Abstract
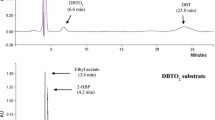
Numerous desulfurizing bacteria from the Rhodococcus genus harbor conserved dsz genes responsible for the degradation of sulfur compounds through 4S pathway. This study describes a newly identified desulfurizing bacterium, Rhodococcus sp. FUM94, which unlike previously identified strains encodes a truncated dsz operon. DNA sequencing revealed a frameshift mutation in the dszA gene, which led to an alteration of 66 amino acids and deletion of other C-terminal 66 amino acids. The resulting DszA polypeptide was shorter than DszA in Rhodococcus sp. IGTS8 reference strain. Despite the truncation, desulfurizing activity of the operon was observed and attributed to the removal of an overlap of dszA and dszB genes, and lack of active site in the altered region. Desulfurization experiments resulted in specific production rate of 6.3 mmol 2-hydroxy biphenyl (kgDCW)−1 h−1 at 2 g l−1 biocatalyst concentration and 68.8% biodesulfurization yield at 20 g l−1 biocatalyst concentration, both at 271 μM dibenzothiophene concentration which is comparable to similar wild-type biocatalysts.






Similar content being viewed by others
Change history
25 January 2018
The original version of this article unfortunately contained a mistake in the caption of Figs. 5 and 6.
References
McFarland, B. L. (1999). Biodesulfurization. Current Opinion in Microbiology, 2, 257–264. https://doi.org/10.1016/S1369-5274(99)80045-9.
Borgne, L. S., & Quintero, R. (2003). Biotechnological processes for the refining of petroleum. Fuel Processing Technology, 81, 155–169. https://doi.org/10.1016/S0378-3820(03)00007-9.
Boniek, D., Figueiredo, D., dos Santos, A. F. B., & Stoianoff, M. A. R. (2015). Biodesulfurization: a mini review about the immediate search for the future technology. Clean Technologies and Environmental Policy, 17, 29–37. https://doi.org/10.1007/s10098-014-0812-x.
Kilbane, J. J. (1990). Sulfur-specific microbial metabolism of organic compounds. Resources, Conservation and Recycling, 3, 69–79. https://doi.org/10.1016/0921-3449(90)90046-7.
Denome, S. A., Olson, E. S., & Young, K. D. (1993). Identification and cloning of genes involved in specific desulfurization of dibenzothiophene by Rhodococcus sp. strain IGTS8. Applied and Environmental Microbiology, 59, 2837–2843.
Denome, S. A., Oldfield, C., Nash, L. J., & Young, K. D. (1994). Characterization of the desulfurization genes from Rhodococcus sp. strain IGTS8. Journal of Bacteriology, 176, 6707–6716. https://doi.org/10.1128/jb.176.21.6707-6716.1994.
Piddington, C. S., Kovacevich, B. R., & Rambosek, J. (1995). Sequence and molecular characterization of a DNA region encoding the dibenzothiophene desulfurization operon of Rhodococcus sp. strain IGTS8. Applied and Environmental Microbiology, 61, 468–475.
Gray, K. A., Pogrebinsky, O. S., Mrachko, G. T., Xi, L., Monticello, D. J., & Squires, C. H. (1996). Molecular mechanisms of biocatalytic desulfurization of fossil fuels. Nature Biotechnology, 14, 1705–1709. https://doi.org/10.1038/nbt1296-1705.
Tanaka, Y., Matsui, T., Konishi, J., Maruhashi, K., & Kurane, R. (2002). Biodesulfurization of benzothiophene and dibenzothiophene by a newly isolated Rhodococcus strain. Applied Microbiology and Biotechnology, 59, 325–328. https://doi.org/10.1007/s00253-002-0985-9.
Yu, B., Xu, P., Shi, Q., & Ma, C. (2006). Deep desulfurization of diesel oil and crude oils by a newly isolated Rhodococcus erythropolis strain. Applied and Environmental Microbiology, 72, 54–58. https://doi.org/10.1128/AEM.72.1.54-58.2006.
Mohebali, G., Ball, A. S., Rasekh, B., & Kaytash, A. (2007). Biodesulfurization potential of a newly isolated bacterium, Gordonia alkanivorans RIPI90A. Enzyme and Microbial Technology, 40, 578–584. https://doi.org/10.1016/j.enzmictec.2006.05.012.
Davoodi-Dehaghani, F., Vosoughi, M., & Ziaee, A. A. (2010). Biodesulfurization of dibenzothiophene by a newly isolated Rhodococcus erythropolis strain. Bioresource Technology, 101, 1102–1105. https://doi.org/10.1016/j.biortech.2009.08.058.
Denis-Larose, C., Labbe, D., Bergeron, H., Jones, A., Greer, C. W., Hawari, J., Grossman, M. J., Sankey, B. M., & Lau, P. C. K. (1997). Conservation of plasmid-encoded dibenzothiophene desulfurization genes in several Rhodococci. Applied and Environmental Microbiology, 63, 2915–2919.
Duarte, G. F., Rosado, A. S., Seldin, L., De Araujo, W., & Van Elsas, J. D. (2001). Analysis of bacterial community structure in sulfurous-oil-containing soils and detection of species carrying dibenzothiophene desulfurization (dsz) genes. Applied and Environmental Microbiology, 67, 1052–1062. https://doi.org/10.1128/AEM.67.3.1052-1062.2001.
Kilbane, J. J., & Robbins, J. (2007). Characterization of the dszABC genes of Gordonia amicalis F.5.25.8 and identification of conserved protein and DNA sequences. Applied Microbiology and Biotechnology, 75, 843–851.
Li, G. Q., Li, S. S., Zhang, M. L., Wang, J., Zhu, L., Liang, F. L., Liu, R. L., & Ma, T. (2008). Genetic rearrangement strategy for optimizing the dibenzothiophene biodesulfurization pathway in Rhodococcus erythropolis. Applied and Environmental Microbiology, 74, 971–976. https://doi.org/10.1128/AEM.02319-07.
Izumi, Y., Ohshiro, T., Ogino, H., Hine, Y., & Shimao, M. (1994). Selective desulfurization of dibenzothiophene by Rhodococcus erythropolis D-1. Applied and Environmental Microbiology, 60, 223–226.
Sambrook, J., & Russell, D. W. (2001). Molecular cloning: a laboratory manual (Third ed.). Cold Spring Harbor: Cold Spring Harbor Laboratory.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685. https://doi.org/10.1038/227680a0.
Maghsoudi, S., Vossoughi, M., Kheirolomoom, A., Tanaka, E., & Katoh, S. (2001). Biodesulfurization of hydrocarbons and diesel fuels by Rhodococcus sp. strain P32C1. Biochemical Engineering Journal, 8, 151–156. https://doi.org/10.1016/S1369-703X(01)00097-3.
Caro, A., Boltes, K., Leton, P., & Garcia-Calvo, E. (2007). Dibenzothiophene biodesulfurization in resting cell conditions by aerobic bacteria. Biochemical Engineering Journal, 35, 191–197. https://doi.org/10.1016/j.bej.2007.01.013.
Oldfield, C., Pogrebinsky, O., Simmonds, J., Olson, E. S., & Kulpa, C. F. (1997). Elucidation of the metabolic pathway for dibenzothiophene desulfurization by Rhodococcus sp. strain IGTS8 (ATCC 53968). Microbiology, 143, 2961–2973. https://doi.org/10.1099/00221287-143-9-2961.
Kaufman, E. R., Harkins, J. B., & Borole, A. P. (1998). Comparison of batchstirred and electrospray reactors for biodesulfurization of dibenzothiophene in crude oil and hydrocarbon feedstock. Applied Biochemistry and Biotechnology, 73, 127–144. https://doi.org/10.1007/BF02785650.
del Olmo, C. H., Alcon, A., Santos, V. E., & Garcia-ochoa, F. (2005). Modeling the production of a Rhodococcus erythropolis IGTS8 biocatalyst for DBT biodesulfurization: influence of media composition. Enzyme and Microbial Technology, 37, 157–166. https://doi.org/10.1016/j.enzmictec.2004.06.016.
Caro, A., Leton, P., Garcia-calvo, E., & Setti, L. (2007). Enhancement of dibenzothiophene biodesulfurization using β-cyclodextrins in oil-to-water media. Fuel, 86, 2632–2636. https://doi.org/10.1016/j.fuel.2007.02.033.
Wang, P., Humphrey, A. E., & Krawiec, S. (1996). Kinetic analyses of desulfurization of dibenzotiophene by Rhodococcus erythropolis in continuous cultures. Applied and Environmental Microbiology, 62, 3066–3068.
Ohshiro, T., Hirata, T., & Izumi, Y. (1996). Desulfurization of dibenzothiophene derivatives by whole cells of Rhodococcus erythropolis H-2. FEMS Microbiology Letters, 142, 65–70. https://doi.org/10.1111/j.1574-6968.1996.tb08409.x.
Yan, H., Kishimoto, M., Omasa, T., Katakura, Y., Suga, K. I., Okumura, K., & Yoshikawa, O. (2000). Increase in desulfurization activity of Rhodococcus erythropolis KA2-5-1 using ethanol feeding. Journal of Bioscience and Bioengineering, 89, 361–366. https://doi.org/10.1016/S1389-1723(00)88959-8.
Guerinik, K., & Al-Mutawah, Q. (2003). Isolation and characterization of oil-desulfurizing bacteria. World Journal of Microbiology and Biotechnology, 19, 941–945. https://doi.org/10.1023/B:WIBI.0000007327.72618.0f.
Yang, J., & Marison, I. W. (2005). Two-stage process design for the biodesulphurisation of a model diesel by a newly isolated Rhodococcus globerulus DAQ3. Biochemical Engineering Journal, 27, 77–82. https://doi.org/10.1016/j.bej.2005.08.012.
Yang, J., Hu, Y., Zhao, D., Wang, S., Lau, P. C. K., & Marison, I. W. (2007). Two-layer continuous-process design for the biodesulfurization of diesel oils under bacterial growth conditions. Biochemical Engineering Journal, 37, 212–218. https://doi.org/10.1016/j.bej.2007.04.012.
Li, G. Q., Ma, T., Li, S. S., Li, H., Liang, F. L., & Liu, R. L. (2007). Improvement of dibenzothiophene desulfurization activity by removing the gene overlap in the dsz operon. Bioscience, Biotechnology, and Biochemistry, 71, 849–854.
Acknowledgements
Some experiments were performed in the Biotechnology Laboratory of the Faculty of Veterinary Medicine, and in the Genetic Engineering Laboratory of the Research Institute of Biotechnology, Ferdowsi University of Mashhad.
Funding
This work was supported by the Ferdowsi University of Mashhad, Graduate Students Research Fund, Grant No. 3/22025. The sponsor had no involvement in the study design; collection, analysis, and interpretation of the data; writing the report; and decision for submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no personal or financial conflict of interest. All authors approved the final version of the manuscript. SK conducted the experiments, MAM designed and supervised the study, RG contributed the experimental setup, and HD supervised some experiments and contributed to the interpretation of results.
Additional information
A correction to this article is available online at https://doi.org/10.1007/s12010-018-2703-9.
Rights and permissions
About this article
Cite this article
Khosravinia, S., Mahdavi, M.A., Gheshlaghi, R. et al. Characterization of Truncated dsz Operon Responsible for Dibenzothiophene Biodesulfurization in Rhodococcus sp. FUM94. Appl Biochem Biotechnol 184, 885–896 (2018). https://doi.org/10.1007/s12010-017-2596-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-017-2596-z