Abstract
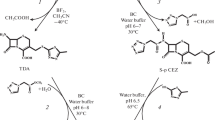
In this study, we investigated the enzymatic synthesis of a semi-synthetic cephalosporin, cefadroclor, from 7-aminodesacetoxymethyl-3-chlorocephalosporanic acid (7-ACCA) and p-OH-phenylglycine methyl ester (D-HPGM) using immobilized penicillin G acylase (IPA) in organic co-solvents. Ethylene glycol (EG) was employed as a component of the reaction mixture to improve the yield of cefadroclor. EG was found to increase the yield of cefadroclor by 15–45%. An investigation of altered reaction parameters including type and concentration of organic solvents, pH of reaction media, reaction temperature, molar ratio of substrates, enzyme loading, and IPA recycling was carried out in the buffer mixture. The best result was a 76.5% conversion of 7-ACCA, which was obtained from the reaction containing 20% EG (v/v), D-HPGM to 7-ACCA molar ratio of 4:1 and pH 6.2, catalyzed by 16 IU mL−1 IPA at 20 °C for 10 h. Under the optimum conditions, no significant loss of IPA activity was found after seven repeated reaction cycles. In addition, cefadroclor exhibited strong inhibitory activity against yeast, Bacillus subtilis NX-2, and Escherichia coli and weaker activity against Staphylococcus aureus and Pseudomonas aeruginosa. Cefadroclor is a potential antibiotic with activity against common pathogenic microorganisms.








Similar content being viewed by others
References
Srirangan, K., Orr, V., Akawi, L., Westbrook, A., Moo-Young, M., & Chou, C. P. (2013). Biotechnological advances on Penicillin G acylase: pharmaceutical implications, unique expression mechanism and production strategies. Biotechnology Advances, 31, 1319–1332.
Illanes, A., Wilson, L., Corrotea, O., Tavernini, L., Zamorano, F., & Aguirre, C. (2007). Synthesis of cephalexin with immobilized penicillin acylase at very high substrate concentrations in fully aqueous medium. Journal of Molecular Catalysis B: Enzymatic, 47, 72–78.
Bengtsson-Palme, J., & Joakim Larsson, D. G. (2016). Concentrations of antibiotics predicted to select for resistant bacteria: proposed limits for environmental regulation. Environment International, 86, 140–149.
Anacona, J. R., Rangel, V., Loroño, M., & Camus, J. (2015). Tetradentate metal complexes derived from cephalexin and 2, 6-diacetylpyridine bis(hydrazone): synthesis, characterization and antibacterial activity. Spectrochimica Acta Part A:Molecular and Biomolecular Spectroscopy, 149, 23–29.
Chen, C., Wu, Q., Liu, B., Lv, D., & Lin, X. (2008). Anhydrous tert-pentanol as a novel media for the efficient enzymatic synthesis of amoxicillin. Enzyme and Microbial Technology, 42, 601–607.
Bruggink, A., Roos, E. C., & de Vroom, E. (1998). Penicillin acylase in the industrial production of beta-lactam antibiotics. Organic Process Research and Development, 2, 128–133.
Justiz, O. H., Fernandez-Lafuente, R., Guisan, J. M., Negri, P., Pagani, G., Pregnolato, M., & Terreni, M. (1997). One-pot chemoenzymatic synthesis of 3′-functionalized cephalosporines (cefazolin) by three consecutive biotransformations in fully aqueous medium. Journal of Organic Chemistry, 62, 9099–9106.
Fernández-Lafuente, R., Rosell, C. M., Piatkowska, B., & Guisán, J. M. (1996). Synthesis of antibiotics (cephaloglycin) catalyzed by penicillin G acylase: evaluation and optimization of different synthetic approaches. Enzyme and Microbial Technology, 19, 9–14.
Illanes, A., Wilson, L., Caballero, E., Fernández-Lafuente, R., & Guisán, J. M. (2006). Crosslinked penicillin acylase aggregates for synthesis of β-lactam antibiotics in organic medium. Applied Biochemistry and Biotechnology, 133, 189–202.
Volpato, G., Rodrigues, R. C., & Fernandez-Lafuente, R. (2010). Use of enzymes in the production of semi-synthetic penicillins and cephalosporins: drawbacks and perspectives. Current Medicinal Chemistry, 17, 3855–3873.
Yang, L., & Wei, D. Z. (2003). Enhanced enzymatic synthesis of a semi-synthetic cephalosporin, cefaclor, with in situ product removal. Biotechnology Letters, 25, 1195–1198.
Wei, D., Zhu, J., & Cao, X. (2002). Enzymatic synthesis of cephalexin in aqueous two-phase systems. Biochemical Engineering Journal, 11, 95–99.
Terreni, M., Ubiali, D., Pagani, G., Hernández-Jústiz, O., Fernández-Lafuente, R., & Guisán, J. M. (2005). Penicillin G acylase catalyzed acylation of 7-ACA in aqueous two-phase systems using kinetically and thermodynamically controlled strategies: improved enzymatic synthesis of 7-[(1-hydroxy-1-phenyl)-acetamido]-3-acetoxymethyl-Δ3-cephem-4-carboxylic acid. Enzyme and Microbial Technology, 36, 672–679.
Wu, Q., Chen, C., Du, L., & Lin, X. (2010). Enzymatic synthesis of amoxicillin via a one-pot enzymatic hydrolysis and condensation cascade process in the presence of organic co-solvents. Applied Biochemistry and Biotechnology, 160, 2026–2035.
Deng, S., Ma, X., Sun, M., Wei, D., & Su, E. (2016). Efficient enzymatic synthesis of ampicillin using mutant penicillin G acylase with bio-based solvent glycerol. Catalysis Communications, 79, 31–34.
van Langen, L. M., van Rantwijk, F., Vedas, V. K., & Sheldon, R. A. (2000). Penicillin acylase-catalyzed peptide synthesis: a chemo-enzymatic route to stereoisomers of 3, 6-diphenylpiperazine-2,5-dione. Tetrahedron: Asymmetry, 11, 1077–1083.
Park, C. B., Lee, S. B., & Ryu, D. D. Y. (2000). Penicillin acylase-catalyzed synthesis of cefazolin in water–solvent mixtures: enhancement effect of ethyl acetate and carbon tetrachloride on the synthetic yield. Journal of Molecular Catalysis B: Enzymatic, 9, 275–281.
Deaguero, A. L., & Bommarius, A. S. (2014). In situ mixed donor synthesis of ampicillin with ethylene glycol co-solvent. Biotechnology and Bioengineering, 111, 1054–1058.
Kim, M. G., & Lee, S. B. (1996). Penicillin acylase-catalyzed synthesis of β-lactam antibiotics in water-methanol mixtures: effect of cosolvent content and chemical nature of substrate on reaction rates and yields. Journal of Molecular Catalysis B: Enzymatic, 1, 201–211.
Illanes, A., Anjarí, S., Arrieta, R., & Aguirre, C. (2002). Optimization of yield in kinetically controlled synthesis of ampicillin with immobilized penicillin acylase in organic media. Applied Biochemistry and Biotechnology, 97, 165–179.
Bahamondes, C., Wilson, L., Aguirre, C., & Illanes, A. (2012). Comparative study of the enzymatic synthesis of cephalexin at high substrate concentration in aqueous and organic media using statistical model. Biotechnology and Bioprocess Engineering, 17, 711–721.
Wang, L., Ye, L., Pan, Y., & Cao, Y. (2012). Two plate-based colorimetric assays for screening α-amino acid ester hydrolase with high synthesis/hydrolysis ratio. Enzyme and Microbial Technology, 51, 107–112.
Wegman, M. A., van Langen, L. M., van Rantwijk, F., & Sheldon, R. A. (2002). A two-step, one-pot enzymatic synthesis of cephalexin from D-phenylglycine nitrile. Biotechnology and Bioengineering, 79, 356–361.
Mark, D. C., & Arthur, J. R. (2015). Laccase-catalyzed synthesis of 2, 3-ethylenedithio-1,4-quinones. Journal of Molecular Catalysis B: Enzymatic, 119, 85–89.
Xu, H., Jiang, M., Li, H., Lu, D., & Ouyang, P. (2005). Efficient production of poly(γ-glutamic acid) by newly isolated Bacillus subtilis NX-2. Process Biochemistry, 40, 519–523.
Shewale, J. G., Kumar, K. K., & Ambekar, G. R. (1987). Evaluation of determination 6-aminopenicillanic acid by p-dimethyl aminobenzaldehyde. Biotechnology Techniques, 1, 69–72.
Andrews, J. M. (2001). Determination of minimum inhibitory concentrations. Journal of Antimicrobial Chemotherapy, 48, 5–16.
Munenori, T., Masayuki, S., Haruka, I., & Hideo, H. (2008). Poly(γ-L-diaminobutanoic acid), a novel poly(amino acid), coproduced with poly(ε-L-lysine) by two strains of Streptomyces celluloflavus. FEMS Microbiology Letters, 286, 110–117.
Kim, M. G., & Lee, S. B. (1996). Penicillin acylase-catalyzed synthesis of pivampicillin: effect of reaction variables and organic cosolvents. Journal of Molecular Catalysis B: Enzymatic, 1, 71–80.
Kim, M. G., & Lee, S. B. (1996). Effect of organic solvents on penicillin acylase-catalyzed reactions: interaction of organic solvents with enzymes. Journal of Molecular Catalysis B: Enzymatic, 1, 181–190.
Wei, D., & Yang, L. (2003). Effects of ethylene glycol on the synthesis of ampicillin using immobilized penicillin G acylase. Journal of Chemical Technology & Biotechnology, 78, 431–436.
van Langen, L. M., de Vroom, E., van Rantwijk, F., & Sheldon, R. (1999). Enzymatic synthesis of β-lactam antibiotics using penicillin-G acylase in frozen media. FEBS Letters, 456, 89–92.
Akhtar, J., Amin, N. S., & Junjie, W. (2012). Optimization studies for catalytic ozonation of cephalexin antibiotic in a batch reactor. Journal of Water Supply: Research and Technology-AQUA, 61, 413–414.
Illanes, A., Anjarı́, M. S., Altamirano, C., & Aguirre, C. (2004). Optimization of cephalexin synthesis with immobilized penicillin acylase in ethylene glycol medium at low temperatures. Journal of Molecular Catalysis B: Enzymatic, 30, 95–103.
Chaney, N., Wilson-Stanford, S., Kastrantas, J., Dahal, N., & Smith, L. (2009). Rapid method for extracting the antibiotic mutacin 1140 from complex fermentation medium yeast extract. Canadian Journal of Microbiology, 55, 1261–1266.
Acknowledgements
This work was supported by the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (11KJD180001), the Natural Science Foundation of Jiangsu Education Ministry (14KJB530005), and the Science and Technology Program of Joint Innovation Fund—A Prospective Joint Research Project in Jiangsu Province (No. BY2014005-10).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, K., Li, S., Pang, X. et al. Enhanced Enzymatic Synthesis of a Cephalosporin, Cefadroclor, in the Presence of Organic Co-solvents. Appl Biochem Biotechnol 182, 29–40 (2017). https://doi.org/10.1007/s12010-016-2308-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-016-2308-0