Abstract
A newly isolated bacterial strain, Bacillus sp. MX47, was actively producing extracellular xylanase only in xylan-containing medium. The xylanase was purified from the culture broth by two chromatographic steps. The xylanase had an apparent molecular weight of 26.4 kDa with an NH2-terminal sequence (Gln-Gly-Gly-Asn-Phe) distinct from that of reported proteins, implying it is a novel enzyme. The optimum pH and temperature for xylanase activity were 8.0 and 40 °C, respectively. The enzyme activity was severely inhibited by many divalent metal ions and EDTA at 5 mM. The xylanase was highly specific to beechwood and oat spelt xylan, however, not active on carboxymethyl cellulose (CMC), avicel, pectin, and starch. Analysis of the xylan hydrolysis products by Bacillus sp. MX47 xylanase indicated that it is an endo-β-1,4-xylanase. It hydrolyzed xylan to xylobiose as the end product. The K m and V max values toward beechwood xylan were 3.24 mg ml−1 and 58.21 μmol min−1 mg−1 protein, respectively.





Similar content being viewed by others
References
Altschul, S. F., Madden, T. L., Schaffer, A. A., Zhang, J., Zhang, A., Miller, W., & Lipman, D. J. (1997). Nucleic Acids Research, 25, 3389–3402.
Bajpai, P. (1997). Advances in Applied Microbiology, 43, 141–194.
Basu, S., Roy, A., Ghosh, A., Bera, A., Chattopadhyay, D., & Chakrabarti, K. (2011). Biodegradation, 22, 153–161.
Bernier, R., Desrochers, M., Jurasek, L., & Paice, M. G. (1983). Applied and Environmental Microbiology, 46, 511–514.
Bustos-Jaimes, I., Mora-Lugo, R., Calcagno, M. L., & Farrés, A. (2010). Biochimica et Biophysica Acta, 1804, 2222–2227.
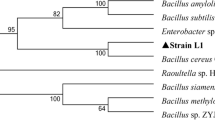
Chun, J., Lee, J. H., Jung, Y. Y., Kim, M. J., Kim, S. I., Kim, B. K., & Lim, Y. W. (2007). International Journal of Systematic Evolution and Microbiololgy, 57, 2259–2261.
Collins, T., Gerday, C., & Feller, G. (2005). FEMS Microbiology Reviews, 29, 3–23.
Felsenstein, J. (1993). PHYLIP (phylogeny inference package), version 3.5c. Distributed by the author. Seattle: Department of Genome Sciences, University of Washington.
Galkiewicz, J. P., & Kellogg, C. A. (2008). Applied and Environmental Microbiology, 74, 7828–7831.
Gallardo, O., Diaz, P., & Pastor, F. I. (2004). Current Microbiology, 48, 276–279.
Ghasemi, S., Ahmadian, G., Sadeghi, M., Zeigler, D. R., Rahimian, H., Ghandili, S., Naghibzadeh, N., & Dehestani, A. (2011). Enzyme and Microbial Technology, 48, 225–231.
Hall, T. A. (1999). Nucleic Acids Symposium Series, 41, 95–98.
Ibrahim, K. S., Muniyandi, J., & Karutha, P. S. (2011). Journal of Microbiology and Biotechnology, 21, 20–27.
Kamble, R. D., & Jadhav, A. R. (2012). International Journal of Microbiology. doi:10.1155/2012/683193.
Kimura, M. (1983). The neutral theory of molecular evolution. Cambridge: Cambridge University Press.
Kluge, A. G., & Farris, F. S. (1969). Systematic Zoology, 18, 1–32.
Knight, B. C., & Proom, H. (1950). Journal of General Microbiology, 4, 508–538.
Laemmli, U. K. (1970). Nature, 30, 545–550.
Lineweaver, H., & Burk, D. (1934). Journal of the American Chemistry Society, 56, 658–666.
Miller, G. L. (1959). Anaytical Biochemistry, 13, 426–428.
Nagar, S., Gupta, V. K., Kumar, D., Kumar, L., & Kuhad, R. C. (2010). Journal of Industrial Microbiology and Biotechnology, 37, 71–83.
Ratanakhanokchaim, K., Kyu, K. L., & Tanticharoen, M. (1999). Applied and Environmental Microbiology, 65, 694–697.
Sa-Pereira, P., Costa-Ferreira, M., & Aires-Barros, M. R. (2002). Journal of Biotechnology, 94, 265–275.
Saitou, N., & Nei, M. (1987). Molecular Biology and Evolution, 4, 406–425.
Satomi, M., La Duc, M. T., & Venkateswaran, K. (2006). International Journal of Systematic and Evolutionary Microbiology, 56, 1735–1740.
Segel, I. H. (1976). Enzyme kinetics. In Biochemical calculations. How to solve mathematical problems in general biochemistry (2nd ed., pp. 214–229). New York: John Wiley and Sons.
Shivaji, S., Chaturvedi, P., Suresh, K., Reddy, G. S. N., Dutt, C. B. S., Wainwright, M., Narlikar, J. V., & Bhargava, P. M. (2006). International Journal of Systematic and Evolutionary Microbiology, 56, 1465–1473.
Su, F., Hua, D., Zhang, Z., Wang, X., Tang, H., Tao, F., Tai, C., Wu, Q., Wu, G., & Xu, P. (2011). Journal of Bacteriology, 193, 6400–6401.
Subramaniyan, S. (2012). Applied Biochemistry and Biotechnology, 166, 1831–1842.
Thomson, J. D., Higgins, D. G., & Gibson, T. J. (1994). Nucleic Acids Research, 22, 4673–4680.
Wang, J., Zhang, W. W., Liu, J. N., Cao, Y. L., Bai, X. T., Gong, Y. S., Cen, P. L., & Yang, M. M. (2010). Molecular Biology Reports, 37, 3297–3302.
Wu, Q., Li, C., Li, C., Chen, H., & Shuliang, L. (2010). Applied Biochemistry and Biotechnology, 160, 129–139.
Yin, L. J., Lin, H. H., Chiang, Y. I., & Jiang, S. T. (2010). Journal of Agricultural and Food Chemistry, 58, 557–562.
Acknowledgments
This work was supported by Grant No. 2012-R1A1B3002174 from the Basic Research Program of the National Research Foundation (KRF) of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Won-Jae Chi and Da Yeon Park contributed equally for this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 301 kb)
Rights and permissions
About this article
Cite this article
Chi, WJ., Park, D.Y., Chang, YK. et al. A Novel Alkaliphilic Xylanase from the Newly Isolated Mesophilic Bacillus sp. MX47: Production, Purification, and Characterization. Appl Biochem Biotechnol 168, 899–909 (2012). https://doi.org/10.1007/s12010-012-9828-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9828-z