Abstract
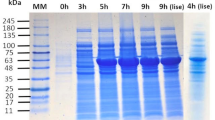
A β-galactosidase gene from Aspergillus oryzae was engineered utilizing codon usage optimization to be constitutively and highly expressed in the Pichia pastoris SMD1168H strain in a high-cell-density fermentation. After fermentation for 96 h in a 50-L fermentor using glucose and glycerol as combined carbon sources, the recombinant enzyme in the culture supernatant had an activity of 4,239.07 U mL−1 with o-nitrophenyl-β-d-galactopyranoside as the substrate, and produced a total of extracellular protein content of 7.267 g L−1 in which the target protein (6.24 g L−1) occupied approximately 86 %. The recombinant β-galactosidase exhibited an excellent lactose hydrolysis ability. With 1,000 U of the enzyme in 100 mL milk, 92.44 % lactose was degraded within 24 h at 60 °C, and the enzyme could also accomplish the hydrolysis at low temperatures of 37, 25, and 10 °C. Thus, this engineered strain had significantly higher fermentation level of A. oryzae lactase than that before optimization and the β-galactosidase may have a good application potential in whey and milk industries.






Similar content being viewed by others
References
Zadow, J. G. (1984). Journal of Dairy Science, 67, 2654–2679.
Adam, A. C., Rubio-Texeria, M., & Polaina, J. (2004). Critical Reviews in Food Science and Nutrition, 44, 553–557.
Husain, Q. (2010). Critical Reviews in Biotechnology, 30, 41–62.
Lifran, E. V., Hourigan, J. A., Sleigh, R. W., & Johnson, R. L. (2000). Food Australia, 52, 120–125.
Jarvis, J. K., & Miller, G. D. (2002). Journal of the National Medical Association, 94, 55–66.
Wilt, T. J., Shaukat, A., Shamliyan, T., Taylor, B. C., MacDonald, R., Tacklind, J., et al. (2010). Lactose intolerance and health. No. 192. AHRQ Publication NO. 10-E004. Rockville: Agency for Healthcare Research and Quality.
Shukla, T. P. (1975). CRC Critical Review Food Technology, 5, 325–356.
Siso, M. I. G. (1996). Biresource Technology, 57, 1–11.
Huerta, L. M., Vera, C., Guerrero, C., Wilson, L., & Illanes, A. (2011). Process Biochemistry, 46, 245–252.
Panesar, P. S., Panesar, R., Singh, R. S., Kennedy, J. F., & Kumar, H. (2006). Journal of Chemistry Technology Biotechnology, 81, 530–543.
Kosseva, M. R., Panesar, P. S., Kaur, G., & Kennedy, J. F. (2009). International Journal of Biological Macromolecules, 45, 437–447.
Becerra, M., Prado, S. D., Cerdán, E., & Siso, M. I. G. (2001). Biotechnology Letters, 23, 33–40.
Becerra, M., Prado, S. D., Siso, M. I., & Cerdán, M. E. (2001). Protein Engineering, 14, 379–386.
Berka, R. M., Hucul, G. A., and Ward, M. (1998) United States Patent, No: 5736374.
Katrolia, P., Yan, Q., Jia, H., Li, Y., Jiang, Z., & Song, C. (2011). Journal of Molecular Catalog B-Enzym, 69, 112–119.
Zhang, W., Fan, Y., & Yao, B. (2005). Acta Microbiologica Sinica, 45, 247–252.
User’s guide, D. N. A. M. A. N. (2005). Lynnon Corporation. Canada: Quebec.
Zhao, X., Huo, K. K., & Li, Y. Y. (2000). Shen Wu Gong Cheng Xue Bao, 16, 308–311.
Sheridan, P. P., & Brenchley, J. E. (2000). Applied and Environmental Microbiology, 66, 2438–2444.
Bendtsen, J. D., Nielsen, H., Heijne, G., & Brunak, S. (2004). Journal of Molecular Biology, 340, 783–795.
Zhao, S., Huang, J., Zhang, C., Deng, L., Hu, N., & Liang, Y. (2010). Journal of Microbiology BioTechniques, 20, 467–473.
Gustafsson, C., Govindarajan, S., & Minshull, J. (2004). Trends in Biotechnology, 22, 346–353.
Gvritishvili, A. G., Leung, K. W., & Tombran-Tink, J. (2010). PLoS One, 5, e15056.
Hu, H., Gao, J., He, J., Yu, B., Zheng, P., Huang, Z., et al. (2013). PLoS One, 8, e58393.
Huang, H., Yang, P., Luo, H., Tang, H., Shao, N., Yuan, T., et al. (2008). Applied Microbiology and Biotechnology, 78, 95–103.
Jia, H., Fan, G., Yan, Q., Liu, Y., Yan, Y., & Jiang, Z. (2012). Journal of Molecular Catalysis B-Enzym, 78, 72–77.
Acknowledgments
We thank all colleagues for assisting in the preparation of this manuscript. We also thank Dr. Fred Bogott at Austin Medical Center, Austin of Minnesota, and Dr. Joshua Liao at Hormel Institute, Austin of Minnesota, for their English editing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Qianqian Zhao and Fei Liu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 449 kb)
Rights and permissions
About this article
Cite this article
Zhao, Q., Liu, F., Hou, Z. et al. High Level Production of β-Galactosidase Exhibiting Excellent Milk-Lactose Degradation Ability from Aspergillus oryzae by Codon and Fermentation Optimization. Appl Biochem Biotechnol 172, 2787–2799 (2014). https://doi.org/10.1007/s12010-013-0684-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0684-2