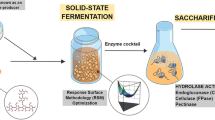
Abstract
Giant reed, miscanthus, and switchgrass are considered prominent lignocellulosic feedstocks to obtain fermentable sugars for biofuel production. The bioconversion into sugars requires a delignifying pre-treatment step followed by hydrolysis with cellulase and other accessory enzymes like xylanase, especially in the case of alkali pre-treatments, which retain the hemicellulose fraction. Blends richer in accessory enzymes than commercial mix can be obtained growing fungi on feedstock-based substrates, thus ten selected Trichoderma isolates, including the hypercellulolytic strain Trichoderma reesei Rut-C30, were grown on giant reed, miscanthus, or switchgrass-based substrates. The produced enzymes were used to saccharify the corresponding feedstocks, compared to a commercial enzymatic mix (6 FPU/g). Feedstocks were acid (H2SO4 0.2–2%, w/v) or alkali (NaOH 0.02–0.2%, w/v) pre-treated. A microplate-based approach was chosen for most of the experimental steps due to the large number of samples. The highest bioconversion was generally obtained with Trichoderma harzianum Or4/99 enzymes (78, 89, and 94% final sugar yields at 48 h for giant reed, miscanthus, and switchgrass, respectively), with significant increases compared to the commercial mix, especially with alkaline pre-treatments. The differences in bioconversion yields were only partially caused by xylanases (maximum R 2 = 0.5), indicating a role for other accessory enzymes.




Similar content being viewed by others
References
Scordia, D., Testa, G., & Cosentino, S. L. (2014). Perennial grasses as lignocellulosic feedstock for second-generation bioethanol production in Mediterranean environment. Italian Journal of Agronomy, 9(581), 84–93.
Fike, J. H., Parrish, D. J., & Fike, W. B. (2013). In B. P. Singh (Ed.), Biofuel crop sustainability: sustainable cellulosic grass crop production (pp. 109–164). Oxford: John Wiley & Sons.
Lewandowski, I., Scurlock, J. M. O., Lindvall, E., & Christou, M. (2003). The development and current status of perennial rhizomatous grasses as energy crops in the US and Europe. Biomass and Bioenergy, 25, 335–361.
Rossa, B., Tuffers, A. V., Naidoo, G., & von Willert, D. J. (1998). Arundo donax L. (Poaceae)—a C3 species with unusually high photosynthetic capacity. Botanica Acta, 111, 216–221.
Corno, L., Pilu, R., & Adani, F. (2014). Arundo donax L.: a non-food crop for bioenergy and bio-compound production. Biotechnology Advances, 32(8), 1535–1549.
Van Dik, J. S., & Pletschke, B. I. (2012). A review of lignocellulose bioconversion using enzymatic hydrolysis and synergistic cooperation between enzymes—factors affecting enzymes, conversion and synergy. Biotechnology Advances, 30, 1458–1480.
Cianchetta, S., Di Maggio, B., Burzi, P. L., & Galletti, S. (2014). Evaluation of selected white-rot fungal isolates for improving the sugar yield from wheat straw. Applied Biochemistry and Biotechnology, 173(2), 609–623.
Mosier, N., Wyman, C., Dale, B., Elander, R., Lee, Y. Y., Holtzapple, M., & Ladisch, M. (2005). Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresource Technology, 96(6), 673–686.
Singh, J., Suhag, M., & Dhaka, A. (2015). Augmented digestion of lignocellulose by steam explosion, acid and alkaline pretreatment methods: a review. Carbohydrate Polymers, 117, 624–631.
Sun, Y., & Cheng, J. J. (2002). Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresource Technology, 83, 1–11.
Chang, V. S., & Holtzapple, M. T. (2000). Fundamental factors affecting biomass enzymatic reactivity. Applied Biochemistry and Biotechnology, 84, 5–37.
Kootstra, A. M. J., Beeftink, H. H., Scott, E. L., & Sanders, J. P. M. (2009). Comparison of dilute mineral and organic acid pretreatment for enzymatic hydrolysis of wheat straw. Biochemical Engineering Journal, 46, 126–131.
McIntosh, S., & Vancov, T. (2011). Optimisation of dilute alkaline pretreatment for enzymatic saccharification of wheat straw. Biomass and Bioenergy, 35(7), 309–403.
Hu, J., Arantes, V., & Saddler, J. N. (2011). The enhancement of enzymatic hydrolysis of lignocellulosic substrates by the addition of accessory enzymes such as xylanase: is it an additive or synergistic effect? Biotechnology for Biofuels, 4(1), 1–14.
Li, J., Zhou, P., Liu, H., Xiong, C., Lin, J., Xiao, W., Gong, Y., & Liu, Z. (2014). Synergism of cellulase, xylanase, and pectinase on hydrolyzing sugarcane bagasse resulting from different pretreatment technologies. Bioresource Technology, 155, 258–265.
Berlin, A., Maximenko, V., Gilkes, N., & Saddler, J. (2007). Optimization of enzyme complexes for lignocellulose hydrolysis. Biotechnology and Bioengineering, 97(2), 287–296.
Alvira, P., Negro, M. J., & Ballesteros, M. (2011). Effect of endoxylanase and α-L-arabinofuranosidase supplementation on the enzymatic hydrolysis of steam exploded wheat straw. Bioresource Technology, 102(6), 4552–4558.
Banerjee, G., Car, S., Scott-Craig, J. S., Borrusch, M. S., & Walton, J. D. (2010a). Rapid optimization of enzyme mixtures for deconstruction of diverse pretreatment/biomass feedstock combinations. Biotechnology for Biofuels, 3, 22.
Banerjee, G., Scott-Craig, J. S., & Walton, J. D. (2010b). Improving enzymes for biomass conversion: a basic research perspective. Bioenergy Research, 3, 82–92.
Juhász, T., Szengyel, Z., Réczey, K., Siika-Aho, M., & Viikari, L. (2005). Characterization of cellulases and hemicellulases produced by Trichoderma reesei on various carbon sources. Process Biochemistry, 40, 3519–3325.
Cianchetta, S., Galletti, S., Burzi, P. L., & Cerato, C. (2012). Hydrolytic potential of Trichoderma sp. strains evaluated by microplate-based screening followed by switchgrass saccharification. Enzyme and Microbial Technology, 50, 304–310.
Jeon, Y. J., Xun, Z., & Rogers, P. L. (2010). Comparative evaluations of cellulosic raw materials for second generation bioethanol production. Letters of Applied Microbiology, 51, 518–524.
Kim, Y., Mosier, N. S., Ladisch, M. R., Pallapolu, V. R., Lee, Y. Y., Garlock, R., Balan, V., Dale, B. E., Donohoe, B. S., Vinzant, T. B., Elander, R. T., Falls, M., Sierra, R., Holtzapple, M. T., Shi, J., Ebrik, M. A., Redmond, T., Yang, B., Wyman, C. E., & Warner, R. E. (2011). Comparative study on enzymatic digestibility of switchgrass varieties and harvests processed by leading pretreatment technologies. Bioresource Technology, 102(24), 11089–11096.
Lygin, A. V., Uptonw, J., Dohlemanz, F. G., Juvik, J., Zabotina, O. A., Widholm, J. M., & Lozovaya, V. V. (2011). Composition of cell wall phenolics and polysaccharides of the potential bioenergy crop—Miscanthus. GCB Bioenergy, 3, 333–345.
Ceotto, E., Castelli, F., Moschella, A., Diozzi, M., & Di Candilo, M. (2015). Cattle slurry fertilization to giant reed (Arundo donax L.): biomass yield and nitrogen use efficiency. Bioenergy Research, 8(3), 1252–1262.
Di Candilo, M., Ceotto, E., & Diozzi, M. (2008). Comparison of 7 ligno-cellulosic biomass feedstock species: 6-years results in the Low Po Valley. Italian Journal of Agronomy, 3(Suppl 3), 481–482.
Monti, A., Bezzi, G., Pritoni, G., & Venturi, G. (2008). Long-term productivity of lowland and upland switchgrass cytotypes as affected by cutting frequency. Bioresource Technology, 99(16), 7425–7432.
Van Soest, P. J., Robertson, J. B., & Lewis, B. A. (1991). Methods for dietary fiber, neutral-detergent fiber and non-starch polysaccharides in relation to animal nutrition. Journal of Dairy Science, 74, 3583–3597.
Hindrichsen, I. K., Wettstein, H. R., Machmüller, A., Knudsen, K. B., Madsen, J., & Kreuzer, M. (2006). Digestive and metabolic utilisation of dairy cows supplemented with concentrates characterised by different carbohydrates. Animal Feed Science and Technology, 126(1), 43–61.
Cianchetta, S., Galletti, S., Burzi, P. L., & Cerato, C. (2010). A novel microplate-based screening strategy to assess the cellulolytic potential of Trichoderma strains. Biotechnology and Bioengineering, 107, 461–468.
Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31, 426–428.
Kubicek, C. P., & Penttilä, M. E. (1998). In G. E. Harman & C. P. Kubicek (Eds.), Trichoderma and Gliocladium, enzymes, biological control and commercial applications, vol. 2: regulation of production of plant polysaccharide degrading enzyme by Trichoderma (pp. 49–67). Bristol: Taylor and Francis Ltd..
Zhang, L., Liu, Y., Niu, X., Liu, Y., & Liao, W. (2012). Effects of acid and alkali treated lignocellulosic materials on cellulase/xylanase production by Trichoderma reesei Rut C-30 and corresponding enzymatic hydrolysis. Biomass and Bioenergy, 37, 16–24.
Dien, B. S., Jung, H. J. G., Vogel, K. P., Casler, M. D., Lamb, J. F., Iten, L., Mitchell, R. B., & Sarath, G. (2006). Chemical composition and response to dilute-acid pretreatment and enzymatic saccharification of alfalfa, reed canarygrass, and switchgrass. Biomass and Bioenergy, 30(10), 880–891.
Gao, K., Boiano, S., Marzocchella, A., & Rehmann, L. (2014). Cellulosic butanol production from alkali-pretreated switchgrass (Panicum virgatum) and phragmites (Phragmites australis). Bioresource Technology, 174, 176–181.
Xu, N., Zhang, W., Ren, S., Liu, F., Zhao, C., Liao, H., Xu, Z., Huang, J., Li, Q., Tu, Y., Yu, B., Wang, Y., Jiang, J., Qin, J., & Peng, L. (2012). Hemicelluloses negatively affect lignocellulose crystallinity for high biomass digestibility under NaOH and H2SO4 pretreatments in Miscanthus. Biotechnology for Biofuels, 5(1), 58.
Bowman, M. J., Dien, B. S., Vermillion, K. E., & Mertens, J. A. (2015). Isolation and characterization of unhydrolyzed oligosaccharides from switchgrass (Panicum virgatum L.) xylan after exhaustive enzymatic treatment with commercial enzyme preparations. Carbohydrate Research, 407, 42–50.
Zhang, J., Siika-aho, M., Tenkanen, M., & Viikari, L. (2011). The role of acetyl xylan esterase in the solubilization of xylan and enzymatic hydrolysis of wheat straw and giant reed. Biotechnology for Biofuels, 4(1), 1–10.
Hu, J., Chandra, R., Arantes, V., Gourlay, K., van Dyk, J. S., & Saddler, J. N. (2015). The addition of accessory enzymes enhances the hydrolytic performance of cellulase enzymes at high solid loading. Bioresource Technology, 186, 149–153.
Polizeli, M. L. T. M., Rizzatti, A. C. S., Monti, R., Terenzi, H. F., Jorge, J. A., & Amorim, D. S. (2005). Xylanases from fungi: properties and industrial applications. Applied Microbiology and Biotechnology, 67(5), 577–591.
Qing, Q., Yang, B., & Wyman, C. E. (2010). Xylooligomers are strong inhibitors of cellulose hydrolysis by enzymes. Bioresource Technology, 101, 9624–9630.
Nlewem, K. C., & Thrash, M. E. (2010). Comparison of different pretreatment methods based on residual lignin effect on the enzymatic hydrolysis of switchgrass. Bioresource Technology, 101(14), 5426–5430.
Jiang, D., Ge, X., Zhang, Q., & Li, Y. (2016). Comparison of liquid hot water and alkaline pretreatments of giant reed for improved enzymatic digestibility and biogas energy production. Bioresource Technology, 216, 60–68.
Komolwanich, T., Tatijarern, P., Prasertwasu, S., Khumsupan, D., Chaisuwan, T., Luengnaruemitchai, A., & Wongkasemjit, S. (2014). Comparative potentiality of Kans grass (Saccharum spontaneum) and Giant reed (Arundo donax) as lignocellulosic feedstocks for the release of monomeric sugars by microwave/chemical pretreatment. Cellulose, 21(3), 1327–1340.
Le Ngoc Huyen, T., Rémond, C., Dheilly, R. M., & Chabbert, B. (2010). Effect of harvesting date on the composition and saccharification of Miscanthus × giganteus. Bioresource Technology, 101(21), 8224–8231.
Scordia, D., Cosentino, S. L., Lee, J. W., & Jeffries, T. W. (2011). Dilute oxalic acid pretreatment for biorefining giant reed (Arundo donax L.) Biomass and Bioenergy, 35(7), 3018–3024.
Anderson, W. F., Dien, B. S., Brandon, S. K., & Peterson, J. D. (2008). Assessment of bermudagrass and bunch grasses as feedstock for conversion to ethanol. Applied Biochemistry and Biotechnology, 145, 13–21.
Guragain, Y. N., Ganesh, K. M., Bansal, S., Sathish, R. S., Rao, N., & Vadlani, P. V. (2014). Low-lignin mutant biomass resources: effect of compositional changes on ethanol yield. Industrial Crops and Products, 61, 1–8.
Berlin, A., Gilkes, N., Kilburn, D., Bura, R., Markov, A., Skomarovsky, A., Okunev, O., Gusakov, A., Maximenko, V., Gregg, D., Sinitsyn, A., & Saddler, J. (2005). Evaluation of novel fungal cellulase preparations for ability to hydrolyze softwood substrates—evidence for the role of accessory enzymes. Enzyme and Microbial Technology, 37, 175–184.
Arantes, V., & Saddler, J. N. (2010). Access to cellulose limits the efficiency of enzymatic hydrolysis: the role of amorphogenesis. Biotechnology for Biofuels, 3, 4.
Kabel, M. A., Van der Maarel, M. J., Klip, G., Voragen, A. G., & Schols, H. A. (2006). Standard assays do not predict the efficiency of commercial cellulase preparations towards plant materials. Biotechnology and Bioengineering, 93(1), 56–63.
Acknowledgments
This research was carried out partially in the framework of the “BIOSEGEN Project” funded by the Italian Ministry of Agricultural, Food and Forestry Policies, D.M. 17532/7303/10, and partially in the framework of the “AGROENER” project funded by the same Ministry, D.M. 26329/ 2016. The authors gratefully acknowledge Prof. Andrea Monti (University of Bologna), Dr. Enrico Ceotto, and Dr. Mario Di Candilo (CREA) for providing samples of the feedstocks studied, Dr. Ciro Vasmara (CREA) for determining their fiber composition, and Dr. Pier Luigi Burzi (CREA) for the technical assistance in laboratory.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cianchetta, S., Bregoli, L. & Galletti, S. Microplate-Based Evaluation of the Sugar Yield from Giant Reed, Giant Miscanthus and Switchgrass after Mild Chemical Pre-Treatments and Hydrolysis with Tailored Trichoderma Enzymatic Blends. Appl Biochem Biotechnol 183, 876–892 (2017). https://doi.org/10.1007/s12010-017-2470-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-017-2470-z