Abstract
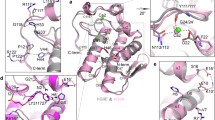
Human non-pancreatic secretory phospholipase A2 (hnpsPLA2) catalyzes the sn-2 acyl hydrolysis of phospholipids. It was reported that hnpsPLA2 is involved in various diseases like inflammation, cancer, and so on. This enzyme also exhibits anti-bacterial and anti-virus activities. It is active over a broad pH range, with higher activity at alkaline conditions. In order to make it suitable as a potential bactericide, high activity at neutral pH is preferable. We have tried to tailor the pH dependence of hnpsPLA2 activity by replacing its surface charged residues. Three surface charge replacements, Arg42Glu, Arg100Glu, and Glu89Lys, showed increased activities at neutral pH, which are 2.3, 2.8, and 2.3 times that of the wild-type enzyme at pH 7. Both the positive-to-negative and negative-to-positive mutations lowered the optimum enzymatic reaction pH of hnpsPLA2, indicating that the enzyme pH profile depends on a delicate balance of charged residues. The activity changes are in good agreement with the recently proposed calcium-coordinated catalytic triad mechanism. This study also provides a general means of enhancing hnpsPLA2 activity at low pH.



Similar content being viewed by others
Abbreviations
- hnpsPLA2 :
-
Human non-pancreatic secretory phospholipase A2
- IBS:
-
Interface binding site
- CD:
-
Circular dichroism
- ANS:
-
1-anilinonaphthalene-8-sulfonate
- DMPC:
-
1,2-dimyristoyl-sn-glycero-3-phosphocholine
- GIIAPLA2 :
-
Group IIA phospholipase A2
- V max :
-
The maximum velocity of the enzymatic activity
References
Crowl, R. M., Stoller, T. J., Conroy, R. R., & Stoner, C. R. (1991). Journal of Biological Chemistry, 266(4), 2647–2651.
Yuan, C., & Tsai, M. (1999). Biochimica et Biophysica Acta, 1441(2–3), 215–22.
Boilard, E., Bourgoin, S. G., Bernatchez, C., Poubelle, P. E., & Surette, M. E. (2003). FASEB Journal, 17(9), 1068–80.
Verheij, H. M., Volwerk, J. J., Jansen, E. H., Puyk, W. C., Dijkstra, B. W., Drenth, J., et al. (1980). Biochemistry, 19(4), 743–50.
Seshadri, K., Vishveshwara, S., & Jain, M. K. (1994). Proceedings of the Indian Academy of Sciences-Chemical Sciences, 106(5), 1177–1189.
Leopoldini, M., Russo, N., & Toscano, M. (2010). Journal of Physical Chemistry B, 114(35), 11584–93.
Thomas, P. G., Russell, A. J., & Fersht, A. R. (1985). Nature, 318(6044), 375–376.
Valenzue, P., & Bender, M. L. (1971). Biochimica et Biophysica Acta, 250(3), 538.
DeSantis, G., & Jones, J. B. (1998). JACS, 120(34), 8582–8586.
Fang, T. Y., & Ford, C. (1998). Protein Engineering, 11(5), 383–388.
Masui, A., Fujiwara, N., Yamamoto, K., Takagi, M., & Imanaka, T. (1998). Journal of Fermentation and Bioengineering, 85(1), 30–36.
Tomschy, A., Brugger, R., Lehmann, M., Svendsen, A., Vogel, K., Kostrewa, D., et al. (2002). Applied and Environmental Microbiology, 68(4), 1907–1913.
Lai, L. H., Wang, Y. L., Xu, X. J., Tang, Y. Q., Ni, Y. S., & Zhang, L. X. (1994). Science in China Series B-Chemistry, 37(1), 1–5.
Huang, C., Zhou, L., Liu, Y., & Lai, L. (2006). Analytical Biochemistry, 351(1), 11–7.
Baker, S. F., Othman, R., & Wilton, D. C. (1998). Biochemistry, 37(38), 13203–11.
Snitko, Y., Koduri, R. S., Han, S. K., Othman, R., Baker, S. F., Molini, B. J., et al. (1997). Biochemistry, 36(47), 14325–33.
Canaan, S., Nielsen, R., Ghomashchi, F., Robinson, B. H., & Gelb, M. H. (2002). Journal of Biological Chemistry, 277(34), 30984–90.
Hansford, K. A., Reid, R. C., Clark, C. I., Tyndall, J. D. A., Whitehouse, M. W., Guthrie, T., et al. (2003). Chembiochem, 4(2–3), 181–185.
Karray, A., Frikha, F., Ben Ali, Y., Gargouri, Y., & Bezzine, S. (2011). Lipids in Health and Disease, 10, 27.
Janssen, M. J., Vermeulen, L., Van der Helm, H. A., Aarsman, A. J., Slotboom, A. J., & Egmond, M. R. (1999). Biochimica et Biophysica Acta, 1440(1), 59–72.
Pan, Y. H., & Bahnson, B. J. (2010). Biochimica et Biophysica Acta, 1804(7), 1443–8.
Russell, A. J., & Fersht, A. R. (1987). Nature, 328(6130), 496–500.
Murakami, M., Taketomi, Y., Sato, H., & Yamamoto, K. (2011). Journal of Biochemistry, 150(3), 233–55.
Cha, J., & Batt, C. A. (1998). Molecules and Cells, 8(4), 374–82.
Noel, J. P., Bingman, C. A., Deng, T. L., Dupureur, C. M., Hamilton, K. J., Jiang, R. T., et al. (1991). Biochemistry, 30(51), 11801–11.
Weiss, J., Wright, G., Bekkers, A. C., van den Bergh, C. J., & Verheij, H. M. (1991). Journal of Biological Chemistry, 266(7), 4162–7.
Bhat, M. K., Pickersgill, R. W., Perry, B. N., Brown, R. A., Jones, S. T., Mueller-Harvey, I., et al. (1993). Biochemistry, 32(45), 12203–8.
Goodenough, P. W., Bhat, K. M., Collins, M. E., Perry, B. N., Pickersgill, R. W., Sumner, I. G., et al. (1991). Protein Engineering, 4(8), 929–34.
Pickersgill, R. W., Sumner, I. G., Collins, M. E., Warwicker, J., Perry, B., Bhat, K. M., et al. (1991). FEBS Letters, 281(1–2), 219–22.
Zhu, H., Dupureur, C. M., Zhang, X., & Tsai, M. D. (1995). Biochemistry, 34(46), 15307–14.
Song, J. K., & Rhee, J. S. (2000). Applied and Environmental Microbiology, 66(3), 890–4.
Li, Y. S., & Tsai, M. D. (1993). JACS, 115(19), 8523–8526.
Kumar, A., Sekharudu, C., Ramakrishnan, B., Dupureur, C. M., Zhu, H. X., Tsai, M. D., et al. (1994). Protein Science, 3(11), 2082–2088.
Sekar, K., Yu, B. Z., Rogers, J., Lutton, J., Liu, X., Chen, X., et al. (1997). Biochemistry, 36(11), 3104–14.
Janssen, M. J., van de Wiel, W. A., Beiboer, S. H., van Kampen, M. D., Verheij, H. M., Slotboom, A. J., et al. (1999). Protein Engineering, 12(6), 497–503.
Edwards, S. H., Thompson, D., Baker, S. F., Wood, S. P., & Wilton, D. C. (2002). Biochemistry, 41(52), 15468–76.
Liu, L. W., Wang, B., Chen, H. G., Wang, S. Y., Wang, M. D., Zhang, S. M., et al. (2009). Process Biochemistry, 44(8), 912–915.
Berg, O. G., Gelb, M. H., Tsai, M. D., & Jain, M. K. (2001). Chemistry Review, 101(9), 2613–54.
Hu, K. K., & Tanaka, T. (2009). Biochim Biophys Acta-Proteins and Proteomics, 1794(12), 1715–1724.
Turunen, O., Vuorio, M., Fenel, F., & Leisola, M. (2002). Protein Engineering, 15(2), 141–5.
Kusano, M., Yasukawa, K., Hashida, Y., & Inouye, K. (2006). Journal of Biochemistry, 139(6), 1017–1023.
Zhou, G. W., Guo, J., Huang, W., Fletterick, R. J., & Scanlan, T. S. (1994). Science, 265(5175), 1059–64.
Acknowledgments
This work was supported in part by the National Natural Science Foundation of China and the Ministry of Science and Technology of China.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 246 kb)
Rights and permissions
About this article
Cite this article
Qiu, S., Lai, L. Tailoring the pH Dependence of Human Non-pancreatic Secretory Phospholipase A2 by Engineering Surface Charges. Appl Biochem Biotechnol 171, 1454–1464 (2013). https://doi.org/10.1007/s12010-013-0437-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0437-2