Abstract
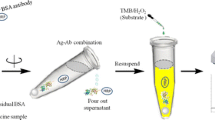
Enzyme-linked immunosorbent assay (ELISA), horseradish peroxidase (HRP)-catalyzed fluorescent reaction, and oxalate chemiluminescence analysis have been combined to develop a highly sensitive, simple, and rapid method for analysis of bovine serum albumin (BSA) based on a pair of specific monoclonal antibodies in vaccines. A typical “sandwich type” immunoassay was used. Reaction of 3-(4-hydroxyphenyl propionate) (PHPPA) with hydrogen peroxide-urea, catalyzed by HRP, produced fluorescence of 3-(4-hydroxyphenyl propionate) dimer, which was detected by chemiluminescence analysis with the bis(2,4,6-trichlorophenyl)oxalate (TCPO)–H2O2–glyoxaline–PHPPA dimer chemiluminescent system. This method exhibited high performance with a linear correlation between response and amount of bovine serum albumin (BSA) in the range 0.1 to 100.0 ng mL−1 (r = 0.9988), and the detection limit was 0.03 ng mL−1 (S/N = 3). Intra- and interassay coefficient variations were all lower than 9.0% at three concentrations (1.0, 20.0, and 80.0 ng mL−1). The proposed method has been used for successful analysis of the amount of residual BSA in vaccines. The results obtained compared well with those obtained by conventional colorimetric ELISA and luminol chemiluminescent ELISA.





Similar content being viewed by others
References
Boye, J. I., Alli, I., & Ismail, A. A. (1996). Interactions involved in the gelation of bovine serum albumin. Journal of Agricultural and Food Chemistry, 44, 996–1004.
Ferry, J. D. (1948). Protein gels. Advances in Protein Chemistry, 4, 1–78.
Kratz, F., Fichtner, I., Beyer, U., Schumacher, P., Roth, T., Fiebig, H. H., & Unger, C. (1997). Antitumor activity of acid labile transferring and albumindoxorubicin conjugates in in vitro and in vivo human tumor xenograft models. European Journal of Cancer, 33, S175.
Gutmann, O., Kuehlewein, R., Reinbold, S., & Niekrawietz, R. (2005). Fast and reliable protein microarray production by a new drop-in-drop technique. Lab on a Chip, 5, 675–681.
Mircean, C., Shmulevich, I., Cogdell, D., Choi, W., Jia, Y., Tabus, I., Hamilton, S. R., & Zhang, W. (2005). Robust estimation of protein expression ratios with lysate microarray technology. Bioinformatics, 21, 1935–1942.
Willison, H. J., Townson, K., Veitch, J., Boffey, J., Isaacs, N., Andersen, S. M., Ling, P. Z. C.-C., & Bundle, D. R. (2004). Synthetic disialylgalactose immunoadsorbents deplete anti-GQ1b antibodies from autoimmune neuropathy sera. Brain, 127, 680–691.
Mirapurkar, S., Nagvekar, U. H., & Sivaprasad, N. (2007). Polyreactivity of monoclonal antibodies produced against thyroid stimulating hormone (hTSH). Journal of Immunoassay Immunochemistry, 28, 119–126.
Studentsov, Y., Schiffman, M., Strickler, H. D., Ho, G. Y. F., Pang, Y. Y. S., Schiller, J., Herrero, R., & Burk, D. (2002). Enhanced enzyme-linked immunosorbent assay for detection of antibodies to virus-like particles of human papillomavirus. Journal of Clinical Microbiology, 40, 1755–1760.
Qiu, B., Stefanos, S., Ma, J., Lalloo, A., Perry, B. A., Leibowitz, M. J., Sinko, P. J., & Stein, S. (2003). A hydrogel prepared by in situ cross-linking of a thiolcontaining poly (ethylene glycol)-based copolymer: a new biomaterial for protein delivery. Biomaterials, 24, 11–18.
Chia, H. H., Yang, Y. Y., Chung, T. S., Ng, S., & Heller, J. (2001). Auto-catalyzed poly (ortho ester) microspheres: a study of their erosion and release mechanism. Journal of Controlled Release, 75, 11–25.
Kyselova, Z., Rackova, L., & Stefek, M. (2003). Pyridoindole antioxidant stobadine protected bovine serum albumin against the hydroxyl radical mediated crosslinking in vitro. Archives of Gerontology and Geriatrics, 36, 221–229.
Hoshi, S., Saito, N., Kusanagi, K., Ihara, T., & Ueda, S. (1998). Adjuvant effects of fluoride on oral immunization of chickens. Veterinary Immunology and Immunopathology, 63, 253–263.
Sprott, G. D., Tolson, D. L., & Patel, G. B. (1997). Archaeosomes as novel antigen delivery systems. FEMS Microbiology Letters, 15, 17–22.
Ramaldes, G. A., Deverre, J. R., Grognet, J. M., Puisieux, F., & Fattal, E. (1996). Use of an enzyme immunoassay for the evaluation of entrapment efficiency and in vitro stability in intestinal fluids of liposomal bovine serum albumin. International Journal of Pharmaceutics, 143, 1–11.
Hilleman, M. R. (2000). Vaccines in historic evolution and perspective: a narrative of vaccine discoveries. Vaccine, 18, 1436–1447.
Ada, G. (2003). Overview of vaccines. Methods in Molecular Medicine, 87, 1–17.
Pastorret, P. P., Brochier, B., & Denis, M. (1995). Environmental safety assessment of vaccines derived from biotechnology. Revue Scientifique et Technique, 14, 963–977.
Huang, C. Y., Liang, C. M., Li, C. Chu, Mei Liang, S. (2009). Albumin fibrillization induces apoptosis via integrin/FAK/Akt pathway. BMC Biotechnology, 9, 1–12.
Khamehchian, S., Madani, R., Golchinfar, F., & Taghavian, M. (2008). Development of a sandwich enzyme-linked immunosorbent assay (ELISA) for determining of bovine serum albumin (BSA) in trivalent measles–mumps–rubella (MMR) vaccines. Human Vaccines, 4, 375–378.
Fiocchi, A., Restani, P., Riva, E., Qualizza, R., Bruni, P., & Restelli, A. R. (1995). Meat allergy: 1—specific IgE to BSA and OSA in atopic, beef sensitive children. Journal of the American College of Nutrition, 14, 239–244.
Fiocchi, A., Restani, P., Riva, E., Mirri, G. P., Santini, I., & Bernardo, L. (1998). Heat treatment modifies the allergenicity of beef and bovine serum albumin. Allergy, 53, 798–802.
Martelli, A., Chiara, A., Corvo, M., Restani, P., & Fiocchi, A. (2002). Beef allergy in children with cow’s milk allergy; cow’s milk allergy in children with beef allergy. Annals of Allergy, Asthma & Immunology, 89, 38–43.
Vicente, S. J., Caballero, M. L., Rodriguez, P. R., Carretero, P., Perez, R., & Blanco, J. G. (2007). Sensitization to serum albumins in children allergic to cow’s milk and epithelia. Pediatric Allergy and Immunology, 18, 503–507.
Tanabc, S., Kobayashi, Y., Takahata, Y., Morimatsu, F., Shibata, R., & Nishimura, T. (2002). Some human B and T cell epitopes of bovine serum albumin, the major beef allergen. Biochemical and Biophysical Research Communications, 293, 1348–1353.
Day, M. J. (2007). Vaccine safety in the neonatal period. Journal of Comparative Pathology, 137, 51–56.
Ter Avest, A. R., Van Steenis, G., & Osterhaus, A. D. (1987). A comparison of an enzyme-linked immunosorbent assay and counter current electrophoresis for the detection of bovine serum albumin in virus vaccines. Journal of Biological Standardization, 15, 245–250.
Yu, Y., Lai, Y., Zheng, X. L., Wu, J. Z., Long, Z. Y., & Liang, C. S. (2007). Synthesis of functionalized CdTe/CdS QDs for spectrofluorimetric detection of BSA. Spectrochim Acta Part A: Molecular and Biomolecular Spectroscopy, 68, 1356–1361.
Rubtosova, M. J., Wittman, C., Egorov, A. M., & Schmid, R. D. (1997). Thermoalkalophilic lipase of Bacillus thermocatenulatus: large-scale production, purification and properties: aggregation behaviour and its effect on activity. Journal of Biotechnology, 56, 89–102.
Dzgoev, A. B., Gazaryan, I. G., Lagrimini, L. M., Ramanathan, K., & Danielsson, B. (1999). High-sensitivity assay for pesticide using a peroxidase as chemiluminescent label. Analytica Chimica Acta, 71, 5258–5261.
Tan, X. J., Song, Z. H., Chen, D. H., & Wang, Z. M. (2011). Study on the chemiluminescence behavior of bovine serum albumin with luminol and its analytical application. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 79, 232–235.
Zhang, K., Song, C. J., Li, Q., Li, Y. M., Sun, Y. J., Yang, K., & Jin, B. Q. (2010). The establishment of a highly sensitive ELISA for detecting bovine serum albumin (BSA) based on a specific pair of monoclonal antibodies (nAb) and its application in vaccine quality control. Human Vaccines, 6, 1–7.
Liang, S. X., Lia, H. F., & Lin, J. M. (2008). Reaction mechanism of surfactant-sensitized chemiluminescence of bis(2,4,6- trichlorophyenyl) oxalate and hydrogen peroxide induced by gold nanoparticles. Luminescence, 23, 381–385.
Acknowledgement
This work was supported financially by the Fundamental Research Funds for the Central Universities (Program No. GK20091004), the National High–tech R&D Program (863 Program) of China (No. 2006AA02A237), and the National Natural Science Foundation of China (No. 30872371).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Pan Xue and Kui Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xue, P., Zhang, K., Zhang, Z. et al. Highly Sensitive Chemiluminescent Analysis of Residual Bovine Serum Albumin (BSA) Based on a Pair of Specific Monoclonal Antibodies and Peroxyoxalate–glyoxaline–PHPPA Dimer Chemiluminescent System in Vaccines. Appl Biochem Biotechnol 166, 1604–1614 (2012). https://doi.org/10.1007/s12010-012-9567-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9567-1