Abstract
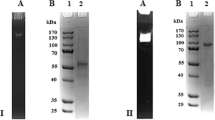
A cinnamyl alcohol dehydrogenase (CAD) from the secondary xylem of Leucaena leucocephala has been purified to homogeneity through successive steps of ammonium sulfate fractionation, DEAE cellulose, Sephadex G-75, and Blue Sepharose CL-6B affinity column chromatographies. CAD was purified to 514.2 folds with overall recovery of 13 % and specific activity of 812. 5 nkat/mg. Native and subunit molecular masses of the purified enzyme were found to be ∼76 and ∼38 kDa, respectively, suggesting it to be a homodimer. The enzyme exhibited highest catalytic efficiency (Kcat/Km 3.75 μM−1 s−1) with cinnamyl aldehyde among all the substrates investigated. The pH and temperature optima of the purified CAD were pH 8.8 and 40 °C, respectively. The enzyme activity was enhanced in the presence of 2.0 mM Mg2+, while Zn2+ at the same concentration exerted an inhibitory effect. The inclusion of 2.0 mM EDTA in the assay system activated the enzyme. The enzyme was inhibited with caffeic acid and ferulic acid in a concentration-dependent manner, while no inhibition was observed with salicylic acid. Peptide mass analysis of the purified CAD by MALDI-TOF showed a significant homology to alcohol dehydrogenases of MDR superfamily.



Similar content being viewed by others
References
Kaur, H., Shaker, K., Heinzel, N., Ralph, J., Galis, I., & Baldwin, I. T. (2012). Plant Physiology, 159, 1545–1570.
Mansell, R. L., Gross, G. G., Stoeckigt, J., Franke, H., & Zenk, M. H. (1974). Phytochemistry, 13, 2427–2436.
Saballos, A., Ejeta, G., Sanchez, E., Kang, C., & Vermerris, W. (2009). Genetics, 181, 783–795.
Boudet, A. (2000). Plant Physiology and Biochemistry, 38, 81–96.
Weng, J. K., & Chapple, C. (2010). New Phytologist, 187, 273–285.
Wyrambik, D., & Grisebach, H. (1975). European Journal of Biochemistry, 59, 9–15.
Halpin, C., Knight, M. E., Grima-Pettenati, J., Goffner, D., Boudet, A., & Schuch, W. (1992). Plant Physiology, 98, 12–16.
Hawkins, S. W., & Boudet, A. M. (1994). Plant Physiology, 104, 75–84.
Pillonel, C., Hunziker, P., & Binder, A. (1992). Journal of Experimental Botany, 43, 299–305.
O’Malley, D. M., Porter, S., & Sederoff, R. R. (1992). Plant Physiology, 98, 1364–1371.
Mee, B., Kelleher, D., Frias, J., Malone, R., Tipton, K. F., Henehan, G. T. M., & Windle, H. J. (2005). FEBS Journal, 272, 1255–1264.
Kutsuki, H., Shimada, M., & Higuchi, T. (1982). Phytochemistry, 21, 19–23.
Li, L., Cheng, X. F., Leshkevich, J., Umezawa, T., Harding, S. A., & Chiang, V. L. (2001). Plant Cell, 13, 1567–1585.
Luderitz, T., & Grisebach, H. (1981). European Journal of Biochemistry, 119, 115–124.
Boerjan, W., Ralph, J., & Baucher, M. (2003). Annual Review of Plant Biology, 54, 519–546.
Kim, S. J., Kim, M. R., Bedgar, D. L., Moinuddin, S. G. A., Cardenas, C. L., Davin, L. B., Kang, C., & Lewis, N. G. (2004). Proceedings of the National Academy of Science, 101, 1455–1460.
Valencia, E., Larroy, C., Ochoa, W. F., Parés, X., Fita, I., & Biosca, J. A. (2004). Journal of Molecular Biology, 341, 1049–1062.
Banfield, M. J., Salvucci, M. E., Baker, E. N., & Smith, C. A. (2001). Journal of Molecular Biology, 306, 239–250.
Rastogi, S. & Dwivedi, U.N. (2003), In: Plant genetic engineering: applications and limitations. (Singh R. P. & Jaiwal, P. K., Eds.) Sci. Tech. Pubs., Houston, U.S.A., pp. 317-360.
Rastogi, S., & Dwivedi, U. N. (2006). Biotechnology Progress, 22, 609–616.
Campbell, M. M., & Ellis, B. E. (1992). Planta, 186, 409–417.
Bradford, M. M. (1976). Analytical Biochemistry, 72, 248–254.
Loulakakis, & Roubelakis-Angelakis (1996). Physiologia Plantarum, 96, 29–35.
Barakate, A., Stephens, J., Goldie, A., Hunter, W. N., Marshall, D., Hancock, R. D., Lapierre, C., Morreel, K., Boerjan, W., & Halpin, C. (2011). Plant Cell, 23, 4492–4506.
Pandey, B., Pandey, V. P., & Dwivedi, U. N. (2011). Protein Expression and Purification, 79, 197–203.
Lee, D., Ellard, M., Wanner, L. A., Davis, K. R., & Douglas, C. J. (1995). Plant Molecular Biology, 28, 871–884.
Acknowledgments
The financial support from Council of Scientific and Industrial Research, New Delhi, is gratefully acknowledged. We are also thankful, for the assistance, to Department of Higher Education, Government of Uttar Pradesh, under the Center of Excellence in Bioinformatics program; Department of Biotechnology (DBT), Government of India, New Delhi, under the BIF program; and Department of Science and Technology under Promotion of University Research and Scientific Excellence (DST-PURSE) program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pandey, B., Pandey, V.P., Shasany, A.K. et al. Purification and Characterization of a Zinc-Dependent Cinnamyl Alcohol Dehydrogenase from Leucaena leucocephala, a Tree Legume. Appl Biochem Biotechnol 172, 3414–3423 (2014). https://doi.org/10.1007/s12010-014-0776-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-0776-7