Abstract
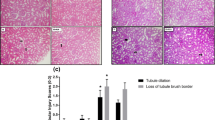
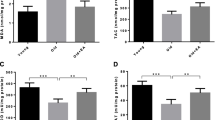
Despite the wide application of iron oxide nanoparticles (IONPs), little is known about the specific mechanism of their nephrotoxic effect. We aimed to evaluate the nephrotoxic effects of iron oxide nanoparticles (IONPs) in vivo and the protective effect of ellagic acid (EA) as a silent information regulator sirtuin 1 (SIRT1) activator against the induced nephrotoxicity. Forty male albino Wistar rats were randomly distributed into four equal groups (10 rats each): the control group (oral saline for 30 days), ellagic acid (EA) group (10 mg/kg b.w. EA, orally for 30 days), IONP group (20 mg/kg b.w. IONP I/P injection at the 24th–30th day), and EA + IONP group (10 mg/kg b.w./day EA for 30 days + 20 mg/kg b.w. IONPs at the 24th–30th day). In the present study, the potent antioxidant and antiapoptotic effects of EA were indicated by the significant overexpression of SIRT1 in renal tissues that leads to significant decreases in renal MDA content, P53 protein level and forkhead-box transcription factor1 (FOXO1) expression, and significant increases in renal GSH level, catalase activity, growth arrest and DNA damage-inducible protein 45 alpha (GADDα45), and renal inhibition of apoptosis protein (KIAP) gene expression levels in the EA + IONP-treated group. These results were confirmed by the improved histopathological renal features with EA administration. In conclusion, the present study provides the first evidence for the usefulness of EA as a sirtuin1 activator in the prevention or treatment of renal damage. Thus, EA could be used as a promising therapy for the prevention of IONP-induced nephrotoxicity.







Similar content being viewed by others
References
Oberdörster G, Oberdörster E, Oberdörster J (2005) Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect 113(7):823–839
Hagens WI, Oomen AG, de Jong WH, Casse FR, Sips AJ (2007) What do we need to know about the kinetic properties of nanoparticles in the body? Regul Toxicol Pharmacol 49(3):217–229
Khanna L, Verma NK, Tripathi SK (2018) Burgeoning tool of biomedical applications superparamagnetic nanoparticles. Alloys Compd 752:332–353. https://doi.org/10.1016/j.jallcom.2018.04.093
Xie W, Guo Z, Gao F, Gao Q, Wang D, Liaw BS, Cai Q, Sun X, Wang X, Zhao L (2018) Shape, size- and structure-controlled synthesis and biocompatibility of iron oxide nanoparticles for magnetic theranostics. Theranostics 8:3284–3307. https://doi.org/10.7150/thno.25220
Jahangirian H, Kalantari K, Izadiyan Z, Rafiee-Moghaddam R, Shameli K, Webster TJ (2019) A review of small molecules and drug delivery applications using gold and iron nanoparticles. Int J Nanomedicine 14:1633–1657
El-Boubbou K (2018) Magnetic iron oxide nanoparticles as drug carriers: clinical relevance. Nanomedicine 13:953–971
Dadfar SM, Roemhild K, Drude NI, Von Stillfried S, Knüchel R, Kiessling F, Lammers T (2019) Iron oxide nanoparticles: diagnostic, therapeutic and theranostic applications. Adv Drug Deliv Rev 138:302–325
Dulińska-Litewka J, Łazarczyk A, Hałubiec P, Szafrański O, Karnas K, Karewicz A (2019) Superparamagnetic iron oxide nanoparticles-current and prospective medical applications. Materials 12:617. https://doi.org/10.3390/ma12040617
Pinel S, Thomas N, Boura C, Barberi-Heyob M (2019) Approaches to physical stimulation of metallic nanoparticles for glioblastoma treatment. Adv Drug Deliv Rev 138:344–357
Mostafalou S, Mohammadi H, Ramazani A, Abdollahi M (2013) Different biokinetics of nanomedicines linking to their toxicity; an overview. Daru J Pharm Sci 21(1):14
Kumar A, Pandey AK, Singh SS, Shanker R, Dhawan A (2011) Cellular uptake and mutagenic potential of metal oxide nanoparticles in bacterial cells. Chemosphere 83:1124–1132. https://doi.org/10.1016/j.chemosphere.2011.01.025
Liu G, Gao J, Ai H, Chen X (2013) Applications and potential toxicity of magnetic iron oxide nanoparticles. Small 9(9–10):1533–1545
Zhu MT, Feng WY, Wang B, Wang TC, Gu YQ, Wang M, Wang Y, Ouyang H, Zhao YL, Chai ZF (2008) Comparative study of pulmonary responses to nano- and submicron-sized ferric oxide in rats. Toxicology 247(2–3):102–111
Ponka P (1999) Cellular iron metabolism. Kidney Int 69:S2–S11
Missaoui WN, Arnold RD, Cummings BS (2018) Toxicological status of nanoparticles: what we know and what we don’t know. Chem Biol Interact. https://doi.org/10.1016/j.cbi.2018.07.015
Poormoosavi SM, Najafzadehvarzi H, Behmanesh MA, Amirgholami R (2018) Protective effects of asparagus officinalis extract against bisphenol a- induced toxicity in Wistar rats. Toxicol Rep 5:427–433. https://doi.org/10.1016/j.toxrep.2018.02.010
Nagy G, Benko I, Kiraly G, Voros O, Tanczos B, Sztrik A (2015) Cellular and nephrotoxicity of selenium species. J Trace Elem Med Biol 30:160–170
Simko M, Mattsson MO (2010) Risks from accidental exposures to engineered nanoparticles and neurological health effects: a critical review. Part Fibre Toxicol 7:42. https://doi.org/10.1186/1743-8977-7-42
Kandeil MA, Mohammed ET, Hashem KS, Aleya L, Abdel-Daim MM (2019) Moringa seed extract alleviates titanium oxide nanoparticles (TiO2-NPs)-induced cerebral oxidative damage, and increases cerebral mitochondrial viability. Environ Sci Pollut Res:1–16. https://doi.org/10.1007/s11356-019-05514-2
Mohammed ET, Safwat GM (2019) Grape seed Proanthocyanidin extract mitigates titanium dioxide nanoparticle (TiO2-NPs)–induced hepatotoxicity through TLR-4/NF-κB signaling pathway. Biol Trace Elem Res:1–11. https://doi.org/10.1007/s12011-019-01955-5
Wu X, Tan Y, Mao H, Zhang M (2010) Toxic effects of iron oxide nanoparticles on human umbilical vein endothelial cells. Int J Nanomedicine 5:385–399. https://doi.org/10.2147/IJN.S10458
Wan R, Mo Y, Feng L, Chien S, Tollerud DJ, Zhang Q (2012) DNA damage caused by metal nanoparticles: involvement of oxidative stress and activation of ATM. Chem Res Toxicol 25:1402–1411. https://doi.org/10.1021/tx200513t
Soong YY, Barlow PJ (2006) Quantification of gallic acid and ellagic acid from longan (Dimocarpus longan Lour.) seed and mango (Mangifera indica L.) kernel and their effects on antioxidant activity. Food Chem 97:524–530
Hassoun EA, Vodhanel J, Abushaban A (2004) The modulatory effects of ellagic acid and vitamin E succinate on TCDD-induced oxidative stress in different brain regions of rats after subchronic exposure. J Biochem Mol Toxicol 18:196–203
Priyadarsini K, Khopde S, Kumar S, Mohan H (2002) Free radical studies of ellagic acid, a natural phenolic antioxidant. J Agric Food Chem 50:2200–2206
Fujimura L, Matsudo Y, Kang M, Takamori Y, Tokuhisa T, Hatano M (2004) Protective role of Nd1 in doxorubicin-induced cardiotoxicity. Cardiovasc Res 64:315–321
Singh K, Khanna AK, Chander R (1999) Hepatoprotective activity of ellagic acid against carbon tetrachloride induced hepatotoxicity in rats. Indian J Exp Biol 37:1025–1026
Sepúlveda L, Ascacio A, Rodriguez-Herrera R, Aguilera-Carbó A, Aguilar CN (2011) Ellagic acid: biological properties and biotecnological development for production processes. Afr J Biotechnol 10(22):4518–4523
Türk G, Çeribaşi AO, Sahna E, Ateşşahin A (2011) Lycopene and ellagic acid prevent testicular apoptosis induced by cisplatin. Phytomedicine 18(5):356–361
Palani S, Santhakumari D, Balachandar S, Ambika S (2015) Protective role of ellagic acid in modulating iron induced nephrotoxicity in rats. Int J Adv Res Biol Sci 2(8):35–42
Pari L, Sivasankari R (2008) Effect of ellagic acid on cyclosporineA-induced oxidative damage in the liver of rats. Fundam Clin Pharmacol 22(4):395–401
Horio Y, Hayashi T, Kuno A, Kunimoto R (2011) Cellular and molecular effects of sirtuins in health and disease. Clin Sci 121:191–203
Luo J, Nikolaev AY, Imai S, Chen D, Su F, Shiloh A, Guarente L, Gu W (2001) Negative control of p53 by Sir2alpha promotes cell survival under stress. Cell 107:137–148
Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y, Tran H, Ross SE, Mostoslavsky R, Cohen HY (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303:2011–2015
Yun JM, Chien A, Jialal I, Devaraj S (2012) Resveratrol up-regulates SIRT1 and inhibits cellular oxidative stress in the diabetic milieu: mechanistic insights. J Nutr Biochem 23(7):699–705. https://doi.org/10.1016/j.jnutbio.2011.03.012
Huang H, Tindall DJ (2007) Dynamic FoxO transcription factors. J Cell Sci 120:2479–2487
Greer EL, Brunet A (2005) FOXO transcription factors at the interface between longevity and tumor suppression. Oncogene. 24(50):7410–7425
Gusev AI, Kurlov AS (2008) Production of nanocrystalline powders by high-energy ball milling: model and experiment. Nanotechnology 19(26):265302. https://doi.org/10.1088/0957-4484/19/26/265302
Celik G, Semiz A, Karakurt S, Arslan S, Adali O, Sen A (2013) A comparative study for the evaluation of two doses of ellagic acid on hepatic drug metabolizing and antioxidant enzymes in the rat. Biomed Res Int 358945. https://doi.org/10.1155/2013/358945
Ma P, Luo Q, Chen J, Gan Y, Du J, Ding S, Xi Z, Yang X (2012) Intraperitoneal injection of magnetic Fe3O4-nanoparticle induces hepatic and renal tissue injury via oxidative stress in mice. Int J Nanomedicine 7:4809–4818. https://doi.org/10.2147/IJN.S34349
Šebeková K, Dušinská M, Simon Klenovics K, Kollárová R, Boor P, Kebis A, Staruchová M, Vlková B, Celec P, Hodosy J, Bačiak L, Tušková R, Beňo M, Tulinská J, Príbojová J, Bilaničová D, Pojana G, Marcomini A, Volkovová K (2014) Comprehensive assessment of nephrotoxicity of intravenously administered sodium-oleate-coated ultrasmall superparamagnetic iron oxide (USPIO) and titanium dioxide (TiO2) nanoparticles in rats. Nanotoxicology 8(2):142–157. https://doi.org/10.3109/17435390.2012.763147
Tietz NW, Finley P, Pruden E, Amerson A (1990) Clinical guide to laboratory tests. Saunders, Philadelphia, pp 232–233
Young DS (2001) Effects of drugs on clinical lab tests, 4th edn. AACC
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Albro PW, Corbett JT, Schroeder JL (1986) Application of the thiobarbiturate assay to the measurement of lipid peroxidation products in microsomes. J Biochem Biophys Methods 13:185–194
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Livak KJ, Thomas S (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-DDCt method. Methods 25:402–408
Bancroft JD, Gamble M (2002) Theory and practice of histological techniques, 5th edn. Churchill Livingstone Pub., Edinburgh, pp 172–175 pp 593-620
Pan X, Redding JE, Wiley PA, Wen L, McConnell JS, Zhang B (2010) Mutagenicity evaluation of metal oxide nanoparticles by the bacterial reverse mutation assay. Chemosphere 79:113–116. https://doi.org/10.1016/jchemosphere2009:12.056
McNamara K, Tofail SAM (2015) Nanosystems: the use of nanoalloys, metallic, bimetallic, and magnetic nanoparticles in biomedical applications. Phys Chem Chem Phys 17(42):27981–27995
Wilhelma C, Billoteya C, Rogerc JN, Bacria J, Gazeau F (2003) Intracellular uptake of anionic superparamagnetic nanoparticles as a function of their surface coating. Biomaterials 24:1001–1011
Cruz DN, Fard A, Clementi A, Ronco C, Maisel A (2012) Role of biomarkers in the diagnosis and management of cardio-renal syndromes. Semin Nephrol 32:79–92
Pujalte I, Passagne I, Brouillaud B, Treguer M, Durand E, Ohayon-Courtes C, L'Azou B (2011) Cytotoxicity and oxidative stress induced by different metallic nanoparticles on human kidney cells. Part Fibre Toxicol 8:10
Stern ST, McNeil SE (2008) Nanotechnology safety concerns revisited. Toxicol Sci 101:4–21
Liu Y, Chen Z, Wang J (2011) Systematic evaluation of biocompatibility of magnetic Fe3O4 nanoparticles with six different mammalian cell lines. J Nanopart Res 13:199–212
Chew DJ, DiBartola SP (1989) Diagnosis and pathophysiology of renal disease. In: Ettinger SJ (ed) Textbook of veterinary internal medicine. W.B. Saunders Company, Philadelphia, pp 1893–1961
Hanini A, Schmitt A, Kacem K, Chau F, Ammar S, Gavard J (2011) Evaluation of iron oxide nanoparticle biocompatibility. Int J Nanomedicine 6:787–794
Atessahín A, Çeríbaßi AO, Yuce A, Bulmus O, Çikim G (2007) Role of ellagic acid against cisplatin-induced nephrotoxicity and oxidative stress in rats. Basic Clin Pharmacol Toxicol 100:121–126. https://doi.org/10.1111/j.1742-7843.2006.00015.x
El-Garhy AM, Abd El-Raouf OM, El-Sayeh BM, Fawzy HM, Abdallah DM (2014) Ellagic acid antiinflammatory and antiapoptotic potential mediate renoprotection in cisplatin nephrotoxic rats. J Biochem Mol Toxicol 10:472–479. https://doi.org/10.1002/jbt.21587
Ahad A, Ganai AA, Mujeeb M, Siddiqui WA (2014) Ellagic acid, an NF-kB inhibitor, ameliorates renal function in experimental diabetic nephropathy. Chem Biol Interact 219:64–75
Sk UH, Bhattacharya S (2006) Prevention of cadmium induced lipid peroxidation, depletion of some antioxidative enzymes and glutathione by a series of novel organoselenocyanates. Environ Toxicol Pharmacol 22:298–230
Durairaj A, Vaiyapuri TS, Kanti MU (2008) Protective activity and antioxidant potential of Lippia nodiflora extract in paracetamol induced hepatotoxicity in rats. Iran J Pharmacol Ther 7:83–89
Gaharwar US, Paulraj R (2015) Iron oxide nanoparticles induced oxidative damage in peripheral blood cells of rat. J Biomed Sci Eng 8:274–286
Samal NK, Paulraj R (2010) Modulatory role of magnetic iron oxide nanoparticles on oxidative stress in rat. J Bionanosci 4:22–28. https://doi.org/10.1166/jbns.2010.1021
Ahamed M, Alhadlaq HA, Alam J, Khan MA, Ali D, Alarafi S (2013) Iron oxide nanoparticle-induced oxidative stress and genotoxicity in human skin epithelial and lung epithelial cell lines. Curr Pharm Des 19:6681–6690
Warpe VS, Mali VR, Arulmozhi S, Bodhankar SL, Mahadik KR (2015) Cardioprotective effect of ellagic acid on doxorubicin induced cardiotoxicity in wistar rats. J Acute Med 5:1–8
Kaur R, Parveen S, Mehan S, Khanna D, Kalra S (2015) Neuroprotective effect of ellagic acid against chronically scopolamine induced Alzheimer’s type memory and cognitive dysfunctions: possible behavioural and biochemical evidences 1(2):45–64
Iino T, Nakahara K, Miki W (2001) Less damaging effect of whisky in rat stomachs in comparison with pure ethanol. Role of ellagic acid, the nonalcoholic component. Digestion 64:214–221
Finkel SE, Kolter R (1999) Evolution of microbial diversity during prolonged starvation. Proc Natl Acad Sci U S A:4023–4027
Imai S (2009) Nicotinamide phosphoribosyltransferase (Nampt): a link between NAD biology, metabolism, and diseases. Curr Pharm Des 15:20–28
Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW (2004) Modulation of NF-kappa B-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J 23(12):2369–2380
Nakagawa S, Naganuma T, Shioi G, Hirose T (2011) Paraspeckles are subpopulation-specific nuclear bodies that are not essential in mice. J Cell Biol 193:31–39
Satoh N, Maniwa Y, Bermudez VP, Nishimura K, Nishio W, Yoshimura M, Okita Y, Ohbayashi C, Hurwitz J, Hayashi Y (2011) Oncogenic phosphatase Wip1 is a novel prognostic marker for lung adenocarcinoma patient survival. Cancer Sci 102(5):1101–1106. https://doi.org/10.1111/j.1349-7006.2011.01898.x
Ogawa T, Wakai C, Saito T, Murayama A, Mimura Y, Youfu S (2011) Distribution of the longevity gene product, SIRT1, in developing mouse organs. Congenit Anom (Kyoto) 51:70–79. https://doi.org/10.1111/j.1741-4520.2010.00304.x
Grubisha LC, Kretzer AM, Bruns TD (2005) Isolation and characterization of microsatellite loci from the truffle-like ectomycorrhizal fungi Rhizopogon occidentalis and Rhizopogon vulgaris. Mol Ecol Notes 5:608–610
Dong YJ, Liu N, Xiao Z, Sun T, Wu SH, Sun WX (2014) Renal protective effect of sirtuin 1. J Diabetes 843786:8. https://doi.org/10.1155/2014/843786
Tikoo K, Tripathi DN, Kabra DG, Sharma V, Gaikwad AB (2007) Intermittent fasting prevents the progression of type I diabetic nephropathy in rats and changes the expression of Sir2 and p53. FEBS Lett 581:1071–1078
de Kreutzenberg SV, Ceolotto G, Papparella I, Bortoluzzi A, Semplicini A, Dalla Man C, Cobelli C, Fadini GP, Avogaro A (2010) Downregulation of the longevity-associated protein sirtuin 1 in insulin resistance and metabolic syndrome: potential biochemical mechanisms. Diabetes 59(4):1006–1015. https://doi.org/10.2337/db09-1187
Deng L, Kakihara T, Fukuda R, Ohta A (2007) Isolation and characterization of a mutant defective in utilization of exogenous phosphatidylethanolamine in Saccharomyces cerevisiae. J Gen Appl Microbiol 53(4):255–258
Hori T, Shang WH, Takeuchi K, Fukagawa T (2013) The CCAN recruits CENP-A to the centromere and forms the structural core for kinetochore assembly. J Cell Biol 200:45–60
Achanta G, Huang P (2004) Role of p53 in sensing oxidative DNA damage in response to reactive oxygen species-generating agents. Cancer Res 64:6233–6239
Vousden KH (2000) p53: death star. Cell 101:691–694
Lu H, Huang H (2011) FOXO1: a potential target for human diseases. Curr Drug Targets 12(9):1235–1244
Wang Y, Zhou Y, Graves DT (2014) FOXO transcription factors: their clinical significance and regulation. Biomed Res Int 2014:925350. https://doi.org/10.1155/2014/925350
Zhan Q (2005) Gadd45a, a p53- and BRCA1-regulated stress protein, in cellular response to DNA damage. Mutat Res 569(1–2):133–143
Hollander MC, Sheikh MS, Bulavin D, Lundren K, Augeri-Henmueller L, Shehee R, Molinaro T, Kim K, Tolosa E, Ashwell JD (1999) Genomic instability in Gadd45a-deficient mice. Nat Genet 23:176–184
Wingert S, Frederic BT, Nadine H, Maike R, Timm S, Michael AR (2016) DNA-damage response gene GADD45A induces differentiation in hematopoietic stem cells without inhibiting cell cycle or survival. Stem Cells 34:699–710
Schmitz KM, Schmitt N, Hoffmann-Rohrer U, Schäfer A, Grummt I, Mayer C (2009) TAF12 recruits Gadd45a and the nucleotide excision repair complex to the promoter of rRNA genes leading to active DNA demethylation. Mol Cell 33:344–353
Li D, Dai C, Zhou Y, Yang X, Zhao K, Xiao X, Tang S (2016) Effect of GADD45a on olaquindox-induced apoptosis in human hepatoma G2 cells: involvement of mitochondrial dysfunction. Environ Toxicol Pharmacol 46:140–146. https://doi.org/10.1016/j.etap.2016.07.012
Takekawa M, Saito H (1998) A family of stress-inducible GADD45-like proteins mediate activation of the stress-responsive MTK1/MEKK4 MAPKKK. Cell 95:521–530
Hildesheim A, Wang SS (2002) Host and viral genetics and risk of cervical cancer. Virus Res 89(2):229–240
Kobayashi Y, Furukawa-Hibi Y, Chen C, Horio Y, Isobe K, Ikeda K, Motoyama N (2005) SIRT1 is critical regulator of FOXO-mediated transcription in response to oxidative stress. Int J Mol Med 16(2):237–243
Motta MC, Divecha N, Lemieux M, Kamel C, Chen D, Gu W, Bultsma Y, McBurney M, Guarente L (2004) Mammalian SIRT1 represses forkhead transcription factors. Cell 116:551–563
Karlsson HL, Gustafsson J, Cronholm P, Möller L (2009) Size-dependent toxicity of metal oxide particles—a comparison between nano- and micrometer size. Toxicol Lett 188:112–118. https://doi.org/10.1016/j.toxlet.2009.03.014
Wagener N, Crnković-Mertens I, Vetter C, Macher-Göppinger S, Bedke J, Gröne EF, Hoppe-Seyler F (2007) Expression of inhibitor of apoptosis protein Livin in renal cell carcinoma and non-tumorous adult kidney. Br J Cancer 97(9):1271–1276. https://doi.org/10.1038/sj.bjc.6604028
Lin JH, Deng G, Huang Q, Morser J (2000) KIAP, a novel member of the inhibitor of apoptosis protein family 279:820–831
Crnkovic-Mertens I, Hoppe-Seyler F, Butz K (2003) Induction of apoptosis in tumor cells by siRNA-mediated silencing of the livin/ML-IAP/KIAP gene. Oncogene 22(51):8330–8336
Alcendor RR, Gao S, Zhai P, Zablocki D, Holle E, Yu X, Tian B, Wagner T, Vatner SF, Sadoshima J (2007) Sirt1 regulates aging and resistance to oxidative stress in the heart. Circ Res 100:1512–1521
Acknowledgments
The authors thank all staff members of the Biochemistry and Histology departments, Faculty of Veterinary Medicine, Beni-Suef University, Egypt, for their help and advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohammed, E.T., Hashem, K.S., Abdelazem, A.Z. et al. Prospective Protective Effect of Ellagic Acid as a SIRT1 Activator in Iron Oxide Nanoparticle-Induced Renal Damage in Rats. Biol Trace Elem Res 198, 177–188 (2020). https://doi.org/10.1007/s12011-020-02034-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02034-w