Abstract
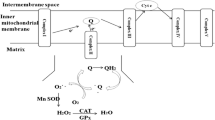
Autism is a neurodevelopmental disorder of childhood with poorly understood etiology and pathology. This pilot study aims to evaluate the levels of antioxidant enzymes, superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px), and levels of malondialdehyde (MDA), a marker of lipid peroxidation, in Egyptian autistic children. Autism is a neurodevelopmental disorder of childhood with poorly understood etiology and pathology. The present study included 20 children with autism diagnosed by DSM-IV-TR criteria and Childhood Autism Rating Scale. Controls included 25 age-matched healthy children. Cases were referred to Outpatient Clinic of Children with Special Needs Department, National Research Center, Cairo, Egypt. We compared levels of SOD, GSH-Px, and MDA in children with autism and controls. In children less than 6 years of age, levels of SOD, and GSH-Px were significantly lower in autistic children compared with their controls, while MDA was significantly higher among patients than controls. In children older than 6 years, there was no significant difference in any of these values between cases and controls. We concluded that children with autism are more vulnerable to oxidative stress in the form of increased lipid peroxidation and deficient antioxidant defense mechanism especially at younger children. We highlight that autistic children might benefit from antioxidants supplementation coupled with polyunsaturated fatty acids. Moreover, early assessment of antioxidant status would have better prognosis as it may decrease the oxidative stress before inducing more irreversible brain damage.
Similar content being viewed by others
References
Bölte S, Poustka F (2002) The relation between general cognitive level and adaptive behavior domains in individuals with autism with and without co-morbid mental retardation. Child Psychiatry Hum Dev 33(2):165–172
Blaxill M, Redwood L, Bernard S (2004) Thimerosal and autism? A plausible hypothesis that should be dismissed. Med Hypotheses 62:788–794
Sung YJ, Dawson G, Munson J, Estes A, Schellenberg GD, Wijsman EM (2005) Genetic investigation of quantitative traits related to autism: use of multivariate polygenic models with ascertainment adjustment. Am J Hum Genet 76:68–81
Fombonne E, Zakarian R, Bennett A, Meng L, McLean-Heywood D (2006) Pervasive developmental disorders in Montreal, Quebec, Canada: prevalence and links with immunizations. Pediatrics 118(1):e139–e150
Kern JK, Jones AM (2006) Evidence of toxicity, oxidative stress, and neuronal insult in autism. J Toxicol Environ Health B Crit Rev 9(6):485–499
Ming X, Johnson WG, Stenroos ES, Mars A, Lambert GH, Buyske S (2010) Genetic variant of glutathione peroxidase 1 in autism. Brain Dev 32(2):105–109
Ng F, Berk M, Dean O, Bush AI (2009) Oxidative stress in psychiatric disorders: evidence base and therapeutic implications. Int J Neuropsychopharmacol 11(6):851–876
Söğüt S, Zoroğlu SS, Ozyurt H, Yilmaz HR, Ozuğurlu F, Sivasli E, Yetkin O, Yanik M, Tutkun H, Savaş HA, Tarakçioğlu M, Akyol O (2003) Changes in nitric oxide levels and antioxidant enzyme activities may have a role in the pathophysiological mechanisms involved in autism. Clin Chim Acta 331(1–2):111–117
Perry G, Nunomura A, Harris S, Smith M, Salomon R (2005) Is Autism a disease of oxidative stress. Oxidative stress in autism symposium, New York State, Institute for basic research in Developmental Disabilities. p. 15
James SJ, Cutler P, Melnyk S, Hernigan S, Janak L, Gaylor DW, Neubrander JA (2004) Metabolic biomarkers of increased oxidative stress and methylation capacity in children with autism. Am J Clin Nutr 80(6):1611–1617
Chauhan A, Chauhan V (2006) Oxidative stress in autism. Pathophysiology 13(3):171–181
American Psychiatric Association (APA) (2000) Diagnostic and statistical manual of mental disorders, 4th edn. Text revision, Washington, DC
Schopler E, Reichler RJ, DeVellis RF, Daly K (1980) Toward objective classification of childhood autism: childhood autism rating scale (CARS). J Autism Dev Disord 10(1):91–103
Tanner JM, Hiernaux J, Jarman S (1969) Growth and physique studies. In: Weiner JS, Lourie JA (eds) Human biology: a guide to field methods, I.B.P. handbook No.9. Blackwell Scientific Publications, Oxford and Edinburgh, pp 1–29
Arthur JR, Boyne R (1985) Superoxide dismutase and glutathione peroxidase activities in neutrophils from selenium deficient and copper deficient cattle. Life Sci 36(16):1569–1575
Anderson PH, Berret S, Patterson DS (1978) Glutathione peroxidase activity in erythrocytes and muscle of cattle and sheep and its relationship to selenium. J Comp Pathol 88(2):181–189
Chauhan VPS, Tsiouris JA, Chauhan A, Sheikh AM, Brown WT, Vaughan M (2002) Increased oxidative stress and decreased activities of Ca2+/Mg2+—ATPase and Na+/K +—ATPase in the red blood cells of the hibernating black bear. Life Sci 71:153–161
Jain SK (1989) Hyperglycemia can cause membrane lipid peroxidation and osmotic fragility in human red blood cells. J Biol Chem 264(35):21340–21345
Coplan J, Jawad A (2005) Modeling clinical outcome of children with autistic spectrum disorders. Pediatrics 116(1):117–122
Charman T, Taylor E, Drew A, Cockerill H, Brown JA, Baird G (2005) Outcome at 7 years of children diagnosed with autism at age 2: predictive validity of assessments conducted at 2 and 3 years of age and pattern of symptom change over time. J Child Psychol Psychiatry 46(5):500–513
Yorbik O, Sayal A, Akay C, Akbiyik DI, Sohmen T (2002) Investigations of antioxidant enzymes in children with autistic disorder. Prostaglandins Leukot Essent Fatty Acids 67(5):341–343
Chauhan A, Chauhan V, Brown WT, Cohen I (2004) Oxidative stress in autism: increased lipid peroxidation and reduced serum levels of ceruloplasmin and transferrin—the antioxidant proteins. Life Sci 75(21):2539–2549
Klein JA, Ackerman SL (2003) Oxidative stress, cell cycle, and neurodegeneration. J Clin Invest 111(6):785–793
Chauhan A, Chauhan V, Cohen IL, Brown WT (2005) Increased lipid peroxidation and membrane rigidity in autism: relationship with behavior abnormalities, oxidative stress. In autism symposium, Institute for Basic Research in Developmental Disabilities Staten Island, New York
Boso M, Emanuele E, Minoretti P, Arra M, Politi P, Ucelli di Nemi S, Barale F (2006) Alterations of circulating endogenous secretory RAGE and S100A9 levels indicating dysfunction of the AGE-RAGE axis in autism. Neurosci Lett 410(3):169–173
Emanuele E, Boso M, Brondino N, Pietra S, Barale F, Ucelli di Nemi S, Politi P (2010) Increased serum levels of high mobility group box 1 protein in patients with autistic disorder. Prog Neuro-Psychopharmacol Biol Psychiatry 34:681–683
Klevay LM (2003) Advances in cardiovascular-copper research. In: Schrauzer GN, (ed) First international bio-minerals symposium: trace elements in nutrition, health and disease, Institute Rosell, Montreal, Canada
Powell SR (2000) The antioxidant properties of zinc. J Nutr 130:1447–1454
Erden-Inal M, Sunal E, Kanbak G (2002) Age related changes in the glutathione redox system. Cell Biochem Funct 20(1):61–66
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meguid, N.A., Dardir, A.A., Abdel-Raouf, E.R. et al. Evaluation of Oxidative Stress in Autism: Defective Antioxidant Enzymes and Increased Lipid Peroxidation. Biol Trace Elem Res 143, 58–65 (2011). https://doi.org/10.1007/s12011-010-8840-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-010-8840-9