Abstract
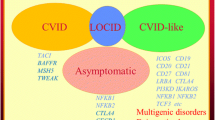
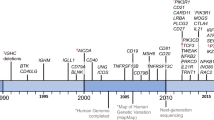
Common variable immunodeficiency disorders (CVID) are the most frequent symptomatic primary immune deficiency in adults. At this time, the causes of these conditions are unknown. Patients with CVID experience immune system failure consequent to late onset antibody failure. They have increased susceptibility to infections and are also at risk of severe autoimmune and inflammatory disorders as a result of immune dysregulation. An increasing number of monogenic causes as well as a digenic disorder have been described in patients with a CVID phenotype. If a causative mutation is identified, patients are removed from the umbrella diagnosis of CVID and are reclassified as having a CVID-like disorder, resulting from a specific mutation. In non-consanguineous populations, next-generation sequencing (NGS) identifies a genetic cause in approximately 25% of patients with a CVID phenotype. It is six years since we published our diagnostic criteria for CVID. There is ongoing debate about diagnostic criteria, the role of vaccine responses and genetic analysis in the diagnosis of CVID. There have been several recent studies, which have addressed some of these uncertainties. Here we review this new evidence from the perspective of our CVID diagnostic criteria and speculate on future approaches, which may assist in identifying and assessing this group of enigmatic disorders.

Similar content being viewed by others
References
Abbott JK, Gelfand EW (2015) Common variable immunodeficiency: diagnosis, management, and treatment. Immunol Allergy Clin N Am 35:637–658
Selenius JS, Martelius T, Pikkarainen S, Siitonen S, Mattila E, Pietikäinen R, Suomalainen P, Aalto AH, Saarela J, Einarsdottir E, Järvinen A, Färkkilä M, Kere J, Seppänen M (2017) Unexpectedly high prevalence of common variable immunodeficiency in Finland. Front Immunol 8:1190
Conley ME, Notarangelo LD, Etzioni A (1999) Diagnostic criteria for primary immunodeficiencies. Representing PAGID (Pan-American Group for Immunodeficiency) and ESID (European Society for Immunodeficiencies). Clin Immunol 93:190–197
Ameratunga R, Brewerton M, Slade C et al (2014) Comparison of diagnostic criteria for common variable immunodeficiency disorder. Front Immunol 5:415
Seppanen M, Aghamohammadi A, Rezaei N (2013) Is there a need to redefine the diagnostic criteria for common variable immunodeficiency? Expert Rev Clin Immunol. 2014 Jan;10(1):1–5
Ameratunga R, Woon ST, Gillis D, Koopmans W, Steele R (2013) New diagnostic criteria for common variable immune deficiency (CVID), which may assist with decisions to treat with intravenous or subcutaneous immunoglobulin. Clin Exp Immunol 174:203–211
Oksenhendler E, Gerard L, Fieschi C et al (2008) Infections in 252 patients with common variable immunodeficiency. Clin Infect Dis 46:1547–1554
Agarwal S, Cunningham-Rundles C (2007) Assessment and clinical interpretation of reduced IgG values. Ann Allergy Asthma Immunol 99:281–283
Chapel H, Cunningham-Rundles C (2009) Update in understanding common variable immunodeficiency disorders (CVIDs) and the management of patients with these conditions. Br J Haematol 145:709–727
Knight AK, Cunningham-Rundles C (2006) Inflammatory and autoimmune complications of common variable immune deficiency. Autoimmun Rev 5:156–159
Cunningham-Rundles C, Bodian C (1999) Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol 92:34–48
Chapel H, Lucas M, Lee M, Bjorkander J, Webster D, Grimbacher B, Fieschi C, Thon V, Abedi MR, Hammarstrom L (2008) Common variable immunodeficiency disorders: division into distinct clinical phenotypes. Blood 112:277–286
Wehr C, Kivioja T, Schmitt C, Ferry B, Witte T, Eren E, Vlkova M, Hernandez M, Detkova D, Bos PR, Poerksen G, von Bernuth H, Baumann U, Goldacker S, Gutenberger S, Schlesier M, Bergeron-van der Cruyssen F, le Garff M, Debre P, Jacobs R, Jones J, Bateman E, Litzman J, van Hagen PM, Plebani A, Schmidt RE, Thon V, Quinti I, Espanol T, Webster AD, Chapel H, Vihinen M, Oksenhendler E, Peter HH, Warnatz K (2008) The EUROclass trial: defining subgroups in common variable immunodeficiency. Blood 111:77–85
Olinder-Nielsen AM, Granert C, Forsberg P, Friman V, Vietorisz A, Bjorkander J (2007) Immunoglobulin prophylaxis in 350 adults with IgG subclass deficiency and recurrent respiratory tract infections: a long-term follow-up. Scand J Infect Dis 39:44–50
Abrahamian F, Agrawal S, Gupta S (2010) Immunological and clinical profile of adult patients with selective immunoglobulin subclass deficiency: response to intravenous immunoglobulin therapy. Clin Exp Immunol 159:344–350
Musher DM, Manof SB, Liss C et al (2010) Safety and antibody response, including antibody persistence for 5 years, after primary vaccination or revaccination with pneumococcal polysaccharide vaccine in middle-aged and older adults. J Infect Dis 201:516–524
Koopmans W, Woon ST, Brooks AE, Dunbar PR, Browett P, Ameratunga R (2013) Clinical variability of family members with the C104R mutation in transmembrane activator and calcium modulator and cyclophilin ligand interactor (TACI). J Clin Immunol 33:68–73
Grabenstein JD, Manoff SB (2012) Pneumococcal polysaccharide 23-valent vaccine: long-term persistence of circulating antibody and immunogenicity and safety after revaccination in adults. Vaccine 30:4435–4444
Tiller TL Jr, Buckley RH (1978) Transient hypogammaglobulinemia of infancy: review of the literature, clinical and immunologic features of 11 new cases, and long-term follow-up. J Pediatr 92:347–353
Pan-Hammarstrom Q, Salzer U, Du L et al (2007) Reexamining the role of TACI coding variants in common variable immunodeficiency and selective IgA deficiency. Nat Genet 39:429–430
Salzer U, Bacchelli C, Buckridge S, Pan-Hammarstrom Q, Jennings S, Lougaris V, Bergbreiter A, Hagena T, Birmelin J, Plebani A, Webster ADB, Peter HH, Suez D, Chapel H, McLean-Tooke A, Spickett GP, Anover-Sombke S, Ochs HD, Urschel S, Belohradsky BH, Ugrinovic S, Kumararatne DS, Lawrence TC, Holm AM, Franco JL, Schulze I, Schneider P, Gertz EM, Schaffer AA, Hammarstrom L, Thrasher AJ, Gaspar HB, Grimbacher B (2009) Relevance of biallelic versus monoallelic TNFRSF13B mutations in distinguishing disease-causing from risk-increasing TNFRSF13B variants in antibody deficiency syndromes. Blood 113:1967–1976
Popa V (1988) Lymphocytic interstitial pneumonia of common variable immunodeficiency. Ann Allergy 60:203–206
Ameratunga R, Becroft DM, Hunter W (2000) The simultaneous presentation of sarcoidosis and common variable immune deficiency. Pathology 32:280–282
Fasano MB, Sullivan KE, Sarpong SB, Wood RA, Jones SM, Johns CJ, Lederman HM, Bykowsky MJ, Greene JM, Winkelstein JA (1996) Sarcoidosis and common variable immunodeficiency. Report of 8 cases and review of the literature. Medicine (Baltimore) 75:251–261
Fuss IJ, Friend J, Yang Z, He JP, Hooda L, Boyer J, Xi L, Raffeld M, Kleiner DE, Heller T, Strober W (2013) Nodular regenerative hyperplasia in common variable immunodeficiency. J Clin Immunol 33:748–758
Malamut G, Ziol M, Suarez F, Beaugrand M, Viallard JF, Lascaux AS, Verkarre V, Bechade D, Poynard T, Hermine O, Cellier C (2008) Nodular regenerative hyperplasia: the main liver disease in patients with primary hypogammaglobulinemia and hepatic abnormalities. J Hepatol 48:74–82
Luzi G, Zullo A, Iebba F, Rinaldi V, Mete LS, Muscaritoli M, Aiuti F (2003) Duodenal pathology and clinical-immunological implications in common variable immunodeficiency patients. Am J Gastroenterol 98:118–121
Malamut G, Verkarre V, Suarez F, Viallard JF, Lascaux AS, Cosnes J, Bouhnik Y, Lambotte O, Béchade D, Ziol M, Lavergne A, Hermine O, Cerf-Bensussan N, Cellier C (2010) The enteropathy associated with common variable immunodeficiency: the delineated frontiers with celiac disease. Am J Gastroenterol 105:2262–2275
Agarwal S, Smereka P, Harpaz N, Cunningham-Rundles C, Mayer L (2011) Characterization of immunologic defects in patients with common variable immunodeficiency (CVID) with intestinal disease. Inflamm Bowel Dis 17:251–259
Bonilla FA, Barlan I, Chapel H, Costa-Carvalho BT, Cunningham-Rundles C, de la Morena MT, Espinosa-Rosales FJ, Hammarström L, Nonoyama S, Quinti I, Routes JM, Tang MLK, Warnatz K (2016) International Consensus Document (ICON): common variable immunodeficiency disorders. J Allergy Clin Immunol Pract 4:38–59
Gathmann B, Mahlaoui N, Gerard L et al (2014) Clinical picture and treatment of 2212 patients with common variable immunodeficiency. J Allergy Clin Immunol 134:116–126
Ameratunga R, Woon ST, Gillis D, Koopmans W, Steele R (2014) New diagnostic criteria for CVID. Expert Rev Clin Immunol 10:183–186
Ameratunga R, Ahn Y, Steele R, Woon ST (2019) Transient hypogammaglobulinemia of infancy: many patients recover in adolescence and adulthood. Clin Exp Immunol in press
Ameratunga R, Woon ST, Neas K, Love DR (2010) The clinical utility of molecular diagnostic testing for primary immune deficiency disorders: a case based review. Allergy, Asthma Clin Immunol 6:12
Ameratunga R, Ahn Y, Steele R, Woon S-T (2019) The natural history of untreated primary hypogammaglobulinemia in adults: implications for the diagnosis and treatment of common variable immunodeficiency disorders (CVID). Front Immunol. 17:10:1541
Smith J, Fernando T, McGrath N, Ameratunga R (2004) Lamotrigine-induced common variable immune deficiency. Neurology 62:833–834
Ameratunga R, Barker R, Steele R et al (2015) Profound reversible hypogammaglobulinemia caused by celiac disease in the absence of protein losing enteropathy. J Clin Immunol 35:589–594
Ameratunga RVCP, Parry S, Kenedi C (2019) Hypogammaglobulinemia factitia. Munchausen syndrome presenting as Common Variable Immune Deficiency. Front Immunol. 10:1541.
Duraisingham SS, Buckland MS, Grigoriadou S, Longhurst HJ (2014) Secondary antibody deficiency. Expert Rev Clin Immunol. 10(5):583–91.
Ameratunga R, Storey P, Barker R, Jordan A, Koopmans W, Woon ST (2015) Application of diagnostic and treatment criteria for common variable immunodeficiency disorder. Expert Rev Clin Immunol 12:257–266
Ameratunga R, Ahn Y, Jordan A, Lehnert K, Brothers S, Woon ST (2018) Keeping it in the family: the case for considering late onset combined immunodeficiency a subset of common variable immunodeficiency disorders. Expert Rev Clin Immunol 14:549–556
Ameratunga R (2018) Assessing disease severity in common variable immunodeficiency disorders (CVID) and CVID-like disorders. Front Immunol. 28:9:2130.
Agarwal S, Cunningham-Rundles C (2013) Treatment of hypogammaglobulinemia in adults: a scoring system to guide decisions on immunoglobulin replacement. J Allergy Clin Immunol 131:1699–1701
Yong PF, Thaventhiran JE, Grimbacher B (2011) “A rose is a rose is a rose,” but CVID is not CVID common variable immune deficiency (CVID), what do we know in 2011? Adv Immunol 111:47–107
Driessen GJ, Dalm VA, van Hagen PM et al (2013) Common variable immunodeficiency and idiopathic primary hypogammaglobulinemia: two different conditions within the same disease spectrum. Haematologica 98:1617–1623
Filion CA, Taylor-Black S, Maglione PJ, Radigan L, Cunningham-Rundles C (2018) Differentiation of common variable immunodeficiency from IgG deficiency. J Allergy Clin Immunol Pract. 7:1277–1284
Orange JS, Ballow M, Stiehm ER, Ballas ZK, Chinen J, de la Morena M, Kumararatne D, Harville TO, Hesterberg P, Koleilat M, McGhee S, Perez EE, Raasch J, Scherzer R, Schroeder H, Seroogy C, Huissoon A, Sorensen RU, Katial R (2012) Use and interpretation of diagnostic vaccination in primary immunodeficiency: a working group report of the Basic and Clinical Immunology Interest Section of the American Academy of Allergy, Asthma & Immunology. J Allergy Clin Immunol 130:S1–S24
Beck SC (2013) Making sense of serotype-specific pneumococcal antibody measurements. Ann Clin Biochem 50:517–519
Jokinen JT, Ahman H, Kilpi TM, Makela PH, Kayhty MH (2004) Concentration of antipneumococcal antibodies as a serological correlate of protection: an application to acute otitis media. J Infect Dis 190:545–550
Russell FM, Carapetis JR, Burton RL, Lin J, Licciardi PV, Balloch A, Tikoduadua L, Waqatakirewa L, Cheung YB, Tang MLK, Nahm MH, Mulholland EK (2011) Opsonophagocytic activity following a reduced dose 7-valent pneumococcal conjugate vaccine infant primary series and 23-valent pneumococcal polysaccharide vaccine at 12 months of age. Vaccine 29:535–544
O’Brien KL, Hochman M, Goldblatt D (2007) Combined schedules of pneumococcal conjugate and polysaccharide vaccines: is hyporesponsiveness an issue? Lancet Infect Dis 7:597–606
Ladhani S, Ramsay M, Flood J et al (2012) Haemophilus influenzae serotype B (Hib) seroprevalence in England and Wales in 2009. Euro Surveill 17
Ameratunga SN, Lennon DR, Entwistle B, Robinson E, Ameratunga RV (1997) The immunogenicity of Haemophilus influenzae: meningococcal protein conjugate vaccine in Polynesian and non-Polynesian New Zealand infants. J Paediatr Child Health 33:138–141
Orange JS, Belohradsky BH, Berger M, Borte M, Hagan J, Jolles S, Wasserman RL, Baggish JS, Saunders R, Grimbacher B (2012) Evaluation of correlation between dose and clinical outcomes in subcutaneous immunoglobulin replacement therapy. Clin Exp Immunol 169:172–181
Smith LL, Buckley R, Lugar P (2014) Diagnostic immunization with bacteriophage PhiX 174 in patients with common variable immunodeficiency/hypogammaglobulinemia. Front Immunol 5:410
Ferry BL, Misbah SA, Stephens P et al (2004) Development of an anti-Salmonella typhi Vi ELISA: assessment of immunocompetence in healthy donors. Clin Exp Immunol 136:297–303
Warnatz K, Denz A, Drager R et al (2002) Severe deficiency of switched memory B cells (CD27(+)IgM(-)IgD(-)) in subgroups of patients with common variable immunodeficiency: a new approach to classify a heterogeneous disease. Blood 99:1544–1551
Koopmans W, Woon ST, Zeng IS et al (2013) Variability of memory B cell markers in a cohort of common variable immune deficiency patients over six months. Scand J Immunol 77:470–475
Kuehn HS, Boisson B, Cunningham-Rundles C, Reichenbach J, Stray-Pedersen A, Gelfand EW, Maffucci P, Pierce KR, Abbott JK, Voelkerding KV, South ST, Augustine NH, Bush JS, Dolen WK, Wray BB, Itan Y, Cobat A, Sorte HS, Ganesan S, Prader S, Martins TB, Lawrence MG, Orange JS, Calvo KR, Niemela JE, Casanova JL, Fleisher TA, Hill HR, Kumánovics A, Conley ME, Rosenzweig SD (2016) Loss of B cells in patients with heterozygous mutations in IKAROS. N Engl J Med 374:1032–1043
Ameratunga R, Koopmans W, Woon ST, Leung E, Lehnert K, Slade CA, Tempany JC, Enders A, Steele R, Browett P, Hodgkin PD, Bryant VL (2017) Epistatic interactions between mutations of TACI (TNFRSF13B) and TCF3 result in a severe primary immunodeficiency disorder and systemic lupus erythematosus. Clin Transl Immunol 6:e159
Boisson B, Wang YD, Bosompem A, Ma CS, Lim A, Kochetkov T, Tangye SG, Casanova JL, Conley ME (2013) A recurrent dominant negative E47 mutation causes agammaglobulinemia and BCR(-) B cells. J Clin Invest 123:4781–4785
Ameratunga R, Lehnert K, Woon ST, Gillis D, Bryant VL, Slade CA, Steele R (2018) Review: diagnosing common variable immunodeficiency disorder in the era of genome sequencing. Clin Rev Allergy Immunol 54:261–268
Ameratunga R, Woon ST (2009) Customised molecular diagnosis of primary immune deficiency disorders in New Zealand: an efficient strategy for a small developed country. N Z Med J 122:46–53
Ameratunga R, Woon ST, Bryant VL, Steele R, Slade C, Leung EY, Lehnert K (2018) Clinical implications of digenic inheritiance and epistasis in primary immunodeficiency disorders. Front Immunol. 26:8:1965
Fliegauf ML, Bryant V, Frede N et al (2015) Haploinsufficiency of the NF-κB1 subunit p50 in common variable immunodeficiency. Am J Hum Genet 97:389–403
Maffucci P, Filion CA, Boisson B et al (2016) Genetic diagnosis using whole exome sequencing in common variable immunodeficiency. Front Immunol 7:220
Woon ST, Ameratunga R (2016) Comprehensive genetic testing for primary immunodeficiency disorders in a tertiary hospital: 10-year experience in Auckland, New Zealand. Allergy, Asthma Clin Immunol 12:65
Ameratunga R, Woon ST, Brewerton M, Koopmans W, Jordan A, Brothers S, Singh R (2011) Primary immune deficiency disorders in the South Pacific: the clinical utility of a customized genetic testing program in New Zealand. Ann N Y Acad Sci 1238:53–64
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424
Rodriguez-Cortez VC, Del Pino-Molina L, Rodriguez-Ubreva J et al (2015) Monozygotic twins discordant for common variable immunodeficiency reveal impaired DNA demethylation during naive-to-memory B-cell transition. Nat Commun 6:7335
Bateson (1909) Discussion on the influence of heredity on disease, with special reference to tuberculosis, cancer, and diseases of the nervous system: introductory address. Proc R Soc Med 2:22–30
Unger S, Seidl M, Schmitt-Graeff A, Böhm J, Schrenk K, Wehr C, Goldacker S, Dräger R, Gärtner BC, Fisch P, Werner M, Warnatz K (2014) Ill-defined germinal centers and severely reduced plasma cells are histological hallmarks of lymphadenopathy in patients with common variable immunodeficiency. J Clin Immunol 34:615–626
Romberg N, Le Coz C, Glauzy S et al (2019) Patients with common variable immunodeficiency with autoimmune cytopenias exhibit hyperplastic yet inefficient germinal center responses. J Allergy Clin Immunol 143:258–265
Ameratunga R, Lindsay K, Woon S-T, Jordan A, Anderson NE, Koopmans W (2015) New diagnostic criteria could distinguish common variable immunodeficiency disorder from anticonvulsant-induced hypogammaglobulinemia. Clin Exp Neuroimmunol 6:83–88
Thierry-Carstensen B, Jordan K, Uhlving HH, Dalby T, Sørensen C, Jensen AM, Heilmann C (2012) A randomised, double-blind, non-inferiority clinical trial on the safety and immunogenicity of a tetanus, diphtheria and monocomponent acellular pertussis (TdaP) vaccine in comparison to a tetanus and diphtheria (Td) vaccine when given as booster vaccinations to healthy adults. Vaccine 30:5464–5471
Hawdon N, Nix EB, Tsang RS, Ferroni G, McCready WG, Ulanova M (2012) Immune response to Haemophilus influenzae type b vaccination in patients with chronic renal failure. Clin Vaccine Immunol 19:967–969
Dentinger CM, Hennessy TW, Bulkow LR, Reasonover AL, Romero-Steiner S, Holder P, Leon PG, Carlone GM, Parks DJ, Parkinson AJ, Singleton RJ, Levine OS, Butler JC (2006) Immunogenicity and reactogenicity to Haemophilus influenzae type B (Hib) conjugate vaccine among rural Alaska adults. Hum Vaccin 2:24–28
Goldacker S, Draeger R, Warnatz K, Huzly D, Salzer U, Thiel J, Eibel H, Schlesier M, Peter HH (2007) Active vaccination in patients with common variable immunodeficiency (CVID). Clin Immunol 124:294–303
Lee H, Nahm MH, Kim KH (2010) The effect of age on the response to the pneumococcal polysaccharide vaccine. BMC Infect Dis 10:60
Ballow M (2012) Vaccines in the assessment of patients for immune deficiency. J Allergy Clin Immunol 130:283–284 e5
Henckaerts I, Goldblatt D, Ashton L, Poolman J (2006) Critical differences between pneumococcal polysaccharide enzyme-linked immunosorbent assays with and without 22F inhibition at low antibody concentrations in pediatric sera. Clin Vaccine Immunol 13:356–360
Concepcion NF, Frasch CE (2001) Pneumococcal type 22f polysaccharide absorption improves the specificity of a pneumococcal-polysaccharide enzyme-linked immunosorbent assay. Clin Diagn Lab Immunol 8:266–272
Balloch A, Licciardi PV, Tang ML (2013) Serotype-specific anti-pneumococcal IgG and immune competence: critical differences in interpretation criteria when different methods are used. J Clin Immunol 33:335–341
Jodar L, Butler J, Carlone G et al (2003) Serological criteria for evaluation and licensure of new pneumococcal conjugate vaccine formulations for use in infants. Vaccine 21:3265–3272
Lee LH, Frasch CE, Falk LA, Klein DL, Deal CD (2003) Correlates of immunity for pneumococcal conjugate vaccines. Vaccine 21:2190–2196
Bonilla FA, Bernstein IL, Khan DA, Ballas ZK, Chinen J, Frank MM, Kobrynski LJ, Levinson AI, Mazer B, Nelson RP Jr, Orange JS, Routes JM, Shearer WT, Sorensen RU (2005) Practice parameter for the diagnosis and management of primary immunodeficiency. Ann Allergy Asthma Immunol 94:S1–S63
Paris K, Sorensen RU (2007) Assessment and clinical interpretation of polysaccharide antibody responses. Ann Allergy Asthma Immunol 99:462–464
Rezaei N, Siadat SD, Aghamohammadi A, Moin M, Pourpak Z, Norouzian D, Mobarakeh JI, Aghasadeghi MR, Nejati M, Read RC (2010) Serum bactericidal antibody response 1 year after meningococcal polysaccharide vaccination of patients with common variable immunodeficiency. Clin Vaccine Immunol 17:524–528
Ochs HD, Davis SD, Wedgwood RJ (1971) Immunologic responses to bacteriophage phi-X 174 in immunodeficiency diseases. J Clin Invest 50:2559–2568
Wolf HM, Thon V, Litzman J, Eibl MM (2015) Detection of impaired IgG antibody formation facilitates the decision on early immunoglobulin replacement in hypogammaglobulinemic patients. Front Immunol 6:32
Funding
We thank A+ Trust, AMRF, ASCIA and IDFNZ for grant support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethics Approval
Ethics approval was not required for this review article. Studies referred to in this publication have complied with ethics requirements of the Health and Disability Ethics Committee (HDEC) of the NZ Ministry of Health and ADHB ethics committee. We thank our patients for participating in these studies for the benefit of others.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ameratunga, R., Woon, ST. Perspective: Evolving Concepts in the Diagnosis and Understanding of Common Variable Immunodeficiency Disorders (CVID). Clinic Rev Allerg Immunol 59, 109–121 (2020). https://doi.org/10.1007/s12016-019-08765-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-019-08765-6