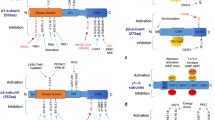
Abstract
Adenosine monophosphate-activated protein kinase (AMPK) is viewed as a privileged therapeutic target for several diseases such as cancer, diabetes, inflammation, obesity, etc. In addition, AMPK has entered the limelight of current drug discovery with its evolution as a key metabolic regulator. AMPK also plays a key role in the maintenance of cellular energy homeostasis. Structurally, AMPK is a heterotrimeric protein, which consists of three protein subunits (α, β, and γ). The crystal structure of AMPK was solved, and several computational studies including homology modeling, molecular docking, molecular dynamics, and QSAR have been reported in order to explore the structure and function of this diverse therapeutic target. In this review, we present a comprehensive up-to-date overview on the computational and molecular modeling approaches that have been carried out on AMPK in order to understand its structure, function, dynamics, and its drug binding landscape. Information provided in this review would be of great interest to a wide pool of researchers involved in the design of new molecules against various diseases where AMPK plays a predominant role.

ᅟ





Similar content being viewed by others
References
Carlson, C. A., & Kim, K. H. (1973). Regulation of hepatic acetyl coenzyme A carboxylase by phosphorylation and dephosphorylation. Journal of Biological Chemistry, 248, 378–80.
Beg, Z. H., Allmann, D. W., & Gibson, D. M. (1973). Modulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity with cAMP. Biochemical and Biophysical Research Communications, 54, 1362–1369.
Carling, D. (2004). The AMP-activated protein kinase cascade—a unifying system for energy control. Trends in Biochemical Sciences, 29, 18–24.
Hardie, D. G., & Carling, D. (1997). The AMP-activated protein kinase—fuel gauge of the mammalian cell? European Journal of Biochemistry, 246, 259–273.
Tripodi, F., Pagliarin, R., Fumagalli, G., Bigi, A., Fusi, P., Orsini, F., Frattini, M., & Coccetti, P. (2012). Synthesis and biological evaluation of 1,4-diaryl-2-azetidinones as specific anticancer agents: activation of adenosine monophosphate activated protein kinase and induction of apoptosis. Journal of Medicinal Chemistry, 55, 2112–2124.
Zhang, W., Wu, R., Zhang, F., Xu, Y., Liu, B., Yang, Y., Zhou, H., Wang, L., Wan, K., Xiao, X., & Zhang, X. (2012). Thiazolidinediones improve hepatic fibrosis in rats with non-alcoholic steatohepatitis by activating the adenosine monophosphate-activated protein kinase signalling pathway. Clinical and Experimental Pharmacology and Physiology, 39, 1026–1033.
Guo, H., Zhao, H., Kanno, Y., Li, W., Mu, Y., Kuang, X., Inouye, Y., Koike, K., Jiang, H., & Bai, H. (2013). A dihydrochalcone and several homoisoflavonoids from Polygonatum odoratum are activators of adenosine monophosphate-activated protein kinase. Bioorganic and Medicinal Chemistry Letters, 23, 3137–3139.
Guh, J. H., Chang, W. L., Yang, J., Lee, S. L., Wei, S., Wang, D., Kulp, S. K., & Chen, C. S. (2010). Development of novel adenosine monophosphate-activated protein kinase activators. Journal of Medicinal Chemistry, 53, 2552–2561.
Bae, E. J., Yang, Y. M., Kim, J. W., & Kim, S. G. (2007). Identification of a novel class of dithiolethiones that prevent hepatic insulin resistance via the adenosine monophosphate-activated protein kinase-p70 ribosomal S6 kinase-1 pathway. Hepatology, 46, 730–739.
Gruzman, A., Babai, G., & Sasson, S. (2009). Adenosine monophosphate-activated protein kinase (AMPK) as a new target for antidiabetic drugs: a review on metabolic, pharmacological and chemical considerations. The Review of Diabetic Studies, 6, 13–36.
Rana, S., Blowers, E. C., & Natarajan, A. (2015). Small molecule adenosine 5′-monophosphate activated protein kinase (AMPK) modulators and human diseases. Journal of Medicinal Chemistry, 58, 2–29.
Hawley, S. A., Fullerton, M. D., Ross, F. A., Schertzer, J. D., Chevtzoff, C., Walker, K. J., Peggie, M. W., Zibrova, D., Green, K. A., Mustard, K. J., Kemp, B. E., Sakamoto, K., Steinberg, G. R., & Hardie, D. G. (2012). The ancient drug salicylate directly activates AMP-activated protein kinase. Science, 336, 918–922.
Chen, Z., Wang, L., & Chen, Y. (2013). Antitumor mechanism of metformin via adenosine monophosphate-activated protein kinase (AMPK) activation. Zhongguo Fei Ai Za Zhi, 16, 427–432.
Xu, Q., Hao, X., Yang, Q., & Si, L. (2009). Resveratrol prevents hyperglycemia-induced endothelial dysfunction via activation of adenosine monophosphate-activated protein kinase. Biochemical and Biophysical Research Communications, 388, 389–394.
Chang, W., Zhang, M., Li, J., Meng, Z., Wei, S., Du, H., Chen, L., & Hatch, G. M. (2013). Berberine improves insulin resistance in cardiomyocytes via activation of 5′-adenosine monophosphate-activated protein kinase. Metabolism, 62, 1159–1167.
Wang, Y., Li, X., Guo, Y., Chan, L., & Guan, X. (2010). Alpha-lipoic acid increases energy expenditure by enhancing adenosine monophosphate-activated protein kinase-peroxisome proliferator-activated receptor-gamma coactivator-1alpha signaling in the skeletal muscle of aged mice. Metabolism, 59, 967–976.
Tseng, S. Y., Chao, T. H., Li, Y. H., Liu, P. Y., Lee, C. H., Cho, C. L., Wu, H. L., & Chen, J. H. (2015). Cilostazol improves high glucose-induced impaired angiogenesis in human endothelial progenitor cells and vascular endothelial cells as well as enhances vasculoangiogenesis in hyperglycemic mice mediated by the adenosine monophosphate-activated protein kinase pathway. Journal of Vascular Surgery. doi:10.1016/j.jvs.2014.10.103.
Tamrakar, P., Ibrahim, B. A., Gujar, A. D., & Briski, K. P. (2015). Estrogen regulates energy metabolic pathway and upstream adenosine 5′-monophosphate-activated protein kinase and phosphatase enzyme expression in dorsal vagal complex metabolosensory neurons during glucostasis and hypoglycemia. Journal of Neuroscience Research, 93, 321–932.
Li, J., Li, J., Yue, Y., Hu, Y., Cheng, W., Liu, R., Pan, X., & Zhang, P. (2014). Genistein suppresses tumor necrosis factor alpha-induced inflammation via modulating reactive oxygen species/Akt/nuclear factor kappaB and adenosine monophosphate-activated protein kinase signal pathways in human synoviocyte MH7A cells. Drug Design, Development and Therapy, 8, 315–323.
Lin, H. Y., Huang, B. R., Yeh, W. L., Lee, C. H., Huang, S. S., Lai, C. H., Lin, H., & Lu, D. Y. (2014). Antineuroinflammatory effects of lycopene via activation of adenosine monophosphate-activated protein kinase-alpha1/heme oxygenase-1 pathways. Neurobiology of Aging, 35, 191–202.
Ohira, M., Endo, K., Saiki, A., Miyashita, Y., Terai, K., Murano, T., Watanabe, F., Tatsuno, I., & Shirai, K. (2012). Atorvastatin and pitavastatin enhance lipoprotein lipase production in L6 skeletal muscle cells through activation of adenosine monophosphate-activated protein kinase. Metabolism, 61, 1452–1460.
Zong, J., Deng, W., Zhou, H., Bian, Z. Y., Dai, J., Yuan, Y., Zhang, J. Y., Zhang, R., Zhang, Y., Wu, Q. Q., Guo, H. P., Li, H. L., & Tang, Q. Z. (2013). 3,3′-Diindolylmethane protects against cardiac hypertrophy via 5′-adenosine monophosphate-activated protein kinase-α2. PLoS One, 8, e53427.
Xu, S. X., Zhou, Z. Q., Li, X. M., Ji, M. H., Zhang, G. F., & Yang, J. J. (2013). The activation of adenosine monophosphate-activated protein kinase in rat hippocampus contributes to the rapid antidepressant effect of ketamine. Behavioural Brain Research, 253, 305–359.
Ikegami, M., Ikeda, H., Ohashi, T., Ohsawa, M., Ishikawa, Y., Kai, M., Kamei, A., & Kamei, J. (2013). Olanzapine increases hepatic glucose production through the activation of hypothalamic adenosine 5′-monophosphate-activated protein kinase. Diabetes, Obesity & Metabolism, 15, 1128–1135.
Tang, H. C., & Chen, C. Y. (2014). In silico design for adenosine monophosphate-activated protein kinase agonist from traditional chinese medicine for treatment of metabolic syndromes. Evidence-based Complementary and Alternative Medicine. doi:10.1155/2014/928589.
Chuang, H. C., Chou, C. C., SK., K., & Chen, C. S. (2014). AMPK as a potential anticancer target—friend or foe? Current Pharmaceutical Design, 2020, 2607–2618.
Zhao, Z., Sui, Y., Gao, W., Cai, B., & Fan, D. (2015). Effects of diet on adenosine monophosphate-activated protein kinase activity and disease progression in an amyotrophic lateral sclerosis model. Journal of International Medical Research, 43, 67–79.
Hawley, S. A., Davison, M., Woods, A., Davies, S. P., Beri, R. K., Carling, D., & Hardie, D. G. (1996). Characterization of the AMP-activated protein kinase from rat liver and identification of threonine 172 as the major site at which it phosphorylates AMP-activated protein kinase. Journal of Biological Chemistry, 271, 27879–27887.
Hong, S. P., Leiper, F. C., Woods, A., Carling, D., & Carlson, M. (2003). Activation of yeast Snf1 and mammalian AMP-activated protein kinase by upstream kinases. Proceedings of the National Academy of Sciences of the United States of America, 100, 8839–8843.
Hawley, S. A., Pan, D. A., Mustard, K. J., Ross, L., Bain, J., Edelman, A. M., Frenguelli, B. G., & Hardie, D. G. (2005). Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for AMP-activated protein kinase. Cell Metabolism, 2, 9–19.
Hardie, D. G. (2004). The AMP-activated protein kinase pathway—new players upstream and downstream. Journal of Cell Science, 117, 5479–5487.
Scott, J. W., Norman, D. G., Hawley, S. A., Kontogiannis, L., & Hardie, D. G. (2002). Protein kinase substrate recognition studied using the recombinant catalytic domain of AMP-activated protein kinase and a model substrate. Journal of Molecular Biology, 2317, 2309–2323.
Davies, S. P., Helps, N. R., Cohen, P. T., & Hardie, D. G. (1995). 5′-AMP inhibits dephosphorylation, as well as promoting phosphorylation, of the AMP-activated protein kinase. Studies using bacterially expressed human protein phosphatase-2C alpha and native bovine protein phosphatase-2AC. FEBS Letters, 377, 421–425.
Cheung, P. C., Salt, I. P., Davies, S. P., Hardie, D. G., & Carling, D. (2000). Characterization of AMP-activated protein kinase gamma-subunit isoforms and their role in AMP binding. Biochemical Journal, 346, 659–669.
Sanders, M. J., Grondin, P. O., Hegarty, B. D., Snowden, M. A., & Carling, D. (2007). Investigating the mechanism for AMP activation of the AMP-activated protein kinase cascade. Biochemical Journal, 403, 139–148.
Hardie, D. G., Carling, D., & Gamblin, S. J. (2011). AMP-activated protein kinase: also regulated by ADP? Trends in Biochemical Sciences, 36, 470–477.
Xiao, B., Sanders, M. J., Underwood, E., Heath, R., Mayer, F. V., Carmena, D., Jing, C., Walker, P. A., Eccleston, J. F., Haire, L. F., Saiu, P., Howell, S. A., Aasland, R., Martin, S. R., Carling, D., & Gamblin, S. J. (2011). Structure of mammalian AMPK and its regulation by ADP. Nature, 472, 230–233.
Chen, L., Wang, J., Zhang, Y. Y., Yan, S. F., Neumann, D., Schlattner, U., Wang, Z. X., & Wu, J. W. (2012). AMP-activated protein kinase undergoes nucleotide-dependent conformational changes. Nature Structural and Molecular Biology, 19, 716–718.
Stapleton, D., Gao, G., Michell, B. J., Widmer, J., Mitchelhill, K., Teh, T., House, C. M., Witters, L. A., & Kemp, B. E. (1994). Mammalian 5′-AMP-activated protein kinase non-catalytic subunits are homologs of proteins that interact with yeast Snf1 protein kinase. Journal of Biological Chemistry, 269, 29343–29346.
Woods, A., Cheung, P. C., Smith, F. C., Davison, M. D., Scott, J., Beri, R. K., & Carling, D. (1996). Characterization of AMP-activated protein kinase beta and gamma subunits. Assembly of the heterotrimeric complex in vitro. Journal of Biological Chemistry, 271, 10282–10290.
Neumann, D., Woods, A., Carling, D., Wallimann, T., & Schlattner, U. (2003). Mammalian AMP-activated protein kinase: functional, heterotrimeric complexes by co-expression of subunits in Escherichia coli. Protein Expression and Purification, 30, 230–237.
Hudson, E. R. (2003). A novel domain in AMP-activated protein kinase causes glycogen storage bodies similar to those seen in hereditary cardiac arrhythmias. Current Biology, 13, 861–866.
Polekhina, G., Gupta, A., Michell, B. J., van Denderen, B., Murthy, S., Feil, S. C., Jennings, I. G., Campbell, D. J., Witters, L. A., Parker, M. W., Kemp, B. E., & Stapleton, D. (2003). AMPK beta subunit targets metabolic stress sensing to glycogen. Current Biology, 13, 867–871.
Oakhill, J. S., Chen, Z. P., Scott, J. W., Steel, R., Castelli, L. A., Ling, N., Macaulay, S. L., & Kemp, B. E. (2010). β-Subunit myristoylation is the gatekeeper for initiating metabolic stress sensing by AMP-activated protein kinase (AMPK). Proceedings of the National Academy of Sciences of the United States of America, 107, 19237–19241.
Xiao, B., Sanders, M. J., Carmena, D., Bright, N. J., Haire, L. F., Underwood, E., Patel, B. R., Heath, R. B., Walker, P. A., Hallen, S., Giordanetto, F., Martin, S. R., Carling, D., & Gamblin, S. J. (2013). Structural basis of AMPK regulation by small molecule activators. Nature Communications, 4, 3017.
Polekhina, G., Gupta, A., van Denderen, B. J., Feil, S. C., Kemp, B. E., Stapleton, D., & Parker, M. W. (2005). Structural basis for glycogen recognition by AMP-activated protein kinase. Structure, 13, 1453–1462.
Walker, J. R., Wybenga-Groot, L., Finerty, P. J., Newman, E., MacKenzie, F. M., Weigelt, J., Sundstrom, M., Arrowsmith, C., Edwards, A., Bochkarev, A., Dhe-Paganon, S. Structure of the glycogen-binding domain of the AMP-activated protein kinase beta2 subunit. Protein data bank. 10.2210/pdb2f15/pdb.
Nayak, V., Zhao, K., Wyce, A., Schwartz, M. F., Lo, W. S., Berger, S. L., & Marmorstein, R. (2006). Structure and dimerization of the kinase domain from yeast Snf1, a member of the Snf1/AMPK protein family. Structure, 14, 477–485.
Littler, D. R., Walker, J. R., Davis, T., Wybenga-Groot, L. E., Finerty, P. J., Newman, E., Mackenzie, F., & Dhe-Paganon, S. (2010). A conserved mechanism of autoinhibition for the AMPK kinase domain: ATP-binding site and catalytic loop refolding as a means of regulation. Acta Crystallographica Section F: Structural Biology and Crystallization Communications, 66, 143–151.
Xia, B., Hu, J. Solution structure of autoinhibitory domain of human AMP-activated protein kinase catalytic subunit. Protein data bank. 10.2210/pdb2ltu/pdb.
Koay, A., Petrie, E., Gorman, M., di Paolo, A., Mobbs, J., Parker, M., Stapleton, D., Gooley, P. Solution NMR structure of the apo-form of the beta2 carbohydrate module of AMP-activated protein kinase. doi:10.2210/pdb2lu3/pdb.
Townley, R., & Shapiro, L. (2007). Crystal structures of the adenylate sensor from fission yeast AMP-activated protein kinase. Science, 315, 1726–1729.
Amodeo, G. A., Rudolph, M. J., & Tong, L. (2007). Crystal structure of the heterotrimer core of Saccharomyces cerevisiae AMPK homologue SNF1. Nature, 449, 492–495.
Jin, X., Townley, R., & Shapiro, L. (2007). Structural insight into AMPK regulation: ADP comes into play. Structure, 15, 1285–1295.
Day, P., Sharff, A., Parra, L., Cleasby, A., Williams, M., Horer, S., Nar, H., Redemann, N., Tickle, I., & Yon, J. (2007). Structure of a CBS-domain pair from the regulatory 1 subunit of human AMPK in complex with AMP and ZMP. Acta Crystallographica Section D: Biological Crystallography, 63, 587–596.
Xiao, B., Heath, R., Saiu, P., Leiper, F. C., Leone, P., Jing, C., Walker, P. A., Haire, L., Eccleston, J. F., Davis, C. T., Martin, S. R., Carling, D., & Gamblin, S. J. (2007). Structural basis for AMP binding to mammalian AMP-activated protein kinase. Nature, 449, 496–500.
Handa, N., Takagi, T., Saijo, S., Kishishita, S., Takaya, D., Toyama, M., Terada, T., Shirouzu, M., Suzuki, A., Lee, S., Yamauchi, T., Okada-Iwabu, M., Iwabu, M., Kadowaki, T., Minokoshi, Y., & Yokoyama, S. (2011). Structural basis for compound C inhibition of the human AMP-activated protein kinase α2 subunit kinase domain. Acta Crystallographica. Section D, Biological Crystallography, 67, 480–487.
Chen, L., Jiao, Z. H., Zheng, L. S., Zhang, Y. Y., Xie, S. T., Wang, Z. X., & Wu, J. W. (2009). Structural insight into the autoinhibition mechanism of AMP-activated protein kinase. Nature, 459, 1146–1149.
Cho, Y. S., Lee, J. I., Shin, D., Kim, H. T., Jung, H. Y., Lee, T. G., Kang, L. W., Ahn, Y. J., Cho, H. S., & Heo, Y. S. (2010). Molecular mechanism for the regulation of human ACC2 through phosphorylation by AMPK. Biochemical and Biophysical Research Communications, 391, 187–92.
Gomez-Garcia, I., Oyenarte, I., & Martinez-Cruz, L. A. (2010). The crystal structure of protein MJ1225 from Methanocaldococcus jannaschii shows strong conservation of key structural features seen in the eukaryal gamma-AMPK. Journal of Molecular Biology, 399, 53–70.
Rudolph, M. J., Amodeo, G. A., & Tong, L. (2010). An inhibited conformation for the protein kinase domain of the Saccharomyces cerevisiae AMPK homolog Snf1. Acta Crystallographica Section F: Structural Biology and Crystallization Communications, 66, 999–1002.
Mayer, F. V., Heath, R., Underwood, E., Sanders, M. J., Carmena, D., McCartney, R. R., Leiper, F. C., Xiao, B., Jing, C., Walker, P. A., Haire, L. F., Ogrodowicz, R., Martin, S. R., Schmidt, M. C., Gamblin, S. J., & Carling, D. (2011). ADP regulates SNF1, the Saccharomyces cerevisiae homolog of AMP-activated protein kinase. Cell Metabolism, 14, 707–714.
Zhan, Y., Chen, Y., Zhang, Q., Zhuang, J., Tian, M., Chen, H., Zhang, L., Zhang, H., He, J., Wang, W., Wu, R., Wang, Y., Shi, C., Yang, K., Li, A., Xin, Y., Li, T. Y., Yang, J. Y., Zheng, Z., Yu, C., Lin, S., Chang, C., Huang, P., Lin, T., & Wu, Q. (2012). Crystal structure of human nur77 ligand-binding domain in complex with ethyl 2-[2,3,4 trimethoxy-6(1-octanoyl)phenyl]acetate. Nature Chemical Biology, 8, 897–904.
Chen, L., Xin, F. J., Wang, J., Hu, J., Zhang, Y. Y., Wan, S., Cao, L. S., Lu, C., Li, P., Yan, S. F., Neumann, D., Schlattner, U., Xia, B., Wang, Z. X., & Wu, J. W. (2013). Conserved regulatory elements in AMPK. Nature, 498, E8–10.
Calabrese, M. F., Rajamohan, F., Harris, M. S., Caspers, N. L., Magyar, R., Withka, J. M., Wang, H., Borzilleri, K. A., Sahasrabudhe, P. V., Hoth, L. R., Geoghegan, K. F., Han, S., Brown, J., Subashi, T. A., Reyes, A. R., Frisbie, R. K., Ward, J., Miller, R. A., Landro, J. A., Londregan, A. T., Carpino, P. A., Cabral, S., Smith, A. C., Conn, E. L., Cameron, K. O., Qiu, X., & Kurumbail, R. G. (2014). Structural basis for AMPK activation: natural and synthetic ligands regulate kinase activity from opposite poles by different molecular mechanisms. Structure, 22, 1161–1172.
Li, X., Wang, L., Zhou, X. E., Ke, J., de Waal, P. W., Gu, X., Tan, M. H., Wang, D., Wu, D., Xu, H. E., & Melcher, K. (2015). Structural basis of AMPK regulation by adenine nucleotides and glycogen. Cell Research, 25, 50–66.
Carling, D., Clarke, P. R., Zammit, V. A., & Hardie, D. G. (1989). Purification and characterization of the AMP-activated protein kinase. Copurification of acetyl-CoA carboxylase kinase and 3-hydroxy-3-methylglutaryl-CoA reductase kinase activities. European Journal of Biochemistry, 186, 129–136.
Zhu, L., Chen, L., Zhou, X. M., Zhang, Y. Y., Zhang, Y. J., Zhao, J., Ji, S. R., Wu, J. W., & Wu, Y. (2011). Structural insights into the architecture and allostery of full-length AMP-activated protein kinase. Structure, 19, 515–522.
Foufelle, F., & Ferre, P. (2005). Role of adenosine monophosphate-activated protein kinase in the control of energy homeostasis. Current Opinion in Clinical Nutrition and Metabolic Care, 8, 355–360.
Gauthier, M. S., O’Brien, E. L., Bigornia, S., Mott, M., Cacicedo, J. M., Xu, X. J., Gokce, N., Apovian, C., & Ruderman, N. (2011). Decreased AMP-activated protein kinase activity is associated with increased inflammation in visceral adipose tissue and with whole-body insulin resistance in morbidly obese humans. Biochemical and Biophysical Research Communications, 404, 382–387.
Peairs, A., Radjavi, A., Davis, S., Li, L., Ahmed, A., Giri, S., & Reilly, C. M. (2009). Activation of AMPK inhibits inflammation in MRL/lpr mouse mesangial cells. Clinical and Experimental Immunology, 156, 542–551.
Gul, T., Balkhi, H. M., & Haq, E. (2013). AMPK: a potent target for treating obesity. International Journal of Pharmaceutics Chemestry Biological Science, 3, 801–813.
Phoenix, K. N., Vumbaca, F., & Claffey, K. P. (2009). Therapeutic metformin/AMPK activation promotes the angiogenic phenotype in the ERalpha negative MDA-MB-435 breast cancer model. Breast Cancer Research and Treatment, 113, 101–111.
Hadad, S. M., Appleyard, V., & Thompson, A. M. (2009). Therapeutic metformin/AMPK activation promotes the angiogenic phenotype in the ERalpha negative MDA-MB-435 breast cancer model. Breast Cancer Research and Treatment, 114, 391.
Shibata, R., Sato, K., Pimentel, D. R., Takemura, Y., Kihara, S., Ohashi, K., Funahashi, T., Ouchi, N., & Walsh, K. (2005). Adiponectin protects against myocardial ischemia-reperfusion injury through AMPK- and COX-2-dependent mechanisms. Nature Medicine, 11, 1096–1103.
Miller, E. J., Li, J., Leng, L., McDonald, C., Atsumi, T., Bucala, R., & Young, L. H. (2008). Macrophage migration inhibitory factor stimulates AMP-activated protein kinase in the ischaemic heart. Nature, 451, 578–582.
Won, J. S., Im, Y. B., Kim, J., Singh, A. K., & Singh, I. (2010). Involvement of AMP-activated-protein-kinase (AMPK) in neuronal amyloido- genesis. Biochemical and Biophysical Research Communications, 399, 487–491.
Morais, A. S., Abarca-Quinones, J., Guigas, B., Viollet, B., Starkel, P., Horsmans, Y., & Leclercq, I. A. (2010). Development of hepatic fibrosis occurs normally in AMPK-deficient mice. Clinical Science, 118, 411–420.
Carling, D., Mayer, F. V., Sanders, M. J., & Gamblin, S. J. (2011). AMP-activated protein kinase: nature’s energy sensor. Nature Chemical Biology, 7, 512–518.
Mor, V., & Unnikrishnan, M. K. (2011). 5′-adenosine monophosphate-activated protein kinase and the metabolic syndrome. Endocrine, Metabolic & Immune Disorders Drug Targets, 11, 206–216.
Baumann, P., Mandl-Weber, S., Emmerich, B., Straka, C., & Schmidmaier, R. (2007). Inhibition of adenosine monophosphate-activated protein kinase induces apoptosis in multiple myeloma cells. Anti-Cancer Drugs, 18, 405–410.
Weisova, P., Davila, D., Tuffy, L. P., Ward, M. W., Concannon, C. G., & Prehn, J. H. (2011). Role of 5′-adenosine monophosphate-activated protein kinase in cell survival and death responses in neurons. Antioxidants and Redox Signaling, 14, 1863–1876.
Filippov, S., Pinkosky, S. L., & Newton, R. S. (2014). LDL-cholesterol reduction in patients with hypercholesterolemia by modulation of adenosine triphosphate-citrate lyase and adenosine monophosphate-activated protein kinase. Current Opinion in Lipidology, 25, 309–315.
Osler, M. E., & Zierath, J. R. (2008). Adenosine 5′-monophosphate-activated protein kinase regulation of fatty acid oxidation in skeletal muscle. Endocrinology, 149, 935–941.
Ropelle, E. R., Pauli, J. R., Zecchin, K. G., Ueno, M., de Souza, C. T., Morari, J., Faria, M. C., Velloso, L. A., Saad, M. J., & Carvalheira, J. B. (2007). A central role for neuronal adenosine 5′-monophosphate-activated protein kinase in cancer-induced anorexia. Endocrinology, 148, 5220–5229.
Huang, H., Chen, C. Y., Chen, H. Y., Tsai, F. J., & Chen, C. Y. C. (2010). Computational screening and QSAR analysis for design of AMP-activated protein kinase agonist. Journal of the Taiwan Institute of Chemical Engineers, 41, 352–359.
Siwiak, M., Edelman, A., & Zielenkiewicz, P. (2012). Structural models of CFTR–AMPK and CFTR–PKA interactions: R-domain flexibility is a key factor in CFTR regulation. Journal of Molecular Modelling, 18, 83–90.
Park, H., Eom, J. W., & Kim, Y. H. (2014). Consensus scoring approach to identify the inhibitors of AMP-activated protein kinase α2 with virtual screening. Journal of Chemical Information and Modeling, 54, 2139–2146.
Zhang, Y., Wang, Y., Bao, C., Xu, Y., Shen, H., Chen, J., Yan, J., & Chen, Y. (2012). Metformin interacts with AMPK through binding to γ subunit. Molecular and Cellular Biochemistry, 368, 69–76.
Raj, C. G. D., Sarojini, B. K., Khan, M. T. H., & Raghavendra, R. (2013). In vivo antidiabetic activity and in silico studies on adenosine monophosphate-activated protein kinase (AMPK) of (2E,5E)-2,5-bis(4-hydroxy-3-methoxybenzylidene) cyclopentanone. Medicinal Chemistry Research, 22, 2430–2436.
Pang, T., Zhang, Z. S., Gu, M., Qiu, B. Y., Yu, L. F., Cao, P. R., Shao, W., Su, M. B., Li, J. Y., Nan, F. J., & Li, J. (2008). Small molecule antagonizes autoinhibition and activates AMP-activated protein kinase in cells. Journal of Biological Chemistry, 283, 16051–16060.
Peng, C., & Head-Gordon, T. (2011). The dynamical mechanism of auto-inhibition of AMP activated protein kinase. PLoS Computational Biology, 7, e1002082.
Chapnik, N., Genzer, Y., Ben-Shimon, A., Niv, M. Y., & Froy, O. (2014). AMPK-derived peptides reduce blood glucose levels but lead to fat retention in the liver of obese mice. Journal of Endocrinology, 221, 89–99.
Chang, Y. H., Ho, T. Y., Wu, C. H., Chen, C. Y., Huang, H. J., Tsai, F. J., Tsai, C. H., & Chen, C. Y. C. (2009). Study of AMP-activated protein kinase agonists by structure-based drug designing. Advances in Materials Research, 79–82, 2187–2190.
Gruzman, A., Shamni, O., Ben Yakir, M., Sandovski, D., Elgart, A., Alpert, E., Cohen, G., Hoffman, A., Katzhendler, Y., Cerasi, E., & Sasson, S. (2008). Novel d-xylose derivatives stimulate muscle glucose uptake by activating AMP-activated protein kinase alpha. Journal of Medicinal Chemistry, 51, 8096–8108.
Wang, Y., Kong, W., Li, Y., Tang, S., Li, Z., Li, Y., Shan, Y., Bi, C., Jiang, J., & Song, D. (2012). Synthesis and structure–activity relationship of berberine analogues in LDLR up-regulation and AMPK activation. Bioorganic and Medicinal Chemistry, 20, 6552–6558.
Doss, C. G. P., & Nagasundaram, N. (2014). Molecular docking and molecular dynamics study on the effect of ERCC1 deleterious polymorphisms in ERCC1-XPF heterodimer. Applied Biochemistry and Biotechnology, 172, 1265–1281.
Roy, D., Kumar, V., Acharya, K. K., & Thirumurugan, K. (2014). Probing the binding of syzygium-derived α-glucosidase inhibitors with N- and C-terminal human maltase glucoamylase by docking and molecular dynamics simulation. Applied Biochemistry and Biotechnology, 172, 102–114.
Sahoo, B. R., Swain, B., Dikhit, M. R., Basu, M., Bej, A., Jayasankar, P., & Samanta, M. (2013). Activation of nucleotide-binding oligomerization domain 1 (NOD1) receptor signaling in labeo rohita by iE-DAP and identification of ligand-binding key motifs in NOD1 by molecular modeling and docking. Applied Biochemistry and Biotechnology, 170, 1282–1309.
Pulaganti, M., Banaganapalli, B., Mulakayala, C., Chitta, S. K., & Anuradha, C. M. (2014). Molecular modeling and docking studies of O-succinylbenzoate synthase of M. tuberculosis—a potential target for antituberculosis drug design. Applied Biochemistry and Biotechnology, 172, 1407–1432.
Pinheiro, A. S., Duarte, J. B. C., Alves, C. N., & Alberto de Molfetta, F. (2015). Virtual screening and molecular dynamics simulations from a bank of molecules of the amazon region against functional NS3-4A protease-helicase enzyme of hepatitis C virus. Applied Biochemistry and Biotechnology, 176, 1709–1721.
Honarparvar, B., Govender, T., Maguire, G. E., Soliman, M. E., & Kruger, H. G. (2014). Integrated approach to structure-based enzymatic drug design: molecular modeling, spectroscopy, and experimental bioactivity. Chemical Reviews, 114, 493–537.
Ramakrishnan, C., & Ramachandran, G. N. (1965). Stereochemical criteria for polypeptide and protein chain conformations. II. Allowed conformations for a pair of peptide units. Biophysical Journal, 5, 909–933.
Lindorff-Larsen, K., Piana, S., Palmo, K., Maragakis, P., Klepeis, J. L., Dror, R. O., & Shaw, D. E. (2010). Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins: Structure, Function, and Bioinformatics, 78, 1950–1958.
SYBYL (2007) Tripos Inc, St Louis.
Molecular Operating Environment (MOE), Chemical Computing Group Inc., 1010 Sherbooke St. West, Suite #910, Montreal, QC, Canada. .
Shoichet, B. K., McGovern, S. L., Wei, B., & Irwin, J. J. (2002). Lead discovery using molecular docking. Current Opinion in Chemical Biology, 6, 439–446.
Lipinski, C. A. (2004). Lead- and drug-like compounds: the rule-of-five revolution. Drug Discovery Today: Technologies, 1, 337–341.
Morris, G. M., Huey, R., Lindstrom, W., Sanner, M. F., Belew, R. K., Goodsell, D. S., & Olson, A. J. (2009). AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. Journal of Computational Chemistry, 30, 2785–2791.
Laskowski, R. A., & Swindells, M. B. (2011). LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. Journal of Chemical Information and Modeling, 51, 2778–2786.
Alder, B. J., & Wainwright, T. (1959). Studies in molecular dynamics. I. General method. Journal of Chemical Physics, 31, 459–466.
Christen, M., Hunenberger, P. H., Bakowies, D., Baron, R., Burgi, R., Geerke, D. P., Heinz, T. N., Kastenholz, M. A., Krautler, V., Oostenbrink, C., Peter, C., Trzesniak, D., & van Gunsteren, W. F. (2005). The GROMOS software for biomolecular simulation: GROMOS05. Journal of Computational Chemistry, 26, 1719–1751.
Chen, Q., & Chen, Y. P. (2006). Mining frequent patterns for AMP-activated protein kinase regulation on skeletal muscle. BMC Bioinformatics, 7, 394.
Patrick, G. L. (2005). An introduction to medicinal chemistry. New York: Oxford University Press.
Acknowledgments
The financial support from the School of Health Sciences, University of KwaZulu-Natal, Westville, Durban, South Africa is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramesh, M., Vepuri, S.B., Oosthuizen, F. et al. Adenosine Monophosphate-Activated Protein Kinase (AMPK) as a Diverse Therapeutic Target: A Computational Perspective. Appl Biochem Biotechnol 178, 810–830 (2016). https://doi.org/10.1007/s12010-015-1911-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1911-9