Abstract
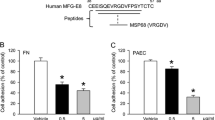
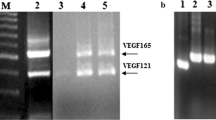
β-lactoglobulin (β-lg), a major whey protein was purified and characterised from buffalo colostrum. The in silico analysis of the tryptic peptides based on LC-CID-MS/MS facilitated the identification of protein as β-lg. The sequences IIVTQ f[1–5] and LSFNPTQLEEQCHV f(149–162) of m/z 933+ and 8512+ were found to match N- and C-extreme of β-lg while IDALNENK f(84–91) and TPEVDDEALEKFDK f(125–138) sequences deduced for m/z 916+ and 8182+ were in compliance to buffalo milk β-lg. Considering the sequence similarity of β-lg to glycodelin, a proven angiogenic protein, similar role for β-lg from buffalo colostrum (BLG-col) was examined. Interestingly, BLG-col exhibited anti-angiogenic activity by potently inhibiting cell proliferation, micro-vessel sprouting, cell migration and tube formation of human umbilical vein endothelial cells (HUVECs) in a dose-dependent manner but having varied effect on Ehrlich ascites tumor cells, MCF-7, MDA-MB 435 and MDA-MB 231 cell lines. The anti-angiogenic potential of BLG-col was found to be vascular endothelial growth factor mediated. The immunolocalisation of BLG-col on the cell surface of HUVECs evidenced using FITC-labelled β-lg antibody indicated its extra-cellular binding. Furthermore, BLG-col interacting HUVEC membrane protein (64 kDa) was detected by immunoblot and its identity was established by matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry analysis, which showed peptide sequence homology to G protein-coupled receptor kinase 4.








Similar content being viewed by others
References
Young, W., & Park, A (2013). Bioactive components in milk and dairy products. New York: Wiley, p 20.
Hernandez-Ledesma, B., Recio, I., & Amigo, L. (2008). β-lactoglobulin as source of bioactive peptides: review article. Amino Acids, 35, 257–265.
Flower, D. R., North, A. C. T., & Sansom, C. E. (2000). The lipocalin protein family-structural and sequence overview. Biochimica et Biophysica Acta, 1482, 9–24.
Yang, M. C., Chen, N. C., Chen, C. J., Wu, C. Y., & Mao, S. J. (2009). Evidence for beta-lactoglobulin involvement in vitamin D transport in vivo—role of the gamma-turn (Leu-Pro-Met) of beta-lactoglobulin in vitamin D binding. FEBS Journal, 276, 2251–2265.
Korhonen, H., & Pihlanto, A. (2006). Bioactive peptides: production and functionality. International Dairy Journal, 16, 945–960.
Sawyer, L., & Kontopidis, G. (2000). The core lipocalin bovine β-lactoglobulin. Biochimica et Biophysica Acta, 1482, 136–148.
Perez, M. D., Sanchaz, L., Aranda, P., Ena, J. M., Oria, R., & Calvo, M. (1992). Effect of beta-lactoglobulin on the activity of pregastic lipase a possible role for this protein in ruminant milk. Biochimica et Biophysica Acta, 1123, 151–155.
Koistinen, H., Koistinen, R., Seppälä, M., Burova, T. V., Choiset, Y., & Haertlé, T. (1999). Glycodelin and β-lactoglobulin, lipocalins with a high structural similarity, differ in ligand binding properties. FEBS Letters, 450, 158–162.
Mingqing, S., Sreemathy, R., Sumathi, R., Lisa, C. F., Ira, R. H., John, A. R., et al. (2001). Angiogenic role for glycodelin in tumorigenesis. PNAS, 98, 9265–9270.
Dvorat, H. F. (2000). VPF/VEGF and the angiogenic response. Seminars in Perinatology, 29, 75–78.
Denekampr, J. (1993). Review article: angiogenesis, neovascular proliferation and vascular pathophysiology as targets for Cancer therapy. British Journal Radiologists, 66, 181–196.
Agarwal, C., Singh, R. P., Dhanalakshmi, S., & Agarwal, R. (2004). Anti-angiogenic efficacy of grape seed extract in endothelial cells. Oncology Reports, 11, 681–685.
Chen, J., Montanari, A. M., & Widmer, W. W. (1997). Two new polymethoxylated flavonoids, a class of compounds with potential anticancer activity, isolated from cold pressed Dancy tangerine peel oil solids. Journal of Agricultural and Food Chemistry, 45, 364–368.
Zhang, Q., Tang, X., Lu, Q., Zhang, Z., Rao, J., & Le, A. D. (2006). Green tea extract and (−)-epigallocatechin-3-gallate inhibit hypoxia- and serum-induced HIF-1alpha protein accumulation and VEGF expression in human cervical carcinoma and hepatoma cells. Molecular Cancer Therapeutics, 5, 1227–1238.
Khan, N., Farrukh, A., Mee-Hyang, K., KyungMann, K., & Hasan, M. (2007). Oral consumption of pomegranate fruit extract inhibits growth and progression of primary lung tumors in mice. Cancer Research, 67, 3475–3482.
Miyazawa, T., Shibata, A., Nakagawa, K., & Tsuzuki, T. (2008). Anti-angiogenic function of tocotrienol. Asia Pacific Journal of Clinical Nutrition, 17, 253–256.
Shimamura, M., & Yamamoto, Y. (2004). Bovine lactoferrin inhibits tumor induced angiogenesis. International Journal of Cancer, 111, 111–116.
Shao, Z. M., Shen, Z. Z., Liu, C. H., Sartippour, M. R., Go, V. L., Heber, D., et al. (2002). Curcumin exerts multiple suppressive effects on human breast carcinoma cells. International Journal of Cancer, 98, 234–240.
Pierce, K. L., Premont, R. T., & Lefkowitz, R. J. (2002). Signalling: seven-transmembrane receptors. Nature Reviews Molecular Cell Biology, 3, 639–650.
Kotresh, A. M. (2009). Structural features of the 5’-franking region of the β-lactoglobulin gene of buffalo (Bubalus bubalis). Buffalo Bulletin, 28, 34–39.
Aparna, H. S., & Salimath, P. V. (1999). Studies on the acidic glycoproteins of buffalo colostrum and their influence on the growth of Bifidobacterium bifidus. Nutrition Research, 19, 295–303.
Hermodson, M., & Mahoney, W. C. (1983). Separation of peptides by reverse phase high performance liquid chromatography. Methods in Enzymology, 9, 352–359.
Rosenfeld, J., Capdevielle, J., Guillemot, J. C., & Ferrara, P. (1992). In-gel digestion for internal sequence analysis after one-or two-dimensional gel electrophoresis. Analytical Biochemistry, 203, 173–179.
Wu, S. L., Hühmer, A. F., Hao, Z., & Karger, B. L. (2007). On-line LC-MS approach combining collision-induced dissociation (CID), electron-transfer dissociation (ETD), and CID of an isolated charge-reduced species for the trace-level characterization of proteins with post translational. Journal of Proteome Research, 6, 4230–4244.
Versari, D., Lerman, L. O., & Lerman, A. (2007). The importance of reendotheialization after arterial injury. Current Pharmaceutical Design, 13, 1811–1824.
Sheela, M. L., Ramakrishna, M. K., & Salimath, B. P. (2006). Angiogenic and proliferative effects of the cytokine VEGF in Ehrlich ascites tumor cells is inhibited by Glycyrrhiza glabra. International Immunopharmacology, 6, 494–498.
Petra, W., Lechner, M., Merschak, P., & Redl, B. (2001). Molecular cloning of a novel lipocalin-1 interacting Human cell membrane receptor using phage display. Journal of Biological Chemistry, 276, 20206–20212.
Rohit, A. C., & Aparna, H. S. (2011). Characterization of β-lactoglobulin from buffalo (Bubalus bubalis) colostrum and its possible interaction with erythrocyte lipocalins interacting membrane receptor. Journal of Biochemistry, 150, 279–288.
William, J. S., & Wanda, P. (2009). Tolerance to water buffalo milk in a child with cow milk allergy. Annals of Allergy, Asthma & Immunology, 102, 349–350.
Rohit, A. C., Sathisha, K., & Aparna, H. S. (2012). A variant peptide of buffalo colostrum β-lactoglobulin inhibits angiotensin I converting enzyme activity. European Journal of Medicinal Chemistry, 53, 211–219.
Bratt, T. (2000). Lipocalins and cancer. Biochim Biophys Acta, 1482, 318–326.
Palupia, N. S., Franck, P., Guimont, C., Linden, G., Dumas, D., Stoltz, J., Nabet, P., Belleville-Nabet, F., & Dousset, B. (2000). Bovine β-lactoglobulin receptors on transformed mammalian cells (hybridomas MARK-3): characterization by flow cytometry. Journal of Biotechnology, 78, 171–184.
Noeske-Jungblut, C., Haendler, B., Donner, P., Alagon, A., Possani, L., & Wolf-Dieter, S. (1995). A highly potent exosite inhibitor of thrombin. Journal of Biological Chemistry, 270, 28629–28634.
Bratt, T., & Scott, G. K. (1995). Protein proteinase inhibitors as modulators of mammalian cell growth. Protein and Peptide Letters, 2, 391–402.
Van't Hof, W., Blankenvoorde, M. F., Veerman, E. C., & Amerongen, A. V. (1997). The salivary lipocalin von Ebner's gland protein is a cysteine proteinase inhibitor. Journal of Biological Chemistry, 272, 1837–1841.
Flower, D. R. (1994). The lipocalin protein family: a role in cell regulation. FEBS Letters, 354, 7–11.
Acknowledgements
We thank University Grant Commission, New Delhi, India for sanction of grant (F31-294/2005-06) to undertake this investigation and Rohit A. Chougule thank ICMR for award of SRF.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 1471 kb)
Rights and permissions
About this article
Cite this article
Chougule, R.A., P., S., Salimath, B.P. et al. Buffalo Colostrum β-lactoglobulin Inhibits VEGF-Induced Angiogenesis by Interacting with G protein-Coupled Receptor Kinase. Appl Biochem Biotechnol 171, 366–381 (2013). https://doi.org/10.1007/s12010-013-0344-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0344-6