Abstract
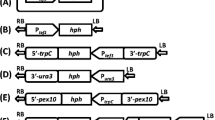
Oleaginous yeast Lipomyces starkeyi, a promising strain of great biotechnical importance, is able to accumulate over 60% of its cell biomass as triacylglycerols (TAGs). It is promising to directly produce the derivatives of TAGs, such as long-chain fatty acid methyl esters and alkanes, in L. starkeyi. However, techniques for genetic modification of this oleaginous yeast are lacking, thus, further research is needed to develop genetic tools and functional elements. Here, we used two exogenous promoters (pGPD and pPGK) from oleaginous yeast Rhodosporidium toruloides to establish a simpler Agrobacterium-mediated transformation (AMT) method for L. starkeyi. Hygromycin-resistant transformants were obtained on antibiotic-contained plate. Mitotic stability test, genotype verification by PCR, and protein expression confirmation all demonstrated the success of this method. Furthermore, the strength of these two promoters was evaluated at the phenotypic level on a hygromycin-gradient plate and at the transcriptional level by real-time quantitative PCR. The PGK promoter strength was 2.2-fold as that of GPD promoter to initiate the expression of the hygromycin-resistance gene. This study provided an easy and efficient genetic manipulation method and elements of the oleaginous yeast L. starkeyi for constructing superior strains to produce advanced biofuels.





Similar content being viewed by others
References
Peralta-Yahya, P. P., Zhang, F., del Cardayre, S. B., & Keasling, J. D. (2012). Microbial engineering for the production of advanced biofuels. Nature, 488, 320–328.
Dellomonaco, C., Clomburg, J. M., Miller, E. N., & Gonzalez, R. (2011). Engineered reversal of the beta-oxidation cycle for the synthesis of fuels and chemicals. Nature, 476, 355–359.
Zhou, Y. J., Buijs, N. A., Zhu, Z., Qin, J., Siewers, V., & Nielsen, J. (2016). Production of fatty acid-derived oleochemicals and biofuels by synthetic yeast cell factories. Nature Communications, 7, 11709.
Liu, L., Redden, H., & Alper, H. S. (2013). Frontiers of yeast metabolic engineering: diversifying beyond ethanol and Saccharomyces. Current Opinion in Biotechnology, 24, 1023–1030.
Alper, H., & Stephanopoulos, G. (2009). Engineering for biofuels: exploiting innate microbial capacity or importing biosynthetic potential? Nature Reviews. Microbiology, 7, 715–723.
Zhao, X., Kong, X. L., Hua, Y. Y., Feng, B., & Zhao, Z. B. (2008). Medium optimization for lipid production through co-fermentation of glucose and xylose by the oleaginous yeast Lipomyces starkeyi. European Journal of Lipid Science and Technology, 110, 405–412.
Angerbauer, C., Siebenhofer, M., Mittelbach, M., & Guebitz, G. M. (2008). Conversion of sewage sludge into lipids by Lipomyces starkeyi for biodiesel production. Bioresource Technology, 99, 3051–3056.
Gong, Z., Wang, Q., Shen, H., Hu, C., Jin, G., & Zhao, Z. K. (2012). Co-fermentation of cellobiose and xylose by Lipomyces starkeyi for lipid production. Bioresource Technology, 117, 20–24.
Liu, J. X., Yue, Q. Y., Gao, B. Y., Ma, Z. H., & Zhang, P. D. (2012). Microbial treatment of the monosodium glutamate wastewater by Lipomyces starkeyi to produce microbial lipid. Bioresource Technology, 106, 69–73.
Nishimura, K., Yamamoto, M., Nakagomi, T., Takiguchi, Y., Naganuma, T., & Uzuka, Y. (2002). Biodegradation of triazine herbicides on polyvinylalcohol gel plates by the soil yeast Lipomyces starkeyi. Applied Microbiology and Biotechnology, 58, 848–852.
Wu, S., Hua, Y. Y., Zhong, C. B., & Zhao, Z. K. (2008). The effect of tryptophol on lipid fermentation by Lipomyces starkeyi. Microbiology, 35, 200–203.
Riley, R., Haridas, S., Wolfe, K. H., et al. (2016). Comparative genomics of biotechnologically important yeasts. Proceedings of the National Academy of Sciences of the United States of America, 113, 9882–9887.
Liu, H. W., Zhao, X., Wang, F. J., et al. (2011). The proteome analysis of oleaginous yeast Lipomyces starkeyi. FEMS Yeast Research, 11, 42–51.
Calvey, C. H., Willis, L. B., & Jeffries, T. W. (2014). An optimized transformation protocol for Lipomyces starkeyi. Current Genetics, 60, 223–230.
Oguro, Y., Yamazaki, H., Shida, Y., Ogasawara, W., Takagi, M., & Takaku, H. (2015). Multicopy integration and expression of heterologous genes in the oleaginous yeast, Lipomyces starkeyi. Bioscience, Biotechnology, and Biochemistry, 79, 512–515.
Michielse, C. B., Hooykaas, P. J. J., van den Hondel, C. A. M. J. J., & Ram, A. F. J. (2005). Agrobacterium-mediated transformation as a tool for functional genomics in fungi. Current Genetics, 48, 1–17.
Lazo, G. R., Stein, P. A., & Ludwig, R. A. (1991). A DNA transformation-competent Arabidopsis genomic library in Agrobacterium. Bio-Technology, 9, 963–967.
Lin, X., Wang, Y., Zhang, S., et al. (2014). Functional integration of multiple genes into the genome of the oleaginous yeast Rhodosporidium toruloides. FEMS Yeast Research, 14, 547–555.
Bundock, P., den Dulk-Ras, A., Beijersbergen, A., & Hooykaas, P. J. (1995). Trans-kingdom T-DNA transfer from Agrobacterium tumefaciens to Saccharomyces cerevisiae. The EMBO Journal, 14, 3206–3214.
Wang, Y., Lin, X., Zhang, S., Sun, W., Ma, S., & Zhao, Z. K. (2016). Cloning and evaluation of different constitutive promoters in the oleaginous yeast Rhodosporidium toruloides. Yeast, 33, 99–106.
Sun, J., Shao, Z., Zhao, H., Nair, N., Wen, F., Xu, J. H., & Zhao, H. (2012). Cloning and characterization of a panel of constitutive promoters for applications in pathway engineering in Saccharomyces cerevisiae. Biotechnology and Bioengineering, 109, 2082–2092.
Partow, S., Siewers, V., Bjorn, S., Nielsen, J., & Maury, J. (2010). Characterization of different promoters for designing a new expression vector in Saccharomyces cerevisiae. Yeast, 27, 955–964.
de Groot, M. J., Bundock, P., Hooykaas, P. J., & Beijersbergen, A. G. (1998). Agrobacterium tumefaciens-mediated transformation of filamentous fungi. Nature Biotechnology, 16, 839–842.
Acknowledgments
We are indebted to professors Alexander Idnurm and Giuseppe Ianiri for their kind suggestions on AMT, to professors Xiaofeng Dai and Tianhong Wang for providing us Agrobacterium tumefaciens AGL1, to professor Lianghui Ji for providing us pTHR1.
This study was funded by the National Natural Science Foundation of China (3150100585), the China Postdoctoral Science Foundation (2016 M591419), and the Educational Commission of Liaoning Province of China (2016 018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Research Involving Human Participants and/or Animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Lin, X., Liu, S., Bao, R. et al. Development of an Agrobacterium-Mediated Transformation Method and Evaluation of Two Exogenous Constitutive Promoters in Oleaginous Yeast Lipomyces starkeyi . Appl Biochem Biotechnol 183, 867–875 (2017). https://doi.org/10.1007/s12010-017-2469-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-017-2469-5