Abstract
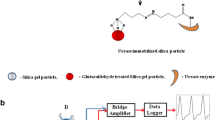
Abnormal concentrations of amino acids in blood and urine can be indicative of several diseases, including cancer and diabetes. Therefore, analyses that examine amino acid concentrations are useful for the diagnosis of such diseases. In this study, we developed an enzyme-immobilized, small reactor column for flow analysis of amino acid concentrations. For the recognition of asparagine and lysine, asparaginyl-tRNA synthetase and lysyl-tRNA synthase were immobilized onto microparticles, respectively, and coupled with coloration reagents for spectrophotometric detection. This assay has some advantages in the analytical field, such as the ability to detect small amounts of analyte, allowing for the use of a small reaction volume, and ensuring a rapid and efficient reaction rate. This approach provided selective quantitation of up to 480 μM of asparagine and lysine in 200 mM Tris–HCl buffer (pH 8.0).





Similar content being viewed by others
References
Miyagi, Y., Higashiyama, M., Gochi, A., Akaike, M., Ishikawa, T., Miura, T., Saruki, N., Bando, E., Kimura, H., Imamura, F., Moriyama, M., Ikeda, I., Chiba, A., Oshita, F., Imaizumi, A., Yamamoto, H., Miyano, H., Horimoto, K., Tochikubo, O., Mitsushima, T., Yamakado, M., & Okamoto, N. (2011). Plasma free amino acid profiling of five types of cancer patients and its application for early detection. PLoS ONE, 6, 1–12.
Lancha, A. H., Jr., Poortmans, J. R., & Pereira, L. O. (2009). The effect of 5 days of aspartate and asparagine supplementation on glucose transport activity in rat muscle. Cell Biochemistry and Function, 27, 552–557.
Marquezi, M. L., Roschel, H. A., dos Santa Costa, A., Sawada, L. A., & Lancha, A. H., Jr. (2003). Effect of aspartate and asparagine supplementation on fatigue determinants in intense exercise. International Journal of Sport Nutrition and Exercise Metabolism, 13, 65–75.
Flakoll, P., Sharp, R., Baier, S., Levenhagen, D., Carr, C., & Nissen, S. (2004). Effect of β-hydroxy-β-methylbutyrate, arginine, and lysine supplementation on strength, functionality, body composition, and protein metabolism in elderly women. Nutrition, 20, 445–451.
Friedman, M. (2004). Applications of the ninhydrin reaction for analysis of amino acids, peptides, and proteins to agricultural and biomedical sciences. Journal of Agricultural and Food Chemistry, 52, 385–406.
Feng, X., Cheng, H., Pan, Y., & Zheng, H. (2015). Development of glucose biosensors based on nanostructured graphene-conducting polyaniline composite. Biosensors and Bioelectronics, 70, 411–417.
Gholivand, M. B., & Khodadadian, M. (2014). Amperometric cholesterol biosensor based on the direct electrochemistry of cholesterol oxidase and catalase on a graphene/ionic liquid-modified glassy carbon electrode. Biosensors and Bioelectronics, 53, 472–478.
Kugimiya, A., & Matsuzaki, E. (2014). Microfluidic analysis of serine levels using seryl-tRNA synthetase coupled with spectrophotometric detection. Applied Biochemistry and Biotechnology, 174, 2527–2536.
Kugimiya, A., & Takamitsu, E. (2013). Spectrophotometric detection of histidine and lysine using combined enzymatic reactions. Materials Science and Engineering C, 33, 4867–4870.
Kugimiya, A., Fukada, R., & Funamoto, D. (2013). A luminol chemiluminescence method for sensing histidine and lysine using enzyme reactions. Analytical Biochemistry, 443, 22–26.
Kugimiya, A., & Fukada, R. (2015) Chemiluminescence detection of serine, proline, glycine, asparagine, leucine, and histidine by using corresponding aminoacyl-tRNA synthetases as recognition elements. Applied Biochemistry and Biotechnology. accepted.
Sekine, S., Nureki, O., Dubois, D. Y., Bernier, S., Chenevert, R., Lapointe, J., Vassylyev, D. G., & Yokoyama, S. (2003). ATP binding by glutamyl-tRNA synthetase is switched to the productive mode by tRNA binding. EMBO Journal, 22, 676–688.
Sekine, S., Shichiri, M., Bernier, S., Chênevert, R., Lapointe, J., & Yokoyama, S. (2006). Structural Bases of Transfer RNA-Dependent Amino Acid Recognition and Activation by Glutamyl-tRNA Synthetase. Structure, 14, 1791–1799.
Finarov, I., Moor, N., Kessler, N., Klipcan, L., & Safro, M. G. (2010). Structure of human cytosolic phenylalanyl-tRNA synthetase: evidence for kingdom-specific design of the active sites and tRNA binding patterns. Structure, 18, 343–353.
Ferreira, L. M. C., Costa, E. T., Lago, L. C., & Angnes, L. (2013). Miniaturized flow system based on enzyme modified PMMA microreactor for amperometric determination of glucose. Biosensors and Bioelectronics, 47, 539–544.
Wang, S., Su, P., & Yang, Y. (2012). Online immobilized enzyme microreactor for the glucose oxidase enzymolysis and enzyme inhibition assay. Analytical Biochemistry, 427, 139–143.
Acknowledgments
This work was partly supported by Hiroshima City University Grant for Special Academic Research (General Studies) and also supported by Grant-in-Aid for Scientific Research C (No. 25330344).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PPTX 71 kb)
Rights and permissions
About this article
Cite this article
Kugimiya, A., Konishi, H. & Fukada, R. Flow Analysis of Amino Acids by Using a Newly Developed Aminoacyl-tRNA Synthetase–Immobilized, Small Reactor Column–Based Assay. Appl Biochem Biotechnol 178, 924–931 (2016). https://doi.org/10.1007/s12010-015-1918-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1918-2