Abstract
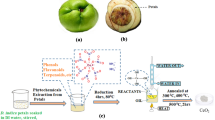
Monodispersed cerium oxide nanoparticle has been synthesized by microwave-mediated hydrothermal as well as microwave-mediated solvothermal synthesis. X-ray diffraction (XRD) data shows that the synthesized particles are single phase. SEM and TEM analysis suggest that particle synthesized by microwave-mediated solvothermal method are less agglomerated. In vitro toxicology study of the synthesized nanoceria particles has shown good free radical scavenging activity for NO and 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical assayed except superoxide radical within a concentration range of 25 to 75 ng ml−1. Nanoceria particle also showed inhibition of Fe-ascorbate-induced lipid peroxidation (LPx) in chick liver mitochondrial fractions. Solvothermally synthesized nanoceria showed better protection against Fe-ascorbate-induced LPx than the hydrothermal one while the hydrothermally synthesized nanoceria showed better DPPH and NO scavenging activity. The ceria nanoparticles also prevented Fe-ascorbate-H2O2-induced carbonylation of bovine serum albumin in a dose-dependent manner. At higher concentration, i.e., 100 ng ml−1, the synthesized nanoparticles showed a reverse trend in all the parameters measured indicating its toxicity at higher doses.







Similar content being viewed by others
References
El-Sayed, M. A. (2001). Some interesting properties of metals confined in time and nanometer space of different shapes. Accounts of Chemical Research, 34(4), 257–264.
Chen, S., & Yang, Y. (2002). Magnetoelectrochemistry of gold nanoparticle quantized capacitance charging. Journal of the American Chemical Society, 124(19), 5280–5281.
Lewis, L. N. (1993). Chemical catalysis by colloids and clusters. Chemical Reviews, 93(8), 2693–2730.
Chen, M. S., & Goodman, D. W. (2004). The structure of catalytically active gold on titania. Science, 306, 252–255.
Kim, S. W., Kim, M., Lee, W. Y., & Hyeon, T. (2002). Fabrication of hollow palladium spheres and their successful application to the recyclable heterogeneous catalyst for suzuki coupling reactions. Journal of the American Chemical Society, 124(26), 7642–7643.
Chen, H. M., Chen, C. K., Chang, Y. C., Tsai, C. W., Liu, R. S., Hu, S. F., Chang, W. S., & Chen, K. H. (2010). Monolayer-quantum dots sensitized zno nanowires-array photoelectrodes: true efficiency for water splitting. Angewandte Chemie International Edition, 49(34), 5966–5969.
Sinha, A. K., Seelan, S., Tsubota, S., & Haruta, M. A. (2004). A three-dimensional mesoporous titanosilicate support for gold nanoparticles: vapor-phase epoxidation of propene with high conversion. Angewandte Chemie International Edition, 43(12), 1546–1548.
Valden, M., Lai, X., & Goodman, D. W. (1998). Onset of catalytic activity of gold clusters on titania with the appearance of nonmetallic properties. Science, 281(5383), 1647–1650.
Zhou, S., Mcllwrath, K., Jackson, G., & Eichhorn, B. (2006). Enhanced CO tolerance for hydrogen activation in Au-Pt dendritic heteroaggregate nanostructures. Journal of the American Chemical Society, 128(6), 1780–1781.
Peyser, L. A., Vinson, A. E., Bartko, A. P., & Dickson, R. M. (2001). Photoactivated fluorescence from individual silver nanoclusters. Science, 291(5501), 103–106.
Chen, H. M., Liu, R. S., Li, H., & Zeng, H. C. (2006). Generating isotropic superparamagnetic interconnectivity for the two-dimensional organization of nanostructured building blocks. Angewandte Chemie International Edition, 45(17), 2713–2717.
Charles Cao, Y. W., Jin, R., & Mirkin, C. A. (2002). Nanoparticles with Raman spectroscopic fingerprints for DNA and RNA detection. Science, 297(5586), 1536–1540.
Taton, T. A., Mirkin, C. A., & Letsinger, R. L. (2000). Scanometric DNA array detection with nanoparticle probes. Science, 289(5485), 1757–1760.
Elghanian, R., Storhoff, J. J., Mucic, R. C., Letsinger, R. L., & Mirkin, C. A. (1997). Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science, 277(5329), 1078–1081.
Park, S. J., Taton, T. A., & Mirkin, C. A. (2002). Array-based electrical detection of DNA with nanoparticle probes. Science, 295(5559), 1503–1506.
Kamat, P. V. (2002). Photophysical, photochemical and photocatalytic aspects of metal nanoparticles. The Journal of Physical Chemistry B, 106(32), 7729–7744.
Rosi, N. L., Giljohann, D. A., Thaxton, C. S., Lytton-Jean, A. K. R., Han, M. S., & Mirkin, C. A. (2006). Oligonucleotide-modified gold nanoparticles for intracellular gene regulation. Science, 312(5776), 1027–1030.
Nie, S., & Emory, S. R. (1997). Probing single molecules and single nanoparticles by surface-enhanced raman scattering. Science, 275(5303), 1102–1106.
Oberdorster, G., Oberdorster, E., & Oberdorster, J. (2005). Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environmental Health Perspectives, 113(7), 823–839.
Warheit, D. B., Webb, T. R., Reed, K. L., Frerichs, S., & Sayes, C. M. (2007). Pulmonary toxicity study in rats with three forms of ultrafine-TiO2 particles: differential responses related to surface properties. Toxicology, 230(1), 90–104.
Limbach, L. K., Li, Y., Grass, R. N., Brunner, T. J., Hintermann, M. A., Muller, M., Gunther, D., & Stark, W. J. (2005). Oxide nanoparticle uptake in human lung fibroblasts: effects of particle size, agglomeration, and diffusion at low concentrations. Environmental Science & Technology, 39(23), 9370–9376.
Korsvik, C., Patil, S., Seal, S., & Self, W. T. (2007). Superoxide dismutase mimetic properties exhibited by vacancy engineered ceria nanoparticles. Chemical Communications, 14(10), 1056–1058.
Heckert, E. G., Karakoti, A. S., Seal, S., & Self, W. T. (2008). The role of cerium redox state in the SOD mimetic activity of nanoceria. Biomaterials, 29(18), 2705–2709.
Robinson, R. D., Spanier, J. E., Zhang, F., Chan, S.-W., & Herman, I. P. (2002). Visible thermal emission from sub-band-gap laser excited cerium dioxide particles. Journal of Applied Physics, 92(4), 1936–1941.
Deshpande, S., Patil, S., Kuchibhatla, S. V. N. T., & Seal, S. (2005). Size dependency variation in lattice parameter and valency states in nanocrystalline cerium oxide. Applied Physics Letters, 87(13), 133113:1–3.
Esch, F., Fabris, S., Zhou, L., Montini, T., Africh, C., Fornasiero, P., Comelli, G., & Rosei, R. (2005). Electron localization determines defect formation on ceria substrates. Science, 309(5735), 752–755.
Chen, J., Patil, S., Seal, S., & McGinnis, J. F. (2006). Rare earth nanoparticles prevent retinal degeneration induced by intracellular peroxides. Nature Nanotechnology, 1(2), 142–150.
Karakoti, A. S., Singh, S., Kumar, A., Malinska, M., Kuchibhatla, S. V. N. T., Wozniak, K., Self, W. T., & Seal, S. (2009). PEGylated nanoceria as radical scavenger with tunable redox chemistry. Journal of the American Chemical Society, 131(40), 14144–14145.
Pirmohamed, T., Dowding, J. M., Singh, S., Wasserman, B., Heckert, E., Karakoti, A. S., King, J. E., Seal, S., & Self, W. T. (2010). Nanoceria exhibit redox state-dependent catalase mimetic activity. Chemical Communications, 46(16), 2736–2738.
Celardo, I., Pedersen, J. Z., Traversa, E., & Ghibelli, L. (2011). Pharmacological potential of cerium oxide nanoparticles. Nanoscale, 3(4), 1411–1420.
Perez, J. M., Asati, A., Nath, S., & Kaittanis, C. (2008). Synthesis of biocompatible dextran-coated nanoceria with pH-dependent antioxidant properties. Small, 4(5), 552–556.
Hirst, S. M., Karakoti, A., Singh, S., Self, W., Tyler, R., Seal, S., & Reilly, C. M. (2013). Bio-distribution and in vivo antioxidant effects of cerium oxide nanoparticles in mice. Environmental Toxicology, 28(2), 107–118.
Cai, X., Seal, S., & McGinnis, J. F. (2014). Sustained inhibition of neovascularization in vldlr −/− mice following intravitreal injection of cerium oxide nanoparticles and the role of the ASK1-P38/JNK-NF-κB pathway. Biomaterials, 35(1), 249–258.
Das, S., Singh, S., Dowding, J. M., Oommen, S., Kumar, A., Sayle, T. X. T., Saraf, S., Patra, C. R., Vlahakis, N. E., Sayle, D. C., Self, W. T., & Seal, S. (2012). The induction of angiogenesis by cerium oxide nanoparticles through the modulation of oxygen in intracellular environments. Biomaterials, 33(31), 7746–7755.
Zheng, F., Jin, Q., & Chen, S. W. (2004). Ceria nanoparticles: size, size distribution, and shape. Journal of Applied Physics, 95(8), 4319–4323.
Czerwinski, F., & Szpunar, J. A. (1997). The nanocrystalline ceria sol-gel coatings for high temperature applications. Journal of Sol-Gel Science and Technology, 9(1), 103–114.
Wang, Z. L., & Feng, X. (2003). Polyhedral shapes of CeO2 nanoparticles. The Journal of Physical Chemistry B, 107(49), 13563–13566.
Zhitomirsky, I., & Petric, A. (2001). Electrochemical deposition of ceria and doped ceria Films. Ceramics International, 27(2), 149–155.
Satyamurthy, S., Leonard, K. J., Dabestani, R. T., & Paranthaman, M. P. (2005). Reverse micellar synthesis of cerium oxide nanoparticles. Nanotechnology, 16(9), 1960–1964.
Sakthivel, T., Das, S., Kumar, A., Reid, D. L., Gupta, A., Sayle, D. C., & Seal, S. (2013). Morphological phase diagram of biocatalytically active ceria nanostructures as a function of processing variables and their properties. ChemPlusChem, 78(12), 1446–1455.
Araujo, V. D., Avansi, W., de Carvalho, H. B., Moreira, M. L., Longo, E., Ribeiro, C., & Bernardi, M. I. B. (2012). CeO2 nanoparticles synthesized by a microwave assisted hydrotrhermal method:evolution from nanosphere to nanorod. Crystal Engineering Communications, 14(3), 1150–1154.
Das, K., Samanta, L., & Chainy, G. B. N. (2000). A modified spectrophotometric assay of superoxide dismutase using nitrite formations by superoxide radicals. Indian Journal of Biochemistry and Biophysics, 37(3), 201–204.
Garrat, D. C. (1964). “In: The quantitative analysis of drug” Chapman and Hall. Japan, 3, 456–458.
Samanta, L., & Chainy, G. B. N. (1997). Comparison of hexa chloro cyclohexane-induced oxidative stress in the testis of immature and adult rats—general considerations. Comparative Biochemistry and Physiology, 118C(3), 319–327.
Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95(2), 351–358.
Levine, R. L., Garland, D., Oliver, C. N., Amici, A., Climent, I., Lenz, A. G., Ahn, B. W., Shaltiel, S., & Stadtman, E. R. (1990). Methods in Enzymology, 186, 464–478.
Fridovich, I. (1986). Oxygen Radicals, Hydrogen peroxide and oxygen toxicity. Free Radicals and Biology. Pryor, W. A. New York, AcademicPress., 1, 239-246.
Denicola, A., Souza, J. M., & Radi, R. (1998). Diffusion of peroxynitrite across erythrocyte membranes. Proceedings of the National Academy of Science, 95(7), 3566–3571.
Marla, S. S., Lee, J., & Groves, J. T. (1997). Peroxynitrite rapidly permeates phospholipid membranes. Proceedings of the National Academy of Science, 94(26), 14243–14248.
Moncada, S., Palmer, R. M., & Higgs, E. A. (1991). Nitric oxide: physiology, pathophysiology, and pharmacology. Pharma Review, 43(2), 109–142.
Tylor, B. S., Kion, Y. M., Wang, Q. I., Sharpio, R. A., Billiar, T. R., & Geller, D. A. (1997). Nitric oxide down-regulates hepatocyte-inducible nitric oxide synthase gene expression. Archives of Surgery, 132, 1177–1183.
Dalle-Donne, I., Rossi, R., Giustarini, D., Milzani, A., & Colombo, R. (2003). Protein carbonyl groups as biomarkers of oxidative stress. Clinica Chimica Acta, 329(1-2), 23–38.
Madian, A. G., & Regnier, F. E. (2010). Proteomic identification of carbonylated proteins and their oxidation sites. Journal of Proteome Research, 9(8), 3766–3780.
Rao, R. S. P., & Moller, I. M. (2011). Pattern of occurrence and occupancy of carbonylation sites in proteins. Proteomics, 11(21), 4166–4173.
Stadtaman, E. R., & Levine, R. L. (2000). Protein oxidation. Annals of the New York Academy of Sciences, 899, 191–208.
Pignatelli, B., Li, C. Q., Boffetta, P., Chen, Q., Ahrens, W., Nyberg, F., Mukeria, A., Bruske-Hohlfeld, I., Fortes, C., Constantinescu, V., Ischiropoulos, H., & Ohshima, H. (2001). Nitrated and oxidized plasma proteins in smokers and lung cancer patients. Cancer Research, 61(2), 778–784.
Yilmaz, I. A., Akçay, T., Cakatay, U., Telci, A., Ataus, S., & Yalçin, V. (2003). Relation between bladder cancer and protein oxidation. International Urology and Nephrology, 35(3), 345–350.
Rossner, P., Jr., Terry, M. B., Gammon, M. D., Agrawal, M., Zhang, F. F., Ferris, J. S., Teitelbaum, S. L., Eng, S. M., Gaudet, M. M., Neugut, A. I., & Santella, R. M. (2007). Plasma protein carbonyl levels and breast cancer risk. Journal of Cellular and Molecular Medicine, 11(5), 1138–1148.
Dalle-Donne, I., Aldini, G., Carini, M., Colombo, R., Rossi, R., & Milzani, A. (2006). Protein carbonylation, cellular dysfunction and disease progression. Journal of Cellular and Molecular Medicine, 10(2), 389–406.
Lin, W., Huang, Y. W., Zhou, X. D., & Ma, Y. (2006). Toxicity of cerium oxide nanoparticles in human lung cancer cells. International Journal of Toxicology, 25(6), 451–457.
Babu, S., Velez, A., Wozniak, K., Szydlowska, J., & Seal, S. (2007). Electron paramagnetic study on radical scavenging properties of ceria nanoparticles. Chemical Physics Letters, 442(4-6), 405–408.
Dowding, J. M., Dosani, T., Kumar, A., Seal, S., & Self, W. T. (2012). Cerium oxide nanoparticles scavenge nitric oxide radical (NO●). Chemical Communications, 48, 4896–4898.
Acknowledgments
Purnendu Parhi acknowledges Department of Science and Technology (DST), project no. SR/FT/CS-91/2011 for funding. Siba Soren acknowledges Rajiv Gandhi Fellowship sponsored by UGC, New Delhi, for fellowship. Author acknowledges the Department of Chemistry and Department of Zoology Ravenshaw University for support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Soren, S., Jena, S.R., Samanta, L. et al. Antioxidant Potential and Toxicity Study of the Cerium Oxide Nanoparticles Synthesized by Microwave-Mediated Synthesis. Appl Biochem Biotechnol 177, 148–161 (2015). https://doi.org/10.1007/s12010-015-1734-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1734-8