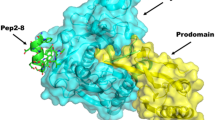
Abstract
Many studies have demonstrated the role of elevated levels of serum cholesterol in the pathogenesis of atherosclerosis and coronary heart disease. Various drugs targeting the key enzymes involved in the cholesterol biosynthesis pathway have been investigated for the treatment of hypercholesterolemia. Human squalene synthase has been one of the most important targets for therapeutic intervention. In the present study, we used the recombinant human squalene synthase as the lure for screening the peptide inhibitors from phage-displayed random peptide library. The tightly bound phages and their derived peptides were further evaluated based on their potential binding capabilities, molecular modeling characteristics and predicted absorption, distribution, metabolism, excretion, toxicity (ADMET) properties. Several hexa-peptides and tetra-peptides were finally synthesized to assay their inhibitory effects toward the recombinant human squalene synthase. The results demonstrated that the hexa-peptide FTACNW and tetra-peptide VACL can inhibit human squalene synthase effectively (with IC50 values near 100 μM) and may have potential to develop further as future hypocholesterolemia agents.



Similar content being viewed by others
Abbreviations
- ADMET:
-
Absorption, distribution, metabolism, excretion, toxicity
- ELISA:
-
Enzyme-linked immune-sorbent assay
- FPP:
-
Farnesyl diphosphate
- hSQS:
-
Human squalene synthase
- HMG-CoA:
-
3-hydroxy-3-methylglutaryl-coenzyme A
- hHMGR:
-
human HMG-CoA reductase
- hHMGRI:
-
human HMG-CoA reductase inhibitor
- IPTG:
-
Isopropyl β-d-1-thiogalactopyranoside
- LDL-R:
-
Low-density lipoprotein receptor
- MTT:
-
3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide
- PCR:
-
Polymerase chain reaction
- PSQPP:
-
Presqualene diphosphate
- SDS-PAGE:
-
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
References
Norenberg, D. (1984). Lipid research clinics program. Journal of the American Medical Association, 252, 2545–2548.
Brown, A. S., Bakker-Arkema, R. G., Yellen, L. R., Henley, W. J., Guthrie, R., Campbell, C. F., Koren, M., Woo, W., McLain, R., & Black, D. M. (1998). Treating patients with documented atherosclerosis to national cholesterol education program- recommended low-density-lipoprotein cholesterol goals with atorvastatin, fluvastatin, lovastatin and simvastatin. Journal of the American College of Cardiology, 32, 665–672.
Hiyoshi, H., Yanagimachi, M., Ito, M., Ohtsuka, I., Yoshida, I., Saeki, T., & Tanaka, H. (2000). Effect of ER-27856, a novel squalene synthase inhibitor, on plasma cholesterol in rhesus monkeys: comparison with 3-hydroxy-3-methylglutaryl-CoA reductase inhibitors. Journal of Lipid Research, 41, 1136–1144.
Satoh, K., Yamato, A., Nakai, T., Hoshi, K., & Ichihara, K. (1995). Effects of 3-hydroxy-3 -methylglutaryl coenzyme A reductase inhibitors on mitochondrial respiration in ischaemic dog hearts. British Journal of Pharmacology, 116, 1894–1898.
Tansey, T. R., & Shechter, I. (2000). Structure and regulation of mammalian squalene synthase. Biochimica et Biophysica Acta, 1529, 49–62.
Do, R., Kiss, R. S., Gaudet, D., & Engert, J. C. (2009). Squalene synthase: a critical enzyme in the cholesterol biosynthesis pathway. Clinical Genetics, 75, 19–29.
Pandit, J., Danley, D. E., Schulte, G. K., Mazzalupo, S., Pauly, T. A., Hayward, C. M., Hamanaka, E. S., & Thompson, J. F. (2000). Crystal structure of human squalene synthase. A key enzyme in cholesterol biosynthesis. The Journal of Biological Chemistry, 275, 30610–30617.
Liao, J. K. (2011). Squalene synthase inhibitor lapaquistat acetate: could anything be better than statins? Circulation, 123, 1925–1928.
Stein, E. A., Bays, H., O’Brien, D., Pedicano, J., Piper, E., & Spezzi, A. (2011). Lapaquistat acetate: development of a squalene synthase inhibitor for the treatment of hypercholesterolemia. Circulation, 123, 1974–1985.
Singh, B. P., Vij, S., & Hati, S. (2014). Functional significance of bioactive peptides derived from soybean. Peptides, 54, 171–179.
Cho, S. J., Juillerat, M. A., & Lee, C. H. (2007). Cholesterol lowering mechanism of soybean protein hydrolysate. Journal of Agriculture and Food Chemistry, 55, 10599–10604.
Toutouzas, K., Drakopoulou, M., Skoumas, I., & Stefanadis, C. (2010). Advancing therapy for hypercholesterolemia. Expert Opinion on Pharmacotherapy, 11, 1659–1672.
Smith, G. P. (1985). Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science, 228, 1315–1317.
Krumpe, L. R., & Mori, H. T. (2006). The use of phage-displayed peptide libraries to develop tumor-targeting drugs. International Journal of Peptide Research and Therapy, 12, 79–91.
Mukhija, S., & Erni, B. (1997). Phage display selection of peptides against enzyme I of the phosphoenolpyruvate-sugar phosphotransferase system (PTS). Molecular Microbiology, 25, 1159–1166.
Lin, K. C., Chen, C. Y., Huang, K. J., Chang, C. W., Lin, S. P., Chang, D. K., Lin, M. R., & Shiuan, D. (2012). A dodecapeptide (YQVTQSKVMSHR) exhibits antibacterial effect and induces cell aggregation in Escherichia coli. Applied Microbiology and Biotechnology, 94, 755–762.
Liu, C. I., Liu, Y., Song, G. Y., Yin, F., Hensler, M. E., Jeng, W. Y., Nizet, V., Wang, A. H. J., & Oldfield, E. (2008). A cholesterol biosynthesis inhibitor blocks Staphylococcus aureus virulence. Science, 319, 1391–1394.
Lin, S. H., Chang, D. K., Chou, M. J., & Shiuan, D. (2015). Peptide inhibitors of human HMG-CoA reductase as potential hypocholesterolemic agents. Biochemistry and Biophysics Research Communication, 456, 104–109.
Thompson, J. F., Danley, D. E., Mazzalupo, S., Milos, P. M., Lira, M. E., & Harwood, H. J. (1998). Truncation of human squalene synthase yields active crystallable protein. Archive of Biochemistry and Biophysics, 350, 283–290.
Yang, W. J., Lai, J. F., Peng, K. C., Chiang, H. J., Weng, C. N., & Shiuan, D. (2005). Epitope mapping of Mycoplasma hyopneumoniae using phage displayed peptide libraries and the immune responses of the selected phagotopes. Journal of Immunological Methods, 304, 15–29.
Mayo, S. L., Olafson, B. D., & Goddartd, W. A. (1990). Dreiding: a genetic force field for molecular simulation. Journal of Physical Chemistry, 94, 8897–8909.
Venkatachalam, C. M., Jiang, X., Oldfield, T., & Waldman, M. (2003). LigandFit : a novel method for the shape-directed rapid docking of ligands to protein active sites. Journal of Molecular Graphics and Modeling, 21, 289–307.
Pak, V. V., Koo, M., Kwon, D. Y., Shakhidoyatov, K. M., & Yun, L. (2010). Peptide fragmentation as an approach in modeling of an active peptide and designing a competitive inhibitory peptide for HMG-CoA reductase. Bioorganic and Medicinal Chemistry, 18, 4300–4309.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
van Meerloo, J., Kaspers, G. J., & Cloos, J. (2001). Cell sensitivity assays: the MTT assay. Methods in Molecular Biology, 731, 237–245.
Sealey-Cardona, M., Cammerer, S., Jones, S., Ruiz-Pérez, L. M., Brun, R., Gilbert, I. H., Urbina, J. A., & González-Pacanowska, D. (2007). Kinetic characterization of squalene synthase from Trypanosoma cruzi: selective inhibition by quinuclidine derivatives. Antimicrobial Agents and Chemotherapy, 51, 2123–2129.
Ichikawa, M., Yokomizo, A., Itoh, M., Usui, H., Shimizu, H., Suzuki, M., Terayama, K., Kanda, A., & Sugita, K. (2011). Discovery of a new 2-aminobenzhydrol template for highly potent squalene synthase inhibitors. Bioorganic and Medicinal Chemistry, 19, 1930–1949.
Liu, C. I., Jeng, W. Y., Chang, W. J., Shih, M. F., Ko, T. P., & Wang, A. H. J. (2014). Structural insights into the catalytic mechanism of human squalene synthase. Acta Crystallographica, Section D, 70, 231–241.
Dale Poulter, C., Capson, T. L., Thompson, M. D., & Bard, R. S. (1989). Squalene synthetase, inhibition by ammonium analogs of carbocationic intermediates in the conversion of presqualene diphosphate to squalene. Journal of the American Chemical Society, 111, 3734–3739.
Flint, O. P., Masters, B. A., Gregg, R. E., & Durham, S. K. (1997). Inhibition of cholesterol synthesis by squalene synthase inhibitors does not induce myotoxicity in vitro. Toxicology and Applied Pharmacology, 145, 91–98.
Nishimoto, T., Amano, Y., Tozawa, R., Ishikawa, E., Imura, Y., Yukimasa, H., & Sugiyama, Y. (2003). Lipid-lowering properties of TAK-475, a squalene synthase inhibitor, in vivo and in vitro. British Journal of Pharmacology, 139, 911–918.
Charlton-Menys, V., & Durrington, P. N. (2007). Squalene synthase inhibitors: clinical pharmacology and cholesterol-lowering potential. Drugs, 67, 11–16.
Craik, D. J., Fairlie, D. P., Liras, S., & Price, D. (2013). The future of peptide-based drugs. Chemical Biology and Drug Design, 81, 136–147.
Vlieghe, P., Lisowski, V., Martinez, J., & Khrestchatisky, M. (2010). Synthetic therapeutic peptides: science and market. Drug Discovery Today, 15, 40–56.
Acknowledgments
This work was partially supported by grants from National Science Council ROC (NSC97 -2311-B259-04-MY3 to D. Shiuan, and NSC99-2113-M-001-024-MY3 to D.-K.Chang.)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shiuan, D., Lin, HK., Chen, YH. et al. Exploration of Peptide Inhibitors of Human Squalene Synthase through Molecular Modeling and Phage Display Technique. Appl Biochem Biotechnol 178, 312–323 (2016). https://doi.org/10.1007/s12010-015-1873-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1873-y