G. Bassotti, M.G. Santaguida, R. Iuorio, S.C. Del Duca, V. Mercuri, A. Picarelli, P. Gargiulo, L. Gargano, M. Centanni, Atypical celiac disease as cause of increased need for thyroxine: a systematic study. J. Clin. Endocrinol. Metab. 97(3), E419–E422 (2012). https://doi.org/10.1210/jc.2011-1851
M. Ruchala, E. Szczepanek-Parulska, A. Zybek, The influence of lactose intolerance and other gastro-intestinal tract disorders on L-thyroxine absorption. Endokrynol. Pol. 63(4), 318–323 (2012)
R. Padwal, D. Brocks, A.M. Sharma, A systematic review of drug absorption following bariatric surgery and its theoretical implications. Obes. Rev. 11(1), 41–50 (2010). https://doi.org/10.1111/j.1467-789X.2009.00614.x
G. Ianiro, F. Mangiola, T.A. Di Rienzo, S. Bibbo, F. Franceschi, A.V. Greco, A. Gasbarrini, Levothyroxine absorption in health and disease, and new therapeutic perspectives. Eur. Rev. Med. Pharmacol. Sci. 18(4), 451–456(2014)
L. Liwanpo, J.M. Hershman, Conditions and drugs interfering with thyroxine absorption. Best Pract. Res. Clin. Endocrinol. Metab. 23(6), 781–792 (2009). https://doi.org/10.1016/j.beem.2009.06.006
M. Lombardo, A. Bellia, F. Mattiuzzo, A. Franchi, C. Ferri, E. Padua, V. Guglielmi, M. D’Adamo, G. Annino, P. Gentileschi, F. Iellamo, D. Lauro, M. Federici, P. Sbraccia, Frequent follow-up visits reduce weight regain in long-term management after bariatric surgery. Bariatr. Surg. Pract. Patient Care 10(3), 119–125(2015).
S. Benvenga, L. Bartolone, S. Squadrito, F. Lo Giudice, F. Trimarchi, Delayed intestinal absorption of levothyroxine. Thyroid 5(4), 249–253 (1995). https://doi.org/10.1089/thy.1995.5.249
T.G. Bach-Huynh, B. Nayak, J. Loh, S.Soldin, J. Jonklaas, Timing of levothyroxine administration affects serum thyrotropin concentration. J. Clin. Endocrinol. Metab. 94(10), 3905–3912 (2009). https://doi.org/10.1210/jc.2009-0860
N. Bolk, T.J. Visser, J. Nijman, I.J. Jongste, J.G. Tijssen, A. Berghout, Effects of evening vs morning levothyroxine intake: a randomized double-blind crossover trial. Arch. Intern. Med. 170(22), 1996–2003 (2010). https://doi.org/10.1001/archinternmed.2010.436
R.D. Utiger, Estrogen, thyroxine binding in serum, and thyroxine therapy. N. Engl. J. Med. 344(23), 1784–1785 (2001). https://doi.org/10.1056/NEJM200106073442310
S.A. Hamed, The effect of antiepileptic drugs on thyroid hormonal function: causes and implications. Expert. Rev. Clin. Pharmacol. 8(6), 741–750 (2015). https://doi.org/10.1586/17512433.2015.1091302
C.S. Yue, C. Scarsi, M.P. Ducharme, Pharmacokinetics and potential advantages of a new oral solution of levothyroxine vs. other available dosage forms. Arzneimittelforschung. 62(12), 631–636 (2012). https://doi.org/10.1055/s-0032-1329951
R. Vita, P. Fallahi, A. Antonelli, S. Benvenga, The administration of L-thyroxine as soft gel capsule or liquid solution. Expert. Opin. Drug. Deliv. 11(7), 1103–1111 (2014). https://doi.org/10.1517/17425247.2014.918101
D. Pabla, F. Akhlaghi, H. Zia, A comparative pH-dissolution profile study of selected commercial levothyroxine products using inductively coupled plasma mass spectrometry. Eur. J. Pharm. Biopharm. 72(1), 105–110 (2009). https://doi.org/10.1016/j.ejpb.2008.10.008S0939-6411(08)00387-1
A. Cassio, S. Monti, A. Rizzello, I. Bettocchi, F. Baronio, G. D’Addabbo, M.O. Bal, A. Balsamo, Comparison between liquid and tablet formulations of levothyroxine in the initial treatment of congenital hypothyroidism. J. Pediatr. 162(6), 1264–1269 (2013). https://doi.org/10.1016/j.jpeds.2012.11.070
R. Vita, F. Di Bari, S. Benvenga, Oral liquid levothyroxine solves the problem of tablet levothyroxine malabsorption due to concomitant intake of multiple drugs. Expert. Opin. Drug. Deliv. 14(4), 467–472 (2017). https://doi.org/10.1080/17425247.2017.1290604
R. Vita, G. Saraceno, F. Trimarchi, S. Benvenga, Switching levothyroxine from the tablet to the oral solution formulation corrects the impaired absorption of levothyroxine induced by proton-pump inhibitors. J. Clin. Endocrinol. Metab. 99(12), 4481–4486 (2014). https://doi.org/10.1210/jc.2014-2684
C. Virili, P. Trimboli, F. Romanelli, M. Centanni, Liquid and softgel levothyroxine use in clinical practice: state of the art. Endocrine 54(1), 3–14 (2016). https://doi.org/10.1007/s12020-016-1035-1.
P. Fallahi, S.M. Ferrari, A. Antonelli, In patients with subclinical hypothyroidism while in therapy with tablet L-T4, the liquid L-T4 formulation is more effective in restoring euthyroidism. Endocr. Pract. 23(2), 170–174 (2017). https://doi.org/10.4158/EP161545.OR
C.P. Lombardi, R. Bocale, A. Barini, A. D’Amore, M. Boscherini, R. Bellantone, Comparative study between the effects of replacement therapy with liquid and tablet formulations of levothyroxine on mood states, self-perceived psychological well-being and thyroid hormone profile in recently thyroidectomized patients. Endocrine 55(1), 51–59 (2017). https://doi.org/10.1007/s12020-016-1003-9.
R. Ferrara, V. lentile, V. Arcoraci, C. Ferrajolo, C. Piccinni, A. Fontana, S. Benvenga, G. Trifiro, Treatment pattern and frequency of serum TSH measurement in users of different levothyroxineformulations: a population-based study during the years 2009–2015. Endocrine (2017).10.1007/s12020-017-1242-4
G.M. Leggio, T. Incognito, G. Privitera, M.R. Marano, F. Drago, Comparative bioavailability of different formulations of levothyroxine and liothyronine in healthy volunteers. J. Endocrinol. Invest. 29(11), RC35–RC38 (2006). 10.1007/BF03349205
P. Fallahi, S.M. Ferrari, I. Ruffilli, A. Antonelli, Reversible normalisation of serum TSH levels in patients with autoimmune atrophic gastritis who received L-T4 in tablet form after switching to an oral liquid formulation: a case series. BMC. Gastroenterol. 16, 22 (2016). https://doi.org/10.1186/s12876-016-0439-y
M.G. Santaguida, C. Virili, S.C. Del Duca, M. Cellini, I. Gatto, N. Brusca, C. De Vito, L. Gargano, M. Centanni, Thyroxine softgel capsule in patients with gastric-related T4 malabsorption. Endocrine 49(1), 51–57 (2015). https://doi.org/10.1007/s12020-014-0476-7
D. Ribichini, G. Fiorini, A. Repaci, V. Castelli, L. Gatta, E. Vaira, R. Pasquali, Tablet and oral liquid Lthyroxineformulation in the treatment of naive hypothyroid patients with Helicobacter pyloriinfection. Endocrine (2016). 10.1007/s12020-016-1167-3
I. Pirola, A.M. Formenti, E. Gandossi, F. Mittempergher, C. Casella, B. Agosti, C. Cappelli, Oral liquid L-thyroxine (L-t4) may be better absorbed compared to L-T4 tablets following bariatric surgery. Obes. Surg. 23(9), 1493–1496 (2013). https://doi.org/10.1007/s11695-013-1015-y
P. Fallahi, S.M. Ferrari, S. Camastra, U. Politti, I. Ruffilli, R. Vita, G. Navarra, S. Benvenga, A. Antonelli, TSH normalization in bariatric surgery patients after the switch from L-thyroxine in tablet to an oral liquid formulation. Obes. Surg. 27(1), 78–82 (2017). https://doi.org/10.1007/s11695-016-2247-4
S.A. Irving, T. Vadiveloo, G.P. Leese, Drugs that interact with levothyroxine: an observational study from the thyroid epidemiology, audit and research study (TEARS). Clin. Endocrinol.82(1), 136–141 (2015). https://doi.org/10.1111/cen.12559
I. Sachmechi, D.M. Reich, M. Aninyei, F. Wibowo, G. Gupta, P.J. Kim, Effect of proton pump inhibitors on serum thyroid-stimulating hormone level in euthyroid patients treated with levothyroxine for hypothyroidism. Endocr. Pract. 13(4), 345–349 (2007). 10.4158/EP.13.4.345
C.J. Diskin, T.J. Stokes, L.M. Dansby, L. Radcliff, T.B. Carter, Effect of phosphate binders upon TSH and L-thyroxine dose in patients on thyroid replacement. Int. Urol. Nephrol. 39(2), 599–602 (2007). https://doi.org/10.1007/s11255-006-9166-6
J.W. Dietrich, K. Gieselbrecht, R.W. Holl, B.O. Boehm, Absorption kinetics of levothyroxine is not altered by proton-pump inhibitor therapy. Horm. Metab. Res. 38(1), 57–59 (2006). https://doi.org/10.1055/s-2006-924980
S. Ananthakrishnan, L.E. Braverman, R.M. Levin, B. Magnani, E.N. Pearce, The effect of famotidine, esomeprazole, and ezetimibe on levothyroxine absorption. Thyroid 18(5), 493–498 (2008). https://doi.org/10.1089/thy.2007.0381
V. Guglielmi, A. Bellia, S. Pecchioli, G. Medea, D. Parretti, D. Lauro, P. Sbraccia, M. Federici, I. Cricelli, C. Cricelli, F. Lapi, What is the actual epidemiology of familial hypercholesterolemia in Italy? Evidence from a national primary care database. Int. J. Cardiol. 223, 701–705 (2016). https://doi.org/10.1016/j.ijcard.2016.08.269
V. Guglielmi, A. Bellia, S. Pecchioli, D. Della-Morte, D. Parretti, I. Cricelli, G. Medea, P. Sbraccia, D. Lauro, C. Cricelli, F. Lapi, Effectiveness of adherence to lipid lowering therapy on LDL-cholesterol in patients with very high cardiovascular risk: a real-world evidence study in primary care. Atherosclerosis 263, 36–41 (2017). https://doi.org/10.1016/j.atherosclerosis.2017.05.018
C. Cricelli, G. Mazzaglia, F. Samani, M. Marchi, A. Sabatini, R. Nardi, G. Ventriglia, A.P. Caputi, Prevalence estimates for chronic diseases in Italy: exploring the differences between self-report and primary care databases. J. Public Health Med. 25(3), 254–257 (2003)
G. Trifiro, P. Morabito, L. Cavagna, C. Ferrajolo, S. Pecchioli, M. Simonetti, E. Bianchini, G. Medea, C. Cricelli, A.P. Caputi, G. Mazzaglia, Epidemiology of gout and hyperuricaemia in Italy during the years 2005-2009: a nationwide population-based study. Annrheumdis 72(5), 694–700 (2013). https://doi.org/10.1136/annrheumdis-2011-201254
F. Lapi, M. Simonetti, R. Michieli, A. Pasqua, M.L. Brandi, B. Frediani, C. Cricelli, G. Mazzaglia, Assessing 5-year incidence rates and determinants of osteoporotic fractures in primary care. Bone 50(1), 85–90 (2012). https://doi.org/10.1016/j.bone.2011.09.048
G. Giussani, C. Cricelli, F. Mazzoleni, I. Cricelli, A. Pasqua, S. Pecchioli, F. Lapi, E. Beghi, Prevalence and incidence of epilepsy in Italy based on a nationwide database. Neuroepidemiology 43(3-4), 228–232 (2014). https://doi.org/10.1159/000368801
P. Sathi, S. Kalyan, C.L. Hitchcock, M. Pudek, J.C. Prior, Progesterone therapy increases free thyroxine levels--data from a randomized placebo-controlled 12-week hot flush trial. Clin. Endocrinol. 79(2), 282–287 (2013). https://doi.org/10.1111/cen.12128
G. Trifiro, F. Parrino, J. Sultana, F. Giorgianni, C. Ferrajolo, E. Bianchini, G. Medea, S. Benvenga, I. Cricelli, C. Cricelli, F. Lapi, Drug interactions with levothyroxine therapy in patients with hypothyroidism: observational study in general practice. Clin. Drug. Investig. 35(3), 187–195 (2015). https://doi.org/10.1007/s40261-015-0271-0
B.R. Haugen, Drugs that suppress TSH or cause central hypothyroidism. Best Pract. Res. Clin. Endocrinol. Metab. 23(6), 793–800 (2009). https://doi.org/10.1016/j.beem.2009.08.003
M. Salas, A. Hofman, B.H. Stricker, Confounding by indication: an example of variation in the use of epidemiologic terminology. Am. J. Epidemiol. 149(11), 981–983 (1999)
L. Romanelli, E. Mhillaj, V. Cuomo, The importance of reporting unexpected drug failure. J. Pharmacovigil. 4(1), 1–2 (2016). https://doi.org/10.4172/2329-6887.1000e148
D. Pincus, T. Gomes, C. Hellings, H. Zheng, J.M. Paterson, M.M. Mamdani, D.N. Juurlink, A population-based assessment of the drug interaction between levothyroxine and warfarin. Clin. Pharmacol. Ther. 92(6), 766–770 (2012). https://doi.org/10.1038/clpt.2012.171
P.N. Taylor, A. Iqbal, C. Minassian, A. Sayers, M.S. Draman, R. Greenwood, W. Hamilton, O. Okosieme, V. Panicker, S.L. Thomas, C. Dayan, Falling threshold for treatment of borderline elevated thyrotropin levels-balancing benefits and risks: evidence from a large community-based study. JAMA Intern Med 174(1), 32–39 (2014). https://doi.org/10.1001/jamainternmed.2013.11312
Y. Ingrasciotta, J. Sultana, F. Giorgianni, A.P. Caputi, V. Arcoraci, D.U. Tari, C. Linguiti, M. Perrotta, A. Nucita, F. Pellegrini, A. Fontana, L. Cavagna, D. Santoro, G. Trifiro, The burden of nephrotoxic drug prescriptions in patients with chronic kidney disease: a retrospective population-based study in Southern Italy. PLoS. ONE. 9(2), e8907 (2014). https://doi.org/10.1371/journal.pone.0089072
W. Willett, An overview of issues related to the correction of non-differential exposure measurement error in epidemiologic studies. Stat. Med. 8(9), 1031–1040 (1989). discussion 1033–1071
I. Donangelo, G.D. Braunstein, Update on subclinical hyperthyroidism. Am. Fam. Physician. 83(8), 933–938 (2011)
A.M. Formenti, G. Mazziotti, R. Giubbini, A. Giustina, Treatment of hypothyroidism: all that glitters is gold? Endocrine 52(3), 411–413 (2016). https://doi.org/10.1007/s12020-016-0882-0
Funding
This study was supported by IBSA. The funding institution had no role in the design and conduct of the study, the collection, management, analysis, and interpretation of the data, the preparation, review, or approval of the manuscript, or the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. I. Cricelli and Dr. F. Lapi provided consultancies in protocol preparation for epidemiological studies and data analyses for Angelini, Alfa Wassermann, Bayer, and IBSA. Dr. C. Cricelli, and Dr. G. Medea provided clinical consultancies for Angelini, Alfa Wassermann, Bayer, and IBSA. Dr. V. Guglielmi, Dr. A. Bellia, Dr. E. Bianchini, Prof. P. Sbraccia, and Prof. D. Lauro, have no conflict of interest to disclose.
Ethical approval
This is an observational, retrospective, non-interventional study. According to a by-law on the classification and implementation of observational drug-related research, as issued by the Italian National Drug Agency (an entity belonging to the Italian Ministry of Health), the present study does not require approval by an Ethics Committee in Italy (Italian Drug Agency note of 3rd August 2007).
Additional information
Valeria Guglielmi and Alfonso Bellia equally contributed.
Rights and permissions
About this article
Cite this article
Guglielmi, V., Bellia, A., Bianchini, E. et al. Drug interactions in users of tablet vs. oral liquid levothyroxine formulations: a real-world evidence study in primary care. Endocrine 59, 585–592 (2018). https://doi.org/10.1007/s12020-017-1412-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-017-1412-4