Abstract
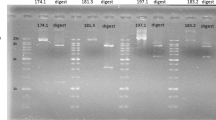
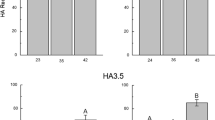
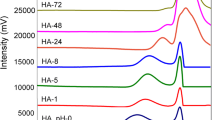
The effects of ethanol/broth proportions and the number of steps at varying pH in the presence or absence of sodium chloride (NaCl) were studied as precipitation strategies for the recovery and purification of high molar mass bio-hyaluronic acid (Bio-HA). Bio-HA was synthesized by Streptococcus zooepidemicus in a culture medium containing glucose and soy peptones. A single-step precipitation was more attractive than multistep precipitation in terms of recovery and purity as well as decreased use of ethanol. The best conditions in the absence and presence of salt were 2:1 ethanol/broth (v/v) at pH 4 (55.0 ± 0.2% purity and 85.0 ± 0.7% recovery) and 2:1 ethanol/broth (v/v) at pH 7 + 2 mol L−1 NaCl (59.0 ± 0.9% purity and 82.0 ± 4.3% recovery). Dynamic light scattering (DLS) spectra showed different particle sizes as a consequence of the changes in the molecular structure of HA, mainly with changes in pH. Although slight changes in distribution were observed, the average HA molar mass was not affected by the precipitation strategy, remaining on the order of 105 Da. Therefore, pH and NaCl modulated the precipitation performance of HA. These findings are relevant to further optimizing the precipitation step, thus minimizing costs in the later stages of HA purification.










Similar content being viewed by others
References
Chong, B. F., Blank, L. M., Mclaughlin, R., & Nielsen, L. K. (2005). Microbial hyaluronic acid production. Applied Microbiology and Biotechnology, 66(4), 341–351.
Kogan, G., Šoltés, L., Stern, R., & Gemeiner, P. (2007). Hyaluronic acid: A natural biopolymer with a broad range of biomedical and industrial applications. Biotechnology Letters, 29, 17–25.
Liang, T. W., Wu, C. C., Cheng, W. T., Chen, Y. C., Wang, C. L., Wang, I. L., & Wang, S. L. (2014). Exopolysaccharides and antimicrobial biosurfactants produced by Paenibacillus macerans TKU029. Applied Biochemistry and Biotechnology, 172(2), 933–950.
Cui, Y., Li, Y., Duan, Q., & Kakuchi, T. (2013). Preparation of hyaluronic acid micro-hydrogel by biotin-avidin-specific bonding for doxorubicin-targeted delivery. Applied Biochemistry and Biotechnology, 169(1), 239–249.
Choi, S. B., Lew, L. C., Hor, K. C., & Liong, M. T. (2014). Fe2+ and Cu2+ increase the production of hyaluronic acid by Lactobacilli via affecting different stages of the pentose phosphate pathyway. Applied Biochemistry and Biotechnology, 173(1), 129–142.
Pires, A. M. B., & Santana, M. H. A. (2010). Metabolic effects of the initial glucose concentration on microbial production of hyaluronic acid. Applied Biochemistry and Biotechnology, 162(6), 1751–1761.
Benedini, L. J., & Santana, M. H. A. (2013). Effects of soy peptone on the inoculum preparation of Streptococcus zooepidemicus for production of hyaluronic acid. Bioresource Technology, 130, 798–800.
Scopes, R. K. (1988). Protein purification (2nd ed.). New York: Springer-verlag Inc.
Ferrari, F. A., Motta, F. L., Bastos, R. G., & Santana, M. H. A. (2013). The solid-state cultivation of Streptococcus zooepidemicus in polyurethane foam as a strategy for the production of hyaluronic acid. Applied Biochemistry and Biotechnology, 170(6), 1491–1502.
Pan, N. C., Pereira, H. C. B., Corradi, M. L., Vasconcelos, A. F. D., & Celligoi, M. A. P. C. (2017). Improvement production of hyaluronic acid by Streptococcus zooepidemicus in sugarcane molasses. Applied Biochemistry and Biotechnology, 182(1), 276–293.
Han, H. Y., Jang, S. H., Kim, E. C., Park, J. K., Han, Y. J., Lee, C., Park, H. S., Kim, Y. C. & Park, H. J. (2004) Patent WO2004016771.
Won, T. Y., Lee, C. & Seo, S. H. (2008) Patent WO2008062998.
Kim, T. H., Kim, H. L., Park, S. Y. & Jang, D. K. (2008) Patent US20080194810.
Murado, M. A., Montemayor, M. I., Cabo, M. L., Vázquez, J. A., & González, M. P. (2012). Optimization of extraction and purification process of hyaluronic acid from fish eyeball. Food and Bioproducts Processing, 90(3), 491–498.
Nimrod, A., Greenman, B., Kanner, D., Landsberg, M. & Beck, Y. (1988) United States Patent 4780414.
Sousa, A. S., Guimarães, A. P., Gonçalves, C. V., Silva, I. J., Cavalcante, C. L., & Azevedo, D. C. S. (2009). Purification and characterization of microbial hyaluronic acid by solvent precipitation and size-exclusion chromatography. Separation Science and Technology, 44(4), 906–923.
Soltes, L., & Mendichi, R. (2003). Molecular characterization of two host-guest associating hyaluronan derivatives. Biomedical Chromatography, 17(6), 376–384.
Gatej, I., Popa, M., & Rinaudo, M. (2004). Role of the pH on Hyaluronan behavior in aqueous solution. Biomacromolecules, 6, 61–67.
Chakrabarti, B., & Balazs, E. A. (1973). Optical properties of hyaluronic acid: Ultraviolet circular dichroism and optical rotatory dispersion. Journal of Molecular Biology, 78(1), 135–141.
Staskus, P. W., & Johnson, W. C. (1988). Double-stranded structure for hyaluronic acid in ethanol-aqueous solution as revealed by circular dichroism of oligomers. Biochemistry, 27(5), 1528–1534.
Cowman, M. K., & Matsuoka, S. (2005). Experimental approaches to hyaluronan structure. Carbohydrate Research, 340(5), 791–809.
Reddy, K. J., & Karunakaran, K. T. (2013). Purification and characterization of hyaluronic acid produced by Streptococcus zooepidemicus strain 3523-7. Journal of BioScience and Biotechnology, 2, 173–179.
Rangaswamy, V., & Jain, D. (2008). An efficient process for production and purification of hyaluronic acid from Streptococcus equi subsp. zooepidemicus. Biotechnology Letters, 30(3), 493–496.
Chen, Y. H., & Wang, Q. (2009). Establishment of CTAB Turbidimetric method to determine hyaluronic acid content in fermentation broth. Carbohydrate Polymers, 78(1), 178–181.
Callou, K. R. A., Sadigov, S., Lajolo, F. M., & Genovese, M. I. (2010). Isoflavones and antioxidant capacity of Comercial soy-based beverages: Effect of storage. Journal of Agricultural and Food Chemistry, 58(7), 4284–4291.
Smith, P. K., Krohn, R. I., Hermanson, G. T., Mallia, A. K., Gartner, F. H., Provenzano, M. D., Fujimoto, E. K., Goeke, N. M., Olson, B. J., & Klenk, D. C. (1985). Measurement of protein using bicinchoninic acid. Analytical Biochemistry, 150(1), 76–85.
Veja, J. A. (2017). Biología y bases racionales para el uso terapéutico del hialuronano en las artropatías: el tamaño es importante. J Cartillage Disease, 4, 11–18.
Funding
This work was supported by the following Brazilian financial institutions: Conselho Nacional de Pesquisa [CNPq-Project no: 142480/2014-2] and Fundação de Amparo a Pesquisa do Estado de São Paulo [FAPESP–Project no: 2015/23134-8].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cavalcanti, A.D.D., Melo, B.A.G., Oliveira, R.C. et al. Recovery and Purity of High Molar Mass Bio-hyaluronic Acid Via Precipitation Strategies Modulated by pH and Sodium Chloride. Appl Biochem Biotechnol 188, 527–539 (2019). https://doi.org/10.1007/s12010-018-02935-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-018-02935-6