Abstract
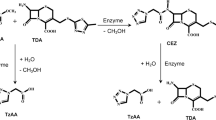
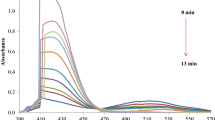
Cephamycin C (CepC) is a β-lactam antibiotic that belongs to the cephalosporin class of drugs. This compound stands out from other cephalosporins for its greater resistance to β-lactamases, which are enzymes produced by pathogenic microorganisms that present a major mechanism of bacterial resistance to β-lactam antibiotics. Cephamycin C is produced by the bacterium Streptomyces clavuligerus. Knowledge about the stability of the compound under different values of pH is important for the development of the process of production, extraction, and purification aimed at obtaining higher yields. Therefore, the stability of cephamycin C under different pH levels (2.2, 6.0, 7.0, 7.6, and 8.7) at 20 °C was evaluated in this study. Ultrafiltered broth from batch fermentations of S. clavuligerus was used in the trials. The results indicated that cephamycin C is a more stable compound than other β-lactam compounds such as penicillin and clavulanic acid. A higher degradation rate was observed at very acidic or basic pH levels, while this rate was lower at quasi-neutral pH levels. After 100 h of trial, the initial CepC showed 46 % degradation at pH 2.2, 71 % degradation at pH 8.7, and varied from 15 to 20 % at quasi-neutral pH levels.



Similar content being viewed by others
Abbreviations
- CepC:
-
Cephamycin C
- kH :
-
Hydrolysis kinetics constant
- ka :
-
Partial constant of hydrolysis in acidic medium
- kn :
-
Partial constant of spontaneous or water hydrolysis
- kb :
-
Partial constant of hydrolysis in basic medium
References
Demain, A. L. (2006). From natural products discovery to commercialization: a success story. Journal of Industrial Microbiology and Biotechnology, 33, 486–495.
Liras, P. (1999). Biosynthesis and molecular genetics of cephamycins. Antonie Van Leeuwenhoek, 75, 109–124.
Kresse, H., Belsey, M. J., & Rovini, H. (2007). The antibacterial drugs market. Natura Reviews Drug Discovery, 6, 19–20.
Baggaley, K. H., Brown, A. G., & Schofield, C. J. (1997). Chemistry and biosynthesis of clavulanic acid and other clavams. Natural Product Reports, Surrey, 14, 309–333.
Oliveira, J. H. H., Granato, A. C., Hirata, D. B., Hokka, C. O., Barboza, M., & Trsic, M. (2009). Ácido clavulânico e cefamicina C: uma perspectiva da biossíntese, processos de isolamento e mecanismo de ação. Química Nova, 32, 2142–2150.
Demain, A. L., & Elander, R. P. (1999). The β-lactam antibiotics: past, present, and future. Antonie van Leeuwenhoek, 75, 5–19.
Baptista Neto, A., Bustamante, M. C., Oliveira, J. H. H. L., Granato, A. C., Bellão, C., Badino, A. C., Barboza, M., & Hokka, C. O. (2012). Preliminary studies for cephamycin C purification technique. Applied Biochemistry and Biotechnology, 166, 208–221.
Baptista Neto, A., Oliveira, J. H. H. L., Bellão, C., Badino, A. C., Barboza, M., & Hokka, C. O. (2012). An alternative methodology for determination of cephamycin C from fermentation broth. Journal of Chromatography & Separation Techniques, 3, 1–5.
Konecny, J., Felber, E., & Gruner, J. (1973). Kinetics of hydrolysis of cephalosporin C. The Journal of Antibiotics, 26, 135–141.
Bellão, C., Antonio, T., Araujo, M. L. G. C., Badino, A. C. (2013). Production of clavulanic acid and cephamycin C by Streptomyces clavuligerus under different fed-batch conditions. Brazilian Journal of Chemical Engineering, 30, 257–266.
Silva, C. S., Rodrigues, Y. R., Hokka, C. O., Barboza, M. (2009). Study of fouling index in tangential filtration applied for separation of clavulanic acid from fermentation broths. In: Antonio Mendez-Vilas (Org.). Current research topics in applied microbiology and microbial biotechnology. World Scientific Publishing Co. Pte. Ltd., Sevilha, 2009, pp. 765–777.
Villela, G. G., Bacila, M., & Tastaldi, H. (1973). Técnicas e experimentos de bioquímica (pp. 28–31). Rio de Janeiro: Guanabara Koogan.
Bersanetti, P. A., Almeida, R. M. R. G., Barboza, M., Araújo, M. L. G. C., & Hokka, C. O. (2005). Kinetic studies on clavulanic acid degradation. Biochemical Engineering Journal, 23, 31–36.
Méndez, R., Alemany, T., & Martín-Villacorta, J. (1992). Stability in aqueous solution of two monocyclic β-lactam antibiotics: aztreonam and nocardicin A. Chemical and Pharmaceutical Bulletin, 40, 3222–3227.
Okamoto, Y., Kiriyama, K., Namiki, Y., Matsushita, J., Fujioka, M., & Yasuda, T. (1996). Degradation kinetics and isomerization of cefdinir: a new oral cephalosporin in aqueous solution. Journal of Pharmaceutical Sciences, 85, 976–983.
Mitchell, S. M., Ullman, J. L., Teel, A. L., & Watts, R. J. (2014). pH and temperature effects on the hydrolysis of three β-lactam antibiotics: ampicillin, cefalotin and cefoxitin. Science of the Total Environment, 466–467, 547–555.
Acknowledgments
The authors thank the Brazilian research funding agencies FAPESP (process no. 05/55079-4), CAPES, and CNPq for their financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brites, L.M., Oliveira, L.M. & Barboza, M. Kinetic Study on Cephamycin C Degradation. Appl Biochem Biotechnol 171, 2121–2128 (2013). https://doi.org/10.1007/s12010-013-0502-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0502-x