Abstract
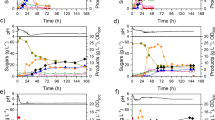
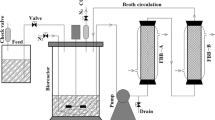
Clostridium acetobutylicum strains used in most Chinese ABE (acetone–butanol–ethanol) plants favorably ferment starchy materials like corn, cassava, etc., rather than sugar materials. This is one major problem of ABE industry in China and significantly limits the exploitation of cheap waste sugar materials. In this work, cane molasses were utilized as substrate in ABE production by Clostridium saccharobutylicum DSM 13864. Under optimum conditions, total solvent of 19.80 g/L (13.40 g/L butanol) was reached after 72 h of fermentation in an Erlenmeyer flask. In a 5-L bioreactor, total solvent of 17.88 g/L was attained after 36 h of fermentation, and the productivity and yield were 0.50 g/L/h and 0.33 g ABE/g sugar consumption, respectively. To further enhance the productivity, a two-stage semicontinuous fermentation process was steadily operated for over 8 days (205 h, 26 cycles) with average productivity (stage II) of 1.05 g/L/h and cell concentration (stage I) of 7.43 OD660, respectively. The average batch fermentation time (stage I and II) was reduced to 21−25 h with average solvent of 15.27 g/L. This study provides valuable process data for the development of industrial ABE fermentation process using cane molasses as substrate.





Similar content being viewed by others
References
Van der Merwe, A.B. (2010) Dissertation, University of Stellenbosch, South Africa.
Qureshi, N., & Blaschek, H. P. (2001). Journal of Industrial Microbiology & Biotechnology, 27, 292–297.
Dürre, P. (2007). Biotechnology Journal, 2, 1525–1534.
Ni, Y., & Sun, Z. H. (2009). Applied Microbiology and Biotechnology, 83, 415–423.
Thang, V. H., Kanda, K., & Kobayashi, G. (2010). Applied Biochemistry and Biotechnology, 161, 157–170.
Qureshi, N., & Blaschek, H. P. (2001). Journal of Industrial Microbiology & Biotechnology, 27, 287–291.
Lee, S. Y., Park, J. H., Jang, S. H., Nielsen, L. K., Kim, J., & Jung, K. S. (2008). Biotechnology and Bioengineering, 101, 209–228.
Liew, S. T., Arbakariya, A., Rosfarizan, M., & Raha, A. R. (2006). Malaysian Journal of Microbiolgy, 2, 42–50.
Pei, J. X., Zuo, W. P., Pang, H., Huang, Z. M., Li, Z. C., Wei, Y. T., et al. (2010). Liquor-Making Science & Technology, 5, 32–35 (in Chinese).
Fan, J. H., Feng, W. L., Di, S. M., Yan, H. R., Jia, J. J., & Wang, Z. P. (2010). Journal of Bioprocess Engineering, 8, 6–9 (in Chinese).
Madihah, M. S., Ariff, A. B., Sahaid, K. M., Suraini, A. A., & Karim, M. I. A. (2001). World Journal of Microbiology and Biotechnology, 17, 567–576.
Liu, Z. Y., Ying, Y., Li, F. L., Ma, C. Q., & Xu, P. (2010). Journal of Industrial Microbiology & Biotechnology, 37, 495–501.
Qureshi, N., Saha, B. C., Dien, B., Hector, R. E., & Cotta, M. A. (2010). Biomass and Bioenergy, 34, 559–565.
Lee, T. M., Ayaaki, I., Sadazo, Y., & Kensuke, F. (1995). Biotechnology Letters, 17, 649–654.
Cho, D. H., Lee, Y. J., Um, Y., Sang, B. I., & Kim, Y. H. (2009). Applied Microbiology and Biotechnology, 83, 1035–1043.
Kotzamanidis, C., Roukas, T., & Skaracis, G. (2002). World Journal of Microbiology and Biotechnology, 18, 441–448.
Patil, S. G., Gokhale, D. V., & Patil, B. G. (1989). Biotechnology Letters, 11, 213–216.
Liu, Y. P., Zheng, P., Sun, Z. H., Ni, Y., Dong, J. J., & Zhu, L. L. (2008). Bioresource Technology, 99, 1736–1742.
Syed, Q. (1994) Ph.D. dissertation, University of the Punjab, Pakistan.
Pătraşcu, E., Râpeanu, G., Bonciu, C., Vicol, C., & Bahrim, G. (2009). Ovidius University Annals of Chemistry, 20, 199–204.
Jiang, L., Wang, J. F., Liang, S. Z., Wang, X. N., Cen, P. L., & Xu, Z. N. (2009). Bioresource Technology, 100, 3403–3409.
Sharma, A., Vivekanand, V., & Singh, R. P. (2008). Bioresource Technology, 99, 3444–3450.
Ikram-ul, H., Ali, S., Qadeer, M. A., & Iqbal, J. (2004). Bioresource Technology, 93, 125–130.
Syed, Q. A., Nadeem, M., & Nelofer, R. (2008). Turkish Journal of Biochemistry, 33, 25–30.
Shaheen, R., Shirley, M., & Jones, D. T. (2000). Journal of Molecular Microbiology and Biotechnology, 2, 115–124.
Qureshi, N., Lolas, A., & Blaschek, H. P. (2001). Journal of Industrial Microbiology & Biotechnology, 26, 290–295.
Qureshi, N., Saha, B. C., Hector, R. E., & Cotta, M. A. (2008). Biomass and Bioenergy, 32, 1353–1358.
Qureshi, N., Saha, B. C., Hector, R. E., Dien, B., Hughes, S., Liu, S., et al. (2010). Biomass and Bioenergy, 34, 566–571.
Fu, T.T. (1974) Dissertation, Texas Tech University, Texas, USA.
Dong, J. J., Zheng, P., Sun, Z. H., Ni, Y., & Liu, Y. P. (2008). Journal of Chemical Industry and Engineering, 59, 1490–1495 (in Chinese).
Wee, Y. J., Kim, J. N., Yun, J. S., & Ryu, H. W. (2004). Enzyme and Microbial Technology, 35, 568–573.
Tsuey, L. S., Ariff, A. B., Mohamad, R., & Rahim, R. A. (2006). Biotechnology and Bioprocess Engineering, 11, 293–298.
Gu, Y., Hu, S. Y., Chen, J., Shao, L. J., He, H. Q., Yang, Y. L., et al. (2009). Journal of Industrial Microbiology & Biotechnology, 36, 1225–1232.
Bae, S. O., & Shoda, M. (2005). Applied Microbiology and Biotechnology, 67, 45–51.
Abou-Zeid, A. A., Fouad, M., & Yassein, M. (1978). Zentralbl Bakteriol Naturwiss, 133(2), 125–134.
Parekh, M., & Blaschek, H. P. (1999). Biotechnology Letters, 21, 45–48.
Oshiro, M., Hanada, K., Tashiro, Y., & Sonomoto, K. (2010). Applied Microbiology and Biotechnology, 87, 1177–1185.
Ezeji, T., Qureshi, N., & Blaschek, H. P. (2007). Biotechnology and Bioengineering, 97, 1460–1469.
Chaiklahan, R., Chirasuwan, N., Siangdung, W., Paithoonrangsarid, K., & Bunnag, B. (2010). Journal of Microbiology and Biotechnology, 20, 609–614.
Bauer, R., Katsikis, N., Varga, S., & Hekmat, D. (2005). Bioprocess and Biosystems Engineering, 5, 37–43.
Sanada, K., Kawaguchi, A., Furuya, T., Ishihara, K., Nakajima, N., & Hamada, H. (2000). Journal of Molecular Catalysis B: Enzymatic, 11, 59–61.
Afschar, A. S., Vaz Rossell, C. E., & Schaller, K. (1990). Applied Microbiology and Biotechnology, 34, 168–171.
Acknowledgments
The authors gratefully acknowledge the support of New Century Excellent Talents in University (NCET-11-0658), the Program of Introducing Talents of Discipline to Universities No. 111-2-06, the Priority Academic Program Development of Jiangsu Higher Education Institutions, and Green Biologics Ltd (Oxfordshire, UK).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Fig. A1
The effect of pH (a), trace element (b), nitrogen sources (c), and content of calcium carbonate (d) on ABE fermentation by C. saccharobutylicum DSM 13864. The fermentation medium and conditions were as follows: (a) 6% cane molasses, 3.2 g/L CaCO3, 2 g/L (NH4)2SO4, 0.5 g/L K2HPO4, pH 5.6−8.0, 35 °C; (b) 6% cane molasses, 3.2 g/L CaCO3, 2 g/L (NH4)2SO4, 0.5 g/L K2HPO4, 0.01 g/L trace element, pH 6.0, 35 °C; (c) 6% cane molasses, 3.2 g/L CaCO3, 2 g/L inorganic nitrogen sources or 2 g/L (NH4)2SO4, and 10 g/L organic nitrogen sources, 0.5 g/L K2HPO4, pH 6.0, 35 °C; (d) 6% cane molasses, 2.4−4.0 g/L CaCO3, 2 g/L (NH4)2SO4, 0.5 g/L K2HPO4, 20 g/L CSP, 0.01 g/L MnSO4·H2O, pH 6.0, 37 °C (JPEG 9 kb)
Rights and permissions
About this article
Cite this article
Ni, Y., Wang, Y. & Sun, Z. Butanol Production from Cane Molasses by Clostridium saccharobutylicum DSM 13864: Batch and Semicontinuous Fermentation. Appl Biochem Biotechnol 166, 1896–1907 (2012). https://doi.org/10.1007/s12010-012-9614-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9614-y