Abstract
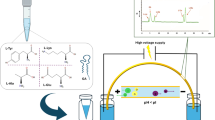
Human saliva quantitative monitoring of clarithromycin (CLA) by chemiluminescence (CL) with flow injection analysis was proposed for the first time, which was based on the quenching effect of CLA on luminol–bovine serum albumin (BSA) CL system with a linear range from 7.5 × 10−4 to 2.0 ng/ml. This proposed approach, offering a maximum sample throughput of 100 h−1, was successfully applied to the quantitative monitoring of CLA levels in human saliva during 24 h after a single oral dose of 250 mg intake, with recoveries of 95.2∼109.0 % and relative standard deviations lower than 6.5 % (N = 7). Results showed that CLA reached maximum concentration of 2.28 ± 0.02 μg/ml at approximately 3 h, and the total elimination ratio was 99.6 % in 24 h. The pharmacokinetic parameters including absorption rate constant (0.058 ± 0.006 h−1), elimination rate constant (0.149 ± 0.009 h−1) and elimination half-life time (4.66 ± 0.08 h) were obtained. A comparison of human saliva and urine monitoring was also given. The mechanism study of BSA–CLA interaction revealed the binding of CLA to BSA is an entropy driven and spontaneous process through hydrophobic interaction, with binding constant K BSA–CLA of 4.78 × 106 l/mol and the number of binding sites n of 0.82 by flow injection–chemiluminescence model. Molecular docking analysis further showed CLA might be in subdomain IIA of BSA, with K BSA–CLA of 6.82 × 105 l/mol and ΔG of −33.28 kJ/mol.






Similar content being viewed by others
References
Herr, A. E., Hatch, A. V., Throckmorton, D. J., Tran, H. M., Brennan, J. S., Giannobile, W. V., & Singh, A. K. (2007). Proceedings of the National Academy of Sciences USA, 104, 5268–5273.
Aps, J. K. M., & Martens, L. C. (2005). Forensic Science International, 150, 119–131.
Swallow, V., Hughes, J., Roberts, D., & Webb, N. J. (2012). Nurse Researcher, 19, 32–37.
Badiyani, B., Kumar, A., & Maru, V. P. (2013). International Journal of Physiology, 1, 52–56.
Liu, J. Y., & Duan, Y. X. (2012). Oral Oncology, 48, 569–577.
Zhang, A. H., Sun, H., & Wang, X. J. (2012). Applied Biochemistry and Biotechnology, 168, 1718–1727.
Cawello, W., Bökens, H., Nickel, B., Andreas, J. O., & Halabi, A. (2013). Epilepsia, 54, 81–88.
Abdel-Rehim, A., & Abdel-Rehim, M. (2013). Biomedical Chromatography, 27, 1188–1191.
Khan, S. A., George, R., Charles, B. G., Taylor, P. J., Heussler, H. S., Cooper, D. M., McGuire, T. M., Pache, D., & Norris, R. L. (2013). Therapeutic Drug Monitoring, 35, 388–395.
Li, X. Z., Yang, T. Y., & Lin, J. X. (2012). Journal of Biomedical Optics, 17, 037003.
Fung, A. O., Damoiseaux, R., Grundeen, S., Panes, J. L., Horton, D. H., Judy, J. W., Moore, T. B. (2012). Malaria Journal, 11, doi:10.1186/1475-2875-11-175. -->
Yoon, A. J., Cheng, B., Philipone, E., Turner, R., & Lamster, I. B. (2012). Journal of Clinical Periodontology, 39, 434–440.
Jhang, C. S., Lee, H., He, Y. S., Liu, J. T., & Lin, C. H. (2012). Electrophoresis, 33, 3073–3078.
Andreou, C., Hoonejani, M. R., Barmi, M. R., Moskovits, M., & Meinhart, C. D. (2013). ACS Nano, 7, 7157–7164.
Lee, H., Jhang, C. S., Liu, J. T., & Lin, C. H. (2012). Journal of Separation Science, 35, 2822–2825.
Gbotosho, G. O., Happi, C. T., Lawal, O., Sijuade, A., Sowunmi, A., Oduola, A. (2012). Malaria Journal, 11, doi:10.1186/1475-2875-11-59.
Henkin, R. I. (2012). Therapeutic Drug Monitoring, 34, 217–221.
Tuyay, J., Coulter, C., Rodrigues, W., & Moore, C. (2012). Drug Testing and Analysis, 4, 395–401.
Hamada, Y., Tokimatsu, I., Mikamo, H., Kimura, M., Seki, M., Takakura, S., Ohmagari, N., Takahashi, Y., Kasahara, K., Matsumoto, K., Okada, K., Igarashi, M., Kobayashi, M., Mochizuki, T., Nishi, Y., Tanigawara, Y., Kimura, T., & Takesue, Y. (2013). Journal of Infection and Chemotherapy, 19, 381–392.
Patsalos, P. N., & Berry, D. J. (2013). Therapeutic Drug Monitoring, 35, 4–29.
Das, R., & Agrawal, Y. K. (2012). Journal of Chromatographic Science, 51, 146–154.
Brown, S. A., Ezejiofor, N. A., & Orisakwe, O. E. (2012). International Current Pharmaceutical Journal, 1, 403–409.
You, J. D., & Song, Z. H. (2013). Instrumentation Science & Technology, 41, 524–534.
Teeninga, N., Guan, Z., Freijer, J., Ruiter, A. F., Ackermans, M. T., Van Kist-van Holthe, J. E., Gelder, T., & Nauta, J. (2013). Therapeutic Drug Monitoring, 35, 485–492.
Noguchi, S., Miura, K., Fujiki, S., & Itai, S. (2012). Acta Crystallographica. Section C, 68, 41–44.
Trevor, A., Katzung, B., Masters, S., Knuidering-Hall, M. (2012). Katzung and Trevor’s Pharmacology Examination and Board Review, 10th ed., Mc Graw-Hill.
Zuckerman, J. M., Qamar, F., & Bono, B. R. (2011). Medical Clinics of North America, 95, 761–791.
Zarogoulidis, P., Papanas, N., Kioumis, I., Chatzaki, E., Maltezos, E., & Zarogoulidis, K. (2012). European Journal of Clinical Pharmacology, 68, 479–503.
Tagliabue, C., Techasaensiri, C., Torres, J. P., Katz, K., Meek, C., Kannan, T. R., Coalson, J. J., Esposito, S., Principi, N., Leff, R., Baseman, J. B., & Hardy, R. D. (2011). Journal of Antimicrobial Chemotherapy, 66, 2323–2329.
Yamazaki, Y., Ushiki, A., Tanabe, Y., Sekiya, K., & Keishi, K. (2012). Journal of Infection and Chemotherapy, 18, 683–688.
Morisset, J., Ptaszynski, S., Al-Mot, S., Dubois, Y., & Bergeron, C. (2012). Annals of Allergy, Asthma & Immunology, 109, 82–83.
Swamy, N., Laurie, S. A., Ruiz-Huidobro, E., & Khan, D. A. (2010). Annals of Allergy, Asthma & Immunology, 105, 489–490.
Couppié, P., Dufour, J., Fior, A., Queuche, F., Clyti, E. E., Guedj, M., Sainte-Marie, D., Vantilke, V., & Nacher, M. (2009). Mycobacterium ulcerans infection in 2008 in French Guyana; HIV immune restoration syndrome; increasing use of two drug therapy rifampicin–clarithromycin treatment, in: WHO Annual Meeting on Buruli Ulcer (p. 17). Geneva: WHO Press.
De Velde, F., Alffenaar, J. W. C., Wessels, A. M. A., Greijdanus, B., & Uges, D. R. (2009). Journal of Chromatography B, 877, 1771–1777.
Shin, J., Pauly, D. F., Johnson, J. A., & Frye, R. F. (2008). Journal of Chromatography B, 871, 130–134.
Gurule, S., Verma, P. R. P., Monif, T., Khuroo, A., & Partani, P. (2008). Journal of Liquid Chromatography & Related Technologies, 31, 2955–2973.
Zhang, X. R., Chen, X. Y., Li, X. Y., & Zhong, D. F. (2004). Journal of Chinese Pharmaceutical Sciences, 13, 166–170.
Oswalda, S., Petersa, J., Vennerb, M., & Siegmund, W. (2011). Journal of Pharmaceutical and Biomedical Analysis, 55, 194–201.
Jiang, Y., Wang, J., Li, H., Wang, Y., & Gu, J. (2007). Journal of Pharmaceutical and Biomedical Analysis, 43, 1460–1464.
Li, W., Jia, H. J., & Zhao, K. (2007). Talanta, 71, 385–390.
Amini, H., & Ahmadiani, A. (2005). Journal of Chromatography B, 81, 193–197.
Lotfipour, F., Valizadeh, H., Hallaj-Nezhadi, S., Milani, M., & Zakeri-Milani, P. (2011). Iranian Journal of Pharmaceutical Research, 9, 27–35.
Bahrami, G., & Mohammadi, B. (2007). Journal of Chromatography B, 850, 417–422.
Zaater, M. F., Tahboub, Y. R., & Ghanem, E. (2012). Journal of Chromatographic Science, 50, 763–768.
Wang, Y. J., Wu, Y. T., Lin, J. Y., Chu, C. H., Huang, H. Y., Wang, Y. C., Chen, J. K., & Yang, C. S. (2012). Journal of Chromatography B, 895–896, 178–181.
Lu, X. M., Chen, L. Y., Wang, D., Liu, J., Wang, Y. J., & Li, F. M. (2008). Chromatographia, 68, 617–622.
Loya, P., & Hamrapurkar, P. D. (2011). JPC-Journal of Planar Chromatography, 24, 534–538.
Luo, M., Chen, X., Zhou, G. H., Xiang, X., Chen, L., Ji, X. H., & He, Z. K. (2012). Chemical Communication, 48, 1126–1128.
Lara, F. J., García-Campaña, A. M., & Aaron, J. J. (2010). Analytica Chimica Acta, 679, 17–30.
Xue, P., Zhang, K., Zhang, Z. J., Li, Y., Liu, F., Sun, Y. J., Zhang, X. M., Song, C. J., Fu, A. H., Jin, B. Q., & Yang, K. (2012). Applied Biochemistry and Biotechnology, 166, 1604–1614.
Li, X., Liu, H. Y., He, X. L., & Song, Z. H. (2010). Applied Biochemistry and Biotechnology, 160, 1065–1073.
Tan, X. J., Song, Z. H., Chen, D. H., & Wang, Z. M. (2011). Spectrochimica Acta Part A, 79, 232–235.
Wang, Z. M., Song, Z. H., & Chen, D. H. (2010). Talanta, 83, 312–319.
Angerer, J., Ewers, U., & Wilhelm, M. (2007). International Journal of Hygiene and Environmental Health, 210, 201–228.
Rockett, J. C., Buck, G. M., Lynch, C. D., & Perreault, S. D. (2004). Environmental Health Perspectives, 112, 94–104.
Van der Edelbroek, P. M., Heijden, J., & Stolk, L. M. (2009). Therapeutic Drug Monitoring, 31, 327–336.
Markway, E. C., & Baker, S. N. (2011). Orthopedics, 34, 877–881.
Reisfield, G. M., Goldberger, B. A., & Bertholf, R. L. (2009). Bioanalysis, 1, 937–952.
Chu, S., Wilson, D. S., Deaton, R. L., Mackenthun, A. V., Eason, C. N., & Cavanaugh, J. H. (1993). The Journal of Clinical Pharmacology, 33, 719–726.
Acknowledgments
The authors gratefully acknowledge the financial support from the National Nature Science Foundation of China (No. 21275118), and the Open Funds from the Key Laboratory of Synthetic and Natural Functional Molecule Chemistry of Ministry of Education and Shaanxi Provincial Key Laboratory of Electroanalytical Chemistry, China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, X., Song, Z. Human Saliva-Based Quantitative Monitoring of Clarithromycin by Flow Injection Chemiluminescence Analysis: A Pharmacokinetic Study. Appl Biochem Biotechnol 172, 1320–1331 (2014). https://doi.org/10.1007/s12010-013-0605-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0605-4