Abstract
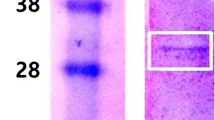
A direct-acting fibrinolytic serine protease named undariase possessing anticoagulant and antiplatelet properties was purified from Undaria pinnatifida. Undariase showed a molecular weight of 50 kDa by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and mass spectrometry. It displayed a strong fibrin zymogram lysis band corresponding to the same molecular mass. The N-terminal sequence of undariase, LTATTCEELAAAPTD, does not match with any known fibrinolytic enzyme. The enzyme was stable and active at high temperatures (35–70 °C). The fibrinolytic activity of undariase was strongly inhibited by phenylmethylsulfonyl fluoride (PMSF) and 4-(amidinophenyl) methanesulfonyl fluoride (APMSF). The K m and V max values for substrate S-2251 were determined as 6.15 mM and 90.91 mM/min/ml, respectively. Undariase resulted in clot lysis by directly cleaving α and β chains of fibrin. Similarly, it preferentially acted on the Aα chain of fibrinogen followed by cleavage of the Bβ chain. It significantly prolonged the PFA-100 closure times of citrated whole human blood. In addition, undariase delayed the coagulation time and increased activated partial thromboplastin time (APTT), prothrombin time (PT), and thrombin time (TT). Undariase exerted a significant protective effect against collagen plus epinephrine-induced pulmonary thromboembolism in mice. It prevented carrageenan-induced thrombus formation in the tail of mice. It also resulted in prolongation of APTT ex vivo. In conclusion, these results suggested a therapeutic potential of undariase for thrombosis.











Similar content being viewed by others
References
Collen, D., & Lijnen, H. R. (1992). Annals of the New York Academy of Sciences, 4, 259–271.
Thorsen, S. (1992). Annals of the New York Academy of Sciences, 4, 52–63.
Collen, D. (1980). Journal of Clinical Pathology. Supplement (Royal College of Pathologists), 14, 24–30.
Collen, D., & Lijnen, H. R. (1991). Blood, 78, 3114–3124.
Mackman, N. (2008). Nature, 451, 914–918.
Fitton, J. H. (2003). Alternative and Complementary Therapies, 9, 29–33.
Moon, S., & Kim, J. (1999). International Journal of Food Sciences and Nutrition, 50, 165–171.
Khan, M. N., Lee, M. C., Kang, J. Y., Park, N. G., Fujii, H., & Hong, Y. K. (2008). Phytotherapy Research, 22, 634–639.
Yubin, J., & Guangmei, Z. (1998). Pharmacological action and application of available antitumor composition of traditional Chinese medicine. China: Heilongjiang.
Hoppe, H. A., Levring, T., & Tanka, Y., (eds.) (1979). Marine algae in pharmaceutical science. Berlin & New York.
Oh, Y. S., Lee, I. K., & Boo, S. M. (1990). Korean Journal of Phycology, 5, 57–71.
Ikeda, K., Kitamura, A., Machida, H., Watanabe, M., Negishi, H., Hiraoka, J., et al. (2003). Clinical and Experimental Pharmacology and Physiology, 30, 44–48.
Hosokawa, M., Kudo, M., Maeda, H., Kohno, H., Tanaka, T., & Miyashita, K. (2004). Biochimica et Biophysica Acta, 1675, 113–119.
Maeda, H., Hosokawa, M., Sashima, T., Funayama, K., & Miyashita, K. (2005). Biochemical and Biophysical Research Communications, 332, 392–397.
Min, S. K., Kwon, O. C., Lee, S., Park, K. H., & Kim, J. K. (2012). Phytotherapy Research, 26, 752–757.
Liu, F., Wang, J., Chang, A. K., Liu, B., Yang, L., Li, Q., et al. (2012). Phytomedicine, 19, 797–803.
Teas, J., & Irhimeh, M. R. (2012). Journal of Applied Phycology, 24, 575–582.
Vishchuk, O. S., Ermakova, S. P., & Zvyagintseva, T. N. (2011). Carbohydrate Research, 346, 2769–2776.
Kim, K. J., & Lee, B. Y. (2012). Nutrition Research, 32, 439–447.
Kim, K. J., Yoon, K. Y., & Lee, B. Y. (2012). Fitoterapia, 83, 1628–1635.
Suetsuna, K., Maekawa, K., & Chen, J. R. (2004). Journal of Nutritional Biochemistry, 15, 267–272.
Wijesinghea, W. A. J. P., & Jeon, Y. J. (2012). Carbohydrate Polymers, 88, 13–20.
Min, S. K., Han, S. M., Kim, H. T., Kwon, O. C., Lee, S., & Kim, J. K. (2012). Blood Coagulation and Fibrinolysis, 23, 359–366.
Matsubara, K., Sumi, H., Hori, K., & Miyazawa, K. (1998). Comparative Biochemistry and Physiology, 119, 177–181.
Matsubara, K., Hori, K., Matsuura, Y., & Miyazawa, K. (1999). Phytochemistry, 52, 993–999.
Matsubara, K., Hori, K., Matsuura, Y., & Miyazawa, K. (2000). Comparative Biochemistry and Physiology, 125, 137–143.
Kim, D. W., Sapkota, K., Choi, J. H., Kim, Y. S., Kim, S., & Kim, S. J. (2013). Process Biochemistry, 48, 340–350.
Choi, J. H., Sapkota, K., Park, S. E., Kim, S., & Kim, S. J. (2013). Biochimie, 95, 1266–1277.
Kim, S. H., Choi, N. S., & Lee, W. Y. (1998). Analytical Biochemistry, 263, 115–116.
Hummel, B. C. W., Schor, J. M., Buck, F. F., Boggiano, E., & De Renzo, E. C. (1965). Analytical Biochemistry, 11, 532–547.
Gaspar, A. R., Crause, J. C., & Neitz, A. W. (1995). Experimental and Applied Acarology, 19, 117–127.
Astrup, T., & Mullertz, S. (1952). Archives of Biochemistry and Biophysics, 40, 346–351.
Datta, G., Dong, A., Witt, J., & Tu, A. T. (1995). Archives of Biochemistry and Biophysics, 317, 365–373.
Gremski, L. H., Chaim, O. M., Paludo, K. S., Sade, Y. B., Otuki, M. F., Richardson, M., et al. (2007). Toxicon, 50, 120–134.
Hagimori, M., Kamiya, S., Yamaguchi, Y., & Arakawa, M. (2009). Pharmacological Research, 60, 320–323.
DiMinno, G., & Silver, M. J. (1983). Journal of Pharmacology and Experimental Therapeutics, 225, 57–60.
Tucker, E. I., Verbout, N. G., Leung, P. Y., Hurst, S., McCarty, O. J., Gailani, D., et al. (2012). Blood, 119, 4762–4768.
Lind, S. E. (1991). Blood, 77, 2547–2552.
Shanmugam, S., & Mody, K. H. (2000). Current Science, 79, 1672–1683.
McLellan, D. S., & Jurd, K. M. (1992). Blood Coagulation and Fibrinolysis, 3, 69–77.
Marder, V. J., & Novokhatny, V. (2010). Journal of Thrombosis and Haemostasis, 8, 433–444.
Vandooren, J., Geurts, N., Martens, E., Van den Steen, P. E., & Opdenakker, G. (2013). Nature Methods, 10, 211–220.
Henry, B. L., Monien, B. H., Bock, P. E., & Desai, U. R. (2007). The Journal of Biological Chemistry, 282, 31891–31899.
Heemskerk, J. W., Bevers, E. M., & Lindhout, T. (2002). Thrombosis and Haemostasis, 88, 186–193.
Chan, K. C., Yin, M. C., & Chao, W. J. (2007). Food and Chemical Toxicology, 45, 502–507.
Markland, F. S. (1998). Toxicon, 36, 1749–1800.
Song, J., Xu, X., Zhang, Y., Guo, M., Yan, X., Wang, S., et al. (2013). Biochimie, 95, 709–718.
Hamza, L., Gargioli, C., Castelli, S., Rufini, S., & Laraba-Djebari, F. (2010). Biochimie, 92, 797–805.
Mukherjee, A. K., Rai, S. K., Thakur, R., Chattopadhyay, P., & Kar, S. K. (2012). Biochimie, 94, 1300–1308.
Rooney, M. M., Farrell, D. H., van Hemel, B. M., de Groot, P. G., & Lord, S. T. (1998). Blood, 92, 2374–2381.
Andrieux, A., Hudry-Clergeon, G., Ryckewaert, J. J., Chapel, A., Ginsberg, M. H., Plow, E. F., et al. (1989). The Journal of Biological Chemistry, 264, 9258–9265.
Levi, M., Keller, T. T., van Gorp, E., & ten Cate, H. (2003). Cardiovascular Research, 60, 26–39.
Levi, M., van der Poll, T., & Büller, H. R. (2004). Circulation, 109, 2698–2704.
Lynch, J., & Shariat-Madar, Z. (2012). Journal of Clinical Toxicology, 2, doi:10.4172/2161-0495.1000e105.
He, J., Chen, S., & Gu, J. (2007). FEBS Letters, 581, 2965–2973.
Yan, F., Yan, J., Sun, W., Yao, L., Wang, J., Qi, Y., et al. (2009). Journal of Thrombosis and Thrombolysis, 28, 444–448.
Arslan, R., Bor, Z., Bektas, N., Meriçli, A. H., & Ozturk, Y. (2011). Thrombosis Research, 127, 210–213.
Yang, H., Wang, Y., Xiao, Y., Wang, Y., Wu, J., Liu, C., et al. (2011). PLoS One, 6, e17519.
Simkhada, J. R., Cho, S. S., Mander, P., Choi, Y. H., & Yoo, J. C. (2012). Thrombosis Research, 129, 176–182.
Bekemeier, H., Hirschelmann, R., & Giessler, A. J. (1985). Agents and Actions, 16, 446–451.
Elsayed, Y. A., Nakagawa, K., Ichikawa, K., Ohkawara, S., & Sueishi, K. (1995). Pathobiology, 63, 328–340.
Davies, M. J., & Thomas, A. C. (1985). British Heart Journal, 53, 363–373.
Jin, Y. R., Hwang, K. A., Cho, M. R., Kim, S. Y., Kim, J. H., Ryu, C. K., et al. (2004). Vascular Pharmacology, 41, 35–41.
Canobbio, I., Cipolla, L., Consonni, A., Momi, S., Guidetti, G., Oliviero, B., et al. (2013). Blood, 121, 648–657.
Kim, M. S., & Lee, K. A. (2006). Journal of Ethnopharmacology, 105, 342–345.
Fan, J., Zhang, Y., Chang, X., Zhang, B., Jiang, D., Saito, M., et al. (2009). Journal of Agricultural and Food Chemistry, 57, 8683–8687.
Klafke, J. Z., Arnoldi da Silva, M., Fortes Rossato, M., Trevisan, G., Banderó Walker, C. I., Martins Leal, C. A., et al. (2012) Evidence-Based Complementary and Alternative Medicine, 954748. doi:10.1155/2012/954748.
Mekhfi, H., Belmekki, F., Ziyyat, A., Legssyer, A., Bnouham, M., & Aziz, M. (2012). Nutrition, 28, 937–941. doi:10.1016/j.nut.2011.11.032.
Colognato, R., Slupsky, J. R., Jendrach, M., Burysek, L., Syrovets, T., & Simmet, T. (2003). Blood, 102, 2645–2652.
Acknowledgments
This research was supported by the Technology Development Program for Fisheries, Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea.
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Choi, JH., Sapkota, K., Kim, MK. et al. Undariase, a Direct-Acting Fibrin(ogen)olytic Enzyme from Undaria pinnatifida, Inhibits Thrombosis In Vivo and Exhibits In Vitro Thrombolytic Properties. Appl Biochem Biotechnol 173, 1985–2004 (2014). https://doi.org/10.1007/s12010-014-0981-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-0981-4