Abstract
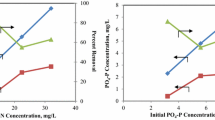
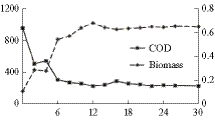
Simultaneous nitrate (N), phosphate (P), and COD removal was investigated in photobioreactors containing both algae and bacteria. The reactors were operated in the semi-batch mode with a hydraulic retention time of 2 days. Reactors were operated in two phases, (1) with 33 % biomass recycle and (2) with no biomass recycle. In both phases, more than 90 % of N and P and 80 % of COD present in synthetic wastewaters with initial N and P concentrations of up to 110 and 25 mg/L, respectively, and initial COD of 45 mg/L could be removed. Biomass growth in reactors did not increase with the increase in initial N and P concentration in either phase. However, biomass growth was slightly more in reactors operated with no biomass recycle. In both phases, N and P uptake was greater in reactors with greater initial N and P concentrations. Also in all cases, N and P uptake in the reactors was far in excess of the stoichiometric requirements for the observed biomass growth. This “luxury uptake” of nitrogen and phosphorus by biomass was responsible for excellent nitrogen and phosphorus removal as observed. However, based on the results of this study, no advantage of biomass recycling could be demonstrated.

Similar content being viewed by others
References
Arceivala, S. J., & Asolekar, S. R. (2007). 3rd Edition, 2007. Tata McGraw-Hill Publishing Company: Limited. Wastewater Treatment for Pollution Control and Reuse.
Olguin, E. J. (2003). Phycoremediation: key issues for cost-effective nutrient removal processes. Biotechnol Adv, 22, 81–91.
Campbell M. N. (2008). Biodiesel: Algae as a renewable source for liquid fuel. Guelph Engineering Journal, (1)2-7, 2008
Vasumathi, K. K., Premalatha, M., & Subramanian, P. (2012). Parameters influencing the design of photobioreactor for the growth of microalgae. Renew Sust Energ Rev, 16, 5442–5450.
Godos, I., Gonzalez, C., Becares, E., Encina, P. A. G., & Munoz, R. (2009). Simultaneous nutrients and carbon removal during pretreated swine slurry degradation in a tubular biofilm photobioreactor. Appl Microbiol Biotechnol, 82, 187–194.
Salces, B. M., Gonzalez, M. C. G., & Fernandez, C. G. (2010). Performance comparison of two photobioreactors configurations (open and closed to the atmosphere) treating anaerobically degraded swine slurry. Bioresour Technol, 101, 5144–5149.
Mallick, N. (2002). Biotechnological potential of immobilized algae for wastewater N, P and metal removal: a review. Biometals, 15, 377–390.
Carvalho, A. P., & Malcata, F. X. (2001). Transfer of carbon dioxide within cultures of microalgae: plain bubbling versus hollow-fiber modules. Biotechnol Prog, 17, 265–272.
Ozbek, B., & Gayik, S. (2001). The studies on the oxygen mass transfer coefficient in a bioreactor. Process Biochem, 36, 729–741.
Singh, R. N., & Sharma, S. (2012). Development of suitable photobioreactor for algae production—a review. Renew Sust Energ Rev, 16, 2347–2353.
Bratbak, G., & Thingstad, T. F. (1985). Phytoplankton-bacteria interactions: an apparent paradox? Analysis of a model system with both; competition and commensalism. Mar Ecol Prog Ser, 25, 23–30.
Munoz, R., Jacinto, M., Guieysse, B., & Mattiasson, B. (2005). Combined carbon and nitrogen removal from acetonitrile using algal-bacterial bioreactors. Appl Microbiol Biotechnol, 67, 699–707.
Shekhar, M. (2011). Microfiltration of algal suspensions. IIT Kanpur India: Master’s dissertation.
Zamfirescu, C., Naterer, G. F., & Dincer, I. (2012). Actinometry measurements of photon flux to supra-molecular complexes for photo-catalytic hydrogen generation. 19th World Dihydrogen Energy Conference 2012, 3–7 June, Toronto, Canada
Ogbonna, J. C., & Tanaka, H. (2000). Light requirement and photosynthetic cell cultivation—development of processes for efficient light utilization in photobioreactors. J Appl Phycol, 12(3–5), 207–218.
Termini, I. D., Prassone, A., Cattaneo, C., & Rovatti, M. (2011). On the nitrogen and phosphorous removal in algal photobioreactors. Ecol Eng, 37, 976–980.
Shriwastav, A., Sudarsan, G., Bose, P., & Tare, V. (2010). Modification of Winkler’s method for determination of dissolved oxygen concentration in small sample volumes. Anal Methods, 10, 1618–1622.
Eaton, A. D., Clesceri, L. S., & Greenberg, A. E. (Eds.) Standard Methods for the Examination of water and waste water. 19th ed., American Public Health Association, Washington DC, APHA, AWWA and WEF.
Sartory, D. P., & Grobbelaar, J. U. (1984). Extraction of chlorophyll a from freshwater phytoplankton for spectrophotometric analysis. Hydrobiologia, 114, 177–187.
Porra, R. J., Thompson, W. A., & Kriedemann, P. E. (1989). Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta, 975, 384–394.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Anal Chem, 28, 350–356.
Maxwell, D. P., Falk, S., Trick, C. G., & Huner, N. P. A. (1994). Growth at low temperature mimics high-light acclimation in chlorella vulgaris. Plant Physiol, 105, 535–543.
Vitova, M., Bisova, K., Hlavova, M., Kawano, S., Zachleder, V., & Cizkova, M. (2011). Chlamydomonas reinhardtii: duration of its cell cycle and phases at growth rates affected by temperature. Planta, 234, 599–608.
Ambrose, R. B., Martin, J. L., & Wool, T. A. (2006). WASP7 Benthic algae—model theory and user’s guide. USA: Environmental Protection Agency.
Chapra, S. C., Pelletier, G. J. & Tao, H. (2007). QUAL2K: A modeling framework for simulating river and stream water quality, Version 2.07. Environmental Protection Agency, USA.
Bich, N. N., Yaziz, M. I., & Bakti, N. A. K. (1999). Combination of chlorella vulgaris and eichhornia crassipes for wastewater nitrogen removal. Water Res, 33(10), 2357–2362.
Aslan, S., & Kapdan, I. K. (2006). Batch kinetics of nitrogen and phosphorous removal from synthetic wastewater by algae. Ecol Eng, 28, 64–70.
Mohammed, T. A., Birima, A. H., Noor, M. J. M. M., Muyibi, S. A., & Idris, A. (2008). Evaluation of using membrane bioreactor for treating municipal wastewater at different operating conditions. Desalination, 221, 502–510.
Wang, L., Min, M., Li, Y., Chen, P., Chen, Y., Liu, Y., Wang, Y., & Ruan, R. (2010). Cultication of green algae chlorella sp. In different wastewaters from municipal wastewater treatment plant. Appl Biochem Biotechnol, 162, 1174–1186.
Su, Y., Mennerich, A., & Urban, B. (2011). Municipal wastewater treatment and biomass accumulation with a wastewater-born and settleable algal-bacterial culture. Water Res, 45, 3351–3358.
Akerstrom, A. M., Mortensen, L. M., Rusten, B., & Gislerod, H. R. (2014). Biomass production and nutrient removal by chlorella sp. as affected by sludge liquor concentration. J Environ Manag, 144, 118–124.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashok, V., Shriwastav, A. & Bose, P. Nutrient Removal Using Algal-Bacterial Mixed Culture. Appl Biochem Biotechnol 174, 2827–2838 (2014). https://doi.org/10.1007/s12010-014-1229-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1229-z