Abstract
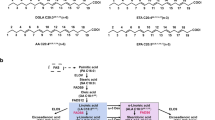
Two recombinant Rhizopus stolonifer ∆6-fatty acid desaturase enzymes with different-length N-termini were cloned and expressed in Saccharomyces cerevisiae strain INVScl: LRsD6D begins with the sequence of the N-terminal of the R. stolonifer ∆6-fatty acid desaturase native, encoding a deduced polypeptide of 459 amino acids (M-S-T-L-D-R-Q-S-I-F-T-I-K-E-L-E-S-I-S-Q-R-I-H-D-G-D-E-E-A-M-K-F), whereas SRsD6D begins with the amino acid sequence of the predicted ORF, encoding a deduced polypeptide of 430 amino acids (M-K-F) and LRsD6D is longer than SRsD6D by 29 amino acids (M-S-T-L-D-R-Q-S-I-F-T-I-K-E-L-E-S-I-S-Q-R-I-H-D-G-D-E-E-A). Bioinformatic analysis characterized the two recombinant ∆6-fatty acid desaturase enzymes with different-length N-termini, including three conserved histidine-rich motifs, hydropathy profile, and a cytochrome b5-like domain in the N-terminus. When the coding sequence was expressed in S. cerevisiae strain INVScl, the coding produced ∆6-fatty acid desaturase activity exhibited by RsD6D, leading to a novel peak corresponding to γ-linolenic acid methyl ester standards, which was detected with the same retention time. The residual activity of LRsD6D was 74 % at 15 °C for 4 h and that of SRsD6D was 43 %. Purified recombinant LRsD6D was more stable than SRsD6D, indicating that the N-terminal extension, containing mostly hydrophobic residues, affected the overall stability of recombinant LRsD6D.



Similar content being viewed by others
References
Los, D. A., & Murata, N. (1998). Structure and expression of fatty acid desaturase. Biochimica et Biophysica Acta, 1394, 3–15.
Vigh, L., Maresca, B., & Harwood, J. L. (1998). Does the membrane’s physical state control the expression of heat shock and other genes? Trends in Biochemical Sciences, 23, 369–374.
Saidi, Y., Finka, A., Muriset, M., Bromberg, Z., Weiss, Y. G., Maathuis, F. J. M., & Goloubinoff, P. (2009). The heat shock response in moss plants is regulated by specific calcium-permeable channels in the plasma membrane. The Plant Cell, 21, 1–15.
Huang, Y. S., Chaudhary, S., Thurmond, J. M., Bobik, J. R. E. G., Yuan, L., Chan, G. M., Kirchner, S. J., Mukerji, P., & Knutzon, D. S. (1999). Cloning of ∆12-and ∆6-desaturase from Mortierella alpina and recombinant production of γ-linolenic acid in S. cerevisiae. Lipids, 34, 649–659.
Murphy, D. J. (1996). Engineering oil production in rapeseed and other oil crops. Trends in Biotechnology, 14, 206–213.
Napier, J. A., Michaelson, L. V., & Stobart, A. K. (1999). Plant desaturase: harvesting the fat of the land. Current Opinion in Plant Biology, 2, 123–127.
Sayanova, O., Smith, M. A., Lapinskes, P., Stobart, A. K., Dobson, G., Christie, W. W., Shewry, P. R., & Napier, J. A. (1997). Expression of a borage desaturase cDNA containing an N-terminal cytochrome b5 domain results in the accumulation of high levels of ∆6-desaturated fatty acid in transgenic tobacco. Proceedings of the National Academy of Sciences of the United States of America, 94, 4211–4216.
Sayanova, O., Beaudoin, F., Libisch, B., Castel, A., Shewry, P. R., & Napier, J. A. (2001). Mutagenesis and heterologous expression in yeast of a plant delta6-fatty acid desaturase. Journal of Experimental Botany, 52, 1581–1585.
Shankin, J., Whittle, E., & Fox, B. G. (1994). Eight histidine residues are catalytically essential in a membrane-associated iron enzyme, stearoyl-CoA desaturase are conserved in alkane hydrocylase and xylene monooxygenase. Biochemistry, 33, 12787–12794.
Gill, I., & Valivety, R. (1997). Polyunsaturated fatty acids, part 1: occurrence, biological activities and applications. Trends in Biotechnology, 15, 401–409.
Zhang, Q., Li, M. C., Sun, Y., Ma, H. T., & Xing, L. J. (2004). Influence of sequence modification flanking AUG codon on ∆6-fatty acid desaturase gene expression. Acta Microbiologica Sinica, 44, 536–539.
Lu, H., Li, J. N., Chai, Y. R., & Zhang, X. K. (2009). Identification and characterization of a novel delta 6-fatty acid desaturase gene from Rhizopus stolonifer. Molecular Biology Reports, 36, 2291–2297.
Elble, R. (1992). A simple and efficient procedure for transformation of yeasts. Biotechniques, 13, 18–20.
Xian, M., Kang, Y. J., Yan, J. C., Liu, Y., Li, W. X., Bi, Y. L., & Zhen, K. J. (2002). ∆6 Desaturase catalyzing linoleic acid to γ-linolenic acid. Chemical Journal of Chinese Universities, 23(4), 624–627.
Petrovskaya, L., Ozerskaya, S., Ivanushkina, N., Kochkina, G., Laurinavichuis, K., Pecheritsina, S., Fattakhova, R., & Tiedje, J. M. (2005). Biodiversity of cryopegs in permafrost. FEMS Microbiology Ecology, 53, 117–128.
Cary, S. C., McDonald, I. R., Barrett, J. E., & Cowan, D. A. (2010). On the rocks: the microbiology of Antarctic Dry Valley soils. Nature Reviews Microbiology, 8, 129–138.
Deming, J. W. (2002). Psychrophiles and polar regions. Current Opinion in Microbiology, 5, 301–309.
Margesin, R., Feller, G., Gerday, C., & Russell, N. J. (2002). Cold-adapted microorganisms: adaptation strategies and biotechnological potential. In G. Bitton (Ed.), The encyclopedia of environmental microbiology (pp. 871–885). New York: Wiley.
Macartney, A., Maresca, B., & Cossins, A. R. (1994). Acyl-CoA desaturases and the adaptive regulation of membrane lipid composition. In A. R. Cossins (Ed.), Temperature adaptation of biological membranes (pp. 63–76). London: Portland.
Chaudhuri, T. K., Horii, K., Yoda, T., Arai, M., Nagata, S., Terada, T. P., Uchiyama, H., Ikura, T., Tsumoto, K., Kataoka, H., Matsushima, M., Kuwajima, K., & Kumagai, I. (1999). Effect of the extra N-terminal methionine residue on the stability and folding of recombinant alpha-lactalbumin expressed in Escherichia coli. Journal of Molecular Biology, 285, 1179–1194.
Yip, K. S., Stillman, T. J., Britton, K. L., Artymiuk, P. J., Baker, P. J., Sedelnikova, S. E., Engel, P. C., Pasquo, A., Chiaraluce, R., & Consalvi, V. (1995). The structure of Pyrococcus furiosus glutamate dehydrogenase reveals a key role for ion-pair networks in maintaining enzyme stability at extreme temperature. Structure, 3, 1147–1158.
Saelensminde, G., Halskau, J., Helland, R., Willassen, N. P., & Jonassen, I. (2007). Structure-dependent relationships between growth temperature of prokaryotes and the amino acid frequency in their proteins. Extremophiles, 11, 585–596.
Feller, G., Arpigny, J. L., Narinx, E., & Gerday, C. (1997). Molecular adaptation of enzymes from psychrophilic organisms. Comparative Biochemistry and Physiology, 118A(495), 499.
Acknowledgments
This research was supported by the Key Project of Chinese Ministry of Education, China National “948” Program, China National “863” Program, etc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, H., Zhu, Y. The Thermostability of Two Kinds of Recombinant ∆6-Fatty Acid Desaturase with Different N-Terminal Sequence Lengths in Low Temperature. Appl Biochem Biotechnol 171, 165–172 (2013). https://doi.org/10.1007/s12010-013-0363-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0363-3