Abstract
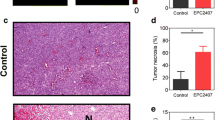
Angiogenesis plays a critical role in progression of malignant gliomas. The development of glioma-specific labeling molecules that can aid detection and visualization of angiogenesis can help surgical planning and improve treatment outcome. The aim of this study was to evaluate if two peptides (GX1 and RGD-GX1) linked to angiogenesis can be used as an MR-imaging markers of angiogenesis. MR imaging was performed in U87 glioblastoma-bearing NOD-SCID mice at different time points between 15 and 120 min post-injection to visualize particle distribution. GX1 and RGD-GX1 exhibited the highest accumulation in U87 glioblastoma at 120 min post i.v. administration. GX1-conjugated agents lead to higher decrease in transverse relaxation time (T 2) (i.e., stronger contrast enhancement) than RGD-GX1-conjugated agents in U87 glioblastoma tumor model. In addition, we tested if U87-IDH1R132 mutated cell line had different pattern of GX1 or RGD-GX1 particle accumulation. Responses in U87-IDH1WT followed a similar pattern with GX1 contrast agents; however, lower contrast enhancement was observed with RGD-GX1 agents. The specific binding of these peptides to human glioblastoma xenograft in the brain was confirmed by magnetic resonance imaging. The contrast enhancement following injection of magnetonanoparticles conjugated to GX1 peptide matched well with CD31 staining and iron staining.






Similar content being viewed by others
References
Ostrom, Q. T., Gittleman, H., Liao, P., Rouse, C., Chen, Y., Dowling, J., Wolinsky, Y., Kruchko, C., & Barnholtz-Sloan, J. (2014). CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro-Oncology, 16(Suppl 4), iv1–i63.
Bello, L., Giussani, C., Carrabba, G., Pluderi, M., Costa, F., & Bikfalvi, A. (2004). Angiogenesis and invasion in gliomas. Cancer Treatment and Research, 117, 263–284.
Skobe, M., Rockwel, L. P., Goldstein, N., Vosseler, S., & Fusenig, N. E. (1997). Halting angiogenesis suppresses carcinoma cell invasion. Nature Med., 3, 1222–1227.
Parsons, D. W., Jones, S., Zhang, X., Lin, J. C., Leary, R. J., Angenendt, P., Mankoo, P., Carter, H., Siu, I. M., Gallia, G. L., Olivi, A., McLendon, R., Rasheed, B. A., Keir, S., Nikolskaya, T., Nikolsky, Y., Busam, D. A., Tekleab, H., Diaz Jr., L. A., Hartigan, J., Smith, D. R., Strausberg, R. L., Marie, S. K., Shinjo, S. M., Yan, H., Riggins, G. J., Bigner, D. D., Karchin, R., Papadopoulos, N., Parmigiani, G., Vogelstein, B., Velculescu, V. E., & Kinzler, K. W. (2008). An integrated genomic analysis of human glioblastoma multiforme. Science, 321(5897), 1807–1812.
Yan, H., Parsons, D. W., Jin, G., McLendon, R., Rasheed, B. A., Yuan, W., Kos, I., Batinic-Haberle, I., Jones, S., Riggins, G. J., Friedman, H., Friedman, A., Reardon, D., Herndon, J., Kinzler, K. W., Velculescu, V. E., Vogelstein, B., & Bigner, D. D. (2009). IDH1 and IDH2 mutations in gliomas. The New England Journal of Medicine, 360(8), 765–773.
Xu, X., Zhao, J., Xu, Z., Peng, B., Huang, Q., Arnold, E., & Ding, J. (2004). Structures of human cytosolic NADP-dependent isocitrate dehydrogenase reveal a novel self-regulatory mechanism of activity. The Journal of Biological Chemistry, 279(32), 33946–33957.
Nobusawa, S., Watanabe, T., Kleihues, P., & Ohgaki, H. (2009). IDH1 mutations as molecular signature and predictive factor of secondary glioblastomas. Clinical Cancer Research, 15(19), 6002–6007.
Smith, J. W., & Cheresh, D. A. (1988). The arg-gly-asp binding domain of the vitronectin receptor: photoaffinity cross-linking implicates amino acid residues 61–203 of the ß subunit. The Journal of Biological Chemistry, 263, 18726–18731.
Zhi, M., Wu, K. C., Dong, L., Hao, Z. M., Deng, T. Z., Hong, L., Liang, S. H., Zhao, P. T., Qiao, T. D., Wang, Y., Xu, X., & Fan, D. M. (2004). Characterization of a specific phage-displayed peptide binding to vasculature of human gastric cancer. Cancer Biology & Therapy, 3, 1232–1235.
Dijkgraaf, I., Kruijtzer, A. W., Liu, S., Soede, A. C., Oyen, W. J. G., Corstens, F. H. M., Liskamp, R. M. J., & Boerman, O. C. (2007). Improved targeting of the avb3 integrin by multimerisation of RGD peptides. European Journal of Nuclear Medicine and Molecular Imaging, 34(2), 267–273.
Liu, S., Kim, Y. S., Hsieh, W. Y., & Sreerama, S. G. (2008). Coligant effect on the solution stability, biodistribution and metabolism of the 99mTc-labeled cyclic RGDfK tetramer. Nuclear Medicine and Biology, 35, 111–121.
Moncelet, D., Bouchaud, V., Mellet, P., Miraux, S., Franconi, J. M., & Voisin, P. (2013). Cellular density effect on RGD ligand internalization in glioblastoma for MRI application. PloS One, 8(12), e82777.
Schottelius, M., & Wester, H. J. (2009). Molecular imaging targeted receptor. Methods, 48, 161–177.
Zhang, X., Xiong, Z., Wu, Y., Cai, W., Tseng, J. R., Gambhir, S. S., & Chen, X. (2006). Quantitative PET imaging of tumor integrin alphavbeta3 expression with 18F-FRGD2. Journal of Nuclear Medicine, 47(1), 113–121.
Desgrosellier, J. S., & Cheresh, D. A. (2010). Integrins in cancer: biological implications and therapeutic opportunities. Nature Reviews. Cancer, 10, 9–22.
Guo, W., & Giancotti, F. G. (2004). Integrin signalling during tumour progression. Nature Reviews. Molecular Cell Biology, 5, 816–826.
Hynes, R. O. (1992). Integrins: versatility, modulation, and signaling in cell adhesión. Cell, 69(1), 11–25.
Jin, Z. H., Furukawa, T., Waki, A., Akaji, K., Coll, J. L., Saga, T., & Fujibayashi, Y. (2010). Effect of multimerization of a linear arg-gly-asp peptide on integrin binding affinity and specificity. Biological & Pharmaceutical Bulletin, 33(3), 370–378.
Wu, H., Chen, H., Pan, D., Ma, Y., Liang, S., Wan, Y., & Fang, Y. (2014). Imaging integrin αvβ 3 and NRP-1 positive gliomas with a novel fluorine-18 labeled RGD-ATWLPPR heterodimeric peptide probe. Molecular Imaging and Biology, 16(6), 781–792.
Chen, B., Cao, S., Zhang, Y., Wang, X., Liu, J., Hui, X., Wan, Y., Du, W., Wang, L., Wu, K., & Fan, D. (2009). A novel peptide (GX1) homing to gastric cancer vasculature inhibits angiogenesis and cooperates with TNF alpha in anti-tumor therapy. BMC Cell Biology, 10, 63.
Chen, K., Sun, X., Niu, G., Ma, Y., Yap, L., Hui, X., Wu, K., Fan, D., Conti, P. S., & Chen, X. (2012). Evaluation of 64Cu labeled GX1: a phage display peptide probe for PET imaging of tumor vasculature. Molecular Imaging and Biology, 14(1), 96–105.
Hu, H., Yin, J., Wang, M., Liang, C., Song, H., Wang, J., Nie, Y., & Liang, J. (2014). GX1 targeting delivery of rmhTNFα evaluated using multimodality imaging. International Journal of Pharmaceutics, 461(1–2), 181–191.
Hui, X., Han, Y., Liang, S., Liu, Z., Liu, J., Hong, L., Zhao, L., He, L., Cao, S., Chen, B., Yan, K., Jin, B., Chai, N., Wang, J., Wu, K., & Fan, D. (2008). Specific targeting of the vasculature of gastric cancer by a new tumor-homing peptide CGNSNPKSC. Journal of Controlled Release, 131, 86–93.
Xin, J., Zhang, X., Liang, J., Xia, L., Yin, J., Nie, Y., Wu, K., & Tian, J. (2013). In vivo gastric cancer targeting and imaging using novel symmetric cyanine dye-conjugated GX1 peptide probes. Bioconjugate Chemistry, 24(7), 1134–1143.
Li, Z. B., Wu, Z., Chen, K., Ryu, E. K., & Chen, X. (2008). 18F-labeled BBN-RGD heterodimer for prostate cancer imaging. Journal of Nuclear Medicine, 49, 453–461.
Liu, Z., Yan, Y., Chin, F. T., Wang, F., & Chen, X. (2009). Dual integrin and gastrin-releasing peptide receptor targeted tumor imaging using 18F-labeled PEGylated RGD-bombesin heterodimer 18F-FB-PEG3-Glu-RGD-BBN. Journal of Medicinal Chemistry, 52(2), 425–432.
Yang, J., Guo, H., Galazzi, F., Berwick, M., Padilla, R. S., & Miao, Y. (2009). Evaluation of a novel arg-gly-asp-conjugated r-melanocyte stimulating hormone hybrid peptide for potential melanoma therapy. Bioconjugate Chemistry, 20, 1634–1642.
Yang, J., Guo, H., & Miao, Y. (2010). Technetium-99m-labeled arg-gly-asp-conjugated alpha-melanocyte stimulating hormone hybrid peptides for human melanoma imaging. Nuclear Medicine and Biology, 37(8), 873–883.
Liu, Z., & Wang, F. (2010). Dual-targeted molecular probes for cancer imaging. Current Pharmaceutical Biotechnology, 11, 610–619.
Oliveira, E. A., & Faintuch, B. L. (2015). Radiolabeling and biological evaluation of the GX1 and RGD-GX1 peptide sequence for angiogenesis targeting. Nuclear Medicine and Biology, 42(2), 123–130.
Oliveira, E. A., Faintuch, B. L., Targino, R. C., Moro, A. M., Martinez, R. C., Pagano, R. L., Fonoff, E. T., Carneiro, C. G., Garcez, A. T., Faria, D. P., & Buchpiguel, C. A. (2016). Evaluation of GX1 and RGD-GX1 peptides as new radiotracers for angiogenesis evaluation in experimental glioma models. Amino Acids, 48(3), 821–831.
Lazovic, J., Soto, H., Piccioni, D., Lou, J. R., Li, S., Mirsadraei, L., Yong, W., Prins, R., Liau, L. M., Ellingson, B. M., Cloughesy, T. F., Lai, A., & Pope, W. B. (2012). Detection of 2-hydroxyglutaric acid in vivo by proton magnetic resonance spectroscopy in U87 glioma cells overexpressing isocitrate dehydrogenase-1 mutation. Neuro-Oncology, 14(12), 1465–1472.
Akhtari, M., Bragin, A., Moats, R., Frew, A., & Mandelkern, M. (2012). Functionalized magnetonanoparticles in imaging brain neuronal activity using MRI. Brain Topography, 25(4), 374–388.
Akhtari, M., Bragin, A., Cohen, M., Moats, R., Brenker, F., Lynch, M. D., Vinters, H. V., & Engel Jr., J. (2008). Functionalized magnetonanoparticles for MRI diagnosis and localization in epilepsy. Epilepsia, 49, 1419–1430.
Akhtari, M., Pope, W., Mathern, G., Moats, R., Frew, A., & Mandelkern, M. (2013). Functionalized magnetonanoparticles in visualization of intracranial tumors on MRI. Molecular Imaging and Biology, 15(3), 299–306.
Akhtari, M., & Engel, J. (2006). Patents PCT/US06/10334. EU06739214.2.
Akhtari, M., & Engel, J. (2009). Pat # WO 2009/123734 A1.
Goldenberg, D. M., Sharkey, R. M., Paganelli, G., Barbet, J., & Chatal, J. F. (2006). Antibody pretargeting advances cancer radioimmunodetection and radioimmunotherapy. Journal of Clinical Oncology, 24, 823–834.
Orlova, A., Magnusson, M., Eriksson, T. L., Nilsson, M., Larsson, B., Höidén-Guthenberg, I., Widström, C., Carlsson, J., Tolmachev, V., Ståhl, S., & Nilsson, F. Y. (2006). Tumor imaging using a picomolar affinity HER2 binding affibody molecule. Cancer Research, 66, 4339–4348.
Decristoforo, C., Faintuch-Linkowski, B., Rey, A., von Guggenberg, E., Rupprich, M., Hernandez-Gonzales, I., Rodrigo, T., & Haubner, R. (2006). [99mTc]HYNIC-RGD for imaging integrin alphavbeta3 expression. Nuclear Medicine and Biology, 33, 945–952.
Decristoforo, C., Santos, I., Pietzsch, H. J., Kuenstler, J. U., Duatti, A., Smith, C. J., Rey, A., Alberto, R., Von Guggenberg, E., & Haubner, R. (2007). Comparison of in vitro and in vivo properties of [99mTc]cRGD peptides labeled using different novel Tc-cores. The Quarterly Journal Nuclear Medicine and Molecular Imaging, 51, 33–41.
Janssen, M. L., Oyen, W. J., Dijkgraaf, I., Massuger, L. F., Frielink, C., Edwards, D. S., Rajopadhye, M., Boonstra, H., Corstens, F. H., & Boerman, O. C. (2002). Tumor targeting with radiolabeled alpha(v)beta(3) integrin binding peptides in a nude mouse model. Cancer Research, 62, 6146–6151.
Su, Z. F., He, J., Rusckowski, M., & Hnatowich, D. J. (2003). In vitro cell studies of technetium-99m labeled RGD-HYNIC peptide, a comparison of tricine and EDDA as co-ligands. Nuclear Medicine and Biology, 30, 141–149.
Denardo, S. J., Yao, Z., Lam, K. M., Song, A., Burker, P. A., Merick, G. R., Lamborn, K. R., O’Donnell, R. T., & Denardo, G. L. (2003). Effect of molecular size of pegylated peptide on the pharmacokinetics and tumor targeting in lymphoma-bearing mice. Clinical Cancer Research, 1(9), 38545–38645.
Vonarbourg, A., Passirani, C., Saulnier, P., & Benoit, J. P. (2006). Parameters influencing the stealthiness of colloidal drug delivery systems. Biomaterials, 27(24), 4356–4373.
Däpp, S., García Garayoa, E., Maes, V., Brans, L., Tourwé, D. A., Müller, C., & Schibli, R. (2011). PEGylation of (99m)Tc-labeled bombesin analogues improves their pharmacokinetic properties. Nuclear Medicine and Biology, 38(7), 997–1009.
Mier, W., Krämer, S., Zitzmann, S., Altmann, A., Leotta, K., Schierbaum, U., Schnölzer, M., Eisenhut, M., & Haberkorn, U. (2013). PEGylation enables the specific tumor accumulation of a peptide identified by phage display. Organic & Biomolecular Chemistry, 11(16), 2706–2711.
Hampton, M. B., & Orrenius, S. (1997). Dual regulation of caspase activity by hydrogen peroxide: implications for apoptosis. FEBS Letters, 414(3), 552–556.
Li, S., Chou, A. P., Chen, W., Chen, R., Deng, Y., Phillips, H. S., Selfridge, J., Zurayk, M., Lou, J. J., Everson, R. G., Wu, K. C., Faull, K. F., Cloughesy, T., Liau, L. M., & Lai, A. (2013). Overexpression of isocitrate dehydrogenase mutant proteins renders glioma cells more sensitive to radiation. Neuro-Oncology, 15(1), 57–68.
Ramsey, M. R., & Sharpless, N. E. (2006). ROS as a tumour suppressor? Nature Cell Biology, 8(11), 1213–1215.
Takahashi, A., Ohtani, N., Yamakoshi, K., Iida, S., Tahara, H., Nakayama, K., Nakayama, K. I., Ide, T., Saya, H., & Hara, E. (2006). Mitogenic signalling and the p16INK4a-Rb pathway cooperate to enforce irreversible cellular senescence. Nature Cell Biology, 8(11), 1291–1297.
Fei, X. F., Zhang, Q. B., Dong, J., Diao, Y., Wang, Z. M., Li, R. J., Wu, Z. C., Wang, A. D., Lan, Q., Zhang, S. M., & Huang, Q. (2010). Development of clinically relevant orthotopic xenograft mouse model of metastatic lung cancer and glioblastoma through surgical tumor tissues injection with trocar. Journal of Experimental & Clinical Cancer Research, 29, 84.
Islam, T., & Harisinghani, M. G. (2009). Overview of nanoparticle use in cancer imaging. Cancer Biomarkers, 5, 61–67.
Molday, H. S. (1984). US Pat 2,452,773.
Muldoon, L. L., Sandor, M., Pinkston, K. E., & Neuwelt, E. A. (2005). Imaging, distribution, and toxicity of superparamagnetic iron oxide magnetic resonance nanoparticles in the rat brain and intracerebral tumor. Neurosurgery, 57, 785–796.
Pope, W. B., Prins, R. M., Albert Thomas, M., Nagarajan, R., Yen, K. E., Bittinger, M. A., Salamon, N., Chou, A. P., Yong, W. H., Soto, H., Wilson, N., Driggers, E., Jang, H. G., Su, S. M., Schenkein, D. P., Lai, A., Cloughesy, T. F., Kornblum, H. I., Wu, H., Fantin, V. R., & Liau, L. M. (2012). Non-invasive detection of 2-hydroxyglutarate and other metabolites in IDH1 mutant glioma patients using magnetic resonance spectroscopy. Journal of Neuro-Oncology, 107(1), 197–205.
Chakraborty, M., Jain, S., & Rani, V. (2011). Nanotechnology: emerging tool for diagnostics and therapeutics. Applied Biochemistry and Biotechnology, 165(5–6), 1178–1187. doi:10.1007/s12010-011-9336-6 Review.
Fernandez-Fernandez, A., Manchanda, R., & McGoron, A. J. (2011). Theranostic applications of nanomaterials in cancer: drug delivery, image-guided therapy, and multifunctional platforms. Applied Biochemistry and Biotechnology, 165(7–8), 1628–1651.
Zhou, X., Chen, L., Wang, A., Ma, Y., Zhang, H., & Zhu, Y. (2015). Multifunctional fluorescent magnetic nanoparticles for lung cancer stem cells research. Colloids and Surfaces. B, Biointerfaces, 20(134), 431–439.
Rosenberger, I., Strauss, A., Dobiasch, S., Weis, C., Szanyi, S., Gil-Iceta, L., Alonso, E., González Esparza, M., Gómez-Vallejo, V., Szczupak, B., Plaza-García, S., Mirzaei, S., Israel, L. L., Bianchessi, S., Scanziani, E., Lellouche, J. P., Knoll, P., Werner, J., Felix, K., Grenacher, L., Reese, T., Kreuter, J., & Jiménez-González, M. (2015). Targeted diagnostic magnetic nanoparticles for medical imaging of pancreatic cancer. Journal of Controlled Release, 17(214), 76–84.
Yan, X., Song, X., Wang, Z. (2016). Construction of specific magnetic resonance imaging/optical dual-modality molecular probe used for imaging angiogenesis of gastric cancer. Artif Cells Nanomed Biotechnol. 1–5.
Liu, C., Liu, D. B., Long, G. X., Wang, J. F., Mei, Q., Hu, G. Y., Qiu, H., & Hu, G. Q. (2013). Specific targeting of angiogenesis in lung cancer with RGD-conjugated ultrasmall superparamagnetic iron oxide particles using a 4.7 T magnetic resonance scanner. Chinese Medical Journal, 126(12), 2242–2247.
Zhang, C., Jugold, M., Woenne, E. C., Lammers, T., Morgenstern, B., Mueller, M. M., Zentgraf, H., Bock, M., Eisenhut, M., Semmler, W., & Kiessling, F. (2007). Specific targeting of tumor angiogenesis by RGD-conjugated ultrasmall superparamagnetic iron oxide particles using a clinical 1.5-T magnetic resonance scanner. Cancer Research, 67, 1555–1562.
Park, J. A., Lee, J. J., Jung, J. C., Yu, D. Y., Oh, C., Ha, S., Kim, T. J., & Chang, Y. (2008). Gd-DOTA conjugate of RGD as a potential tumor-targeting MRI contrast agent. Chembiochem, 9, 2811–2813.
Sun, J., Teng, Z. G., Tian, Y., Wang, J. D., Guo, Y., Kim, D. H., Larson, A. C., & Lu, G. M. (2014). Targeted fluorescent magnetic nanoparticles for imaging of human breast cancer. International Journal of Clinical and Experimental Medicine, 7(12), 4747–4758.
Yang, J., Luo, Y., Xu, Y., Li, J., Zhang, Z., Wang, H., Shen, M., Shi, X., & Zhang, G. (2015). Conjugation of iron oxide nanoparticles with RGD-modified dendrimers for targeted tumor MR imaging. ACS Applied Materials & Interfaces, 7(9), 5420–5428.
Acknowledgements
The authors are grateful for a post-graduate grant by Fundação de Amparo a Pesquisa do Estado de São Paulo, Brazil (FAPESP 2011/12405-0). We thank preclinical imaging and histology unit of Vienna Biocenter Core Facilities (VBCF) for their help with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Funding was provided by Fundação de Amparo a Pesquisa do Estado de São Paulo, Brazil (FAPESP 2011/12405-0).
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures were approved by the University of California, Los Angeles, Institutional Animal Care and Use Committee.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
de Oliveira, E.A., Lazovic, J., Guo, L. et al. Evaluation of Magnetonanoparticles Conjugated with New Angiogenesis Peptides in Intracranial Glioma Tumors by MRI. Appl Biochem Biotechnol 183, 265–279 (2017). https://doi.org/10.1007/s12010-017-2443-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-017-2443-2