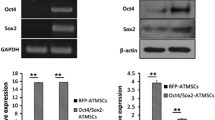
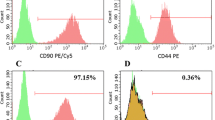
Abstract
Human adipose-derived adult stem cells (hADSCs) can express human telomerase reverse transcriptase phenotypes under an appropriate culture condition. Because adipose tissue is abundant and easily accessible, hADSCs offer a promising source of stem cells for tissue engineering application and other cell-based therapies. However, the shortage of cells number and the difficulty to proliferate, known as the “Hayflick limit” in vitro, limit their further clinical application. Here, hADSCs were transfected with human telomerase reverse transcriptase (hTERT) gene by the lentiviral vector to prolong the lifespan of stem cells and even immortalize them. Following to this, the cellular properties and functionalities of the transfected cell lines were assayed. The results demonstrated that hADSCs had been successfully transfected with hTERT gene (hTERT-ADSCs). Then, hTERT-ADSCs were initially selected by G418 and subsequently expanded over 20 passages in vitro. Moreover, the qualitative and quantitative differentiation criteria for 20 passages of hTERT-ADSCs also demonstrated that hTERT-ADSCs could differentiate into osteogenesis, chondrogenesis, and adipogenesis phenotypes in lineage-specific differentiation media. These findings confirmed that this transfection could prolong the lifespan of hADSCs.




Similar content being viewed by others
Abbreviations
- hADSCs:
-
Human adipose-derived adult stem cells
- hTERT:
-
Human telomerase reverse transcriptase
- s.c.:
-
Subcutaneously
- ALP:
-
Alkaline phosphatase
References
Hornsby, P. J. (2011). Cellular aging and cancer. Critical Reviews in Oncology/Hematology, 79(2), 189–95.
Baird, D. M. (2005). New developments in telomere length analysis. Experimental Gerontology, 40(5), 363–368.
Collins, K. (2011). Single-stranded DNA repeat synthesis by telomerase. Current Opin ion Chemical Biology, 15(5), 643–8.
Mason, M., Schuller, A., & Skordalakes, E. (2011). Telomerase structure function. Current Opinion in Structural Biology, 21(1), 92–100.
Cong, Y. S., & Shay, J. W. (2008). Actions of human telomerase beyond telomeres. Cell Research, 18(7), 725–32.
Bayne, S., Jones, M. E., Li, H., Pinto, A. R., Simpson, E. R., & Liu, J. P. (2008). Estrogen deficiency leads to telomerase inhibition, telomere shortening and reduced cell proliferation in the adrenal gland of mice. Cell Research, 18(11), 1141–50.
Borssén, M., Cullman, I., Norén-Nyström, U., Sundström, C., Porwit, A., Forestier, E., & Roos, G. (2011). hTERT promoter methylation and telomere length in childhood acute lymphoblastic leukemia-associations with immunophenotype and cytogenetic subgroup. Experimental Hematology, 39(12), 1144–51.
Venturini, L., Daidone, M. G., Motta, R., Collini, P., Spreafico, F., Terenziani, M., Piva, L., Radice, P., Perotti, D., & Zaffaroni, N. (2011). Telomere maintenance in Wilms tumors: First evidence for the presence of alternative lengthening of telomeres mechanism. Genes, Chromosomes & Cancer, 50(10), 823–9.
Hao, L. Y., Armanios, M., Strong, M. A., Karim, B., Feldser, D. M., Huso, D., & Greider, C. W. (2005). Short telomeres, even in the presence of telomerase, limit tissue renewal capacity. Cell, 123(6), 1121–1131.
Soares, J., Lowe, M. M., & Jarstfer, M. B. (2011). The catalytic subunit of human telomerase is a unique caspase-6 and caspase-7 substrate. Biochemistry, 50(42), 9046–55.
Cohen, S., Jacob, E., & Manor, H. (2004). Effects of single-stranded DNA binging proteins on primer extension by telomerase. Biochimica et Biophysica Acta, 1679(2), 129–40.
Bianchi, A., & Shore, D. (2008). How telomerase reaches its end: Mechanism of telomerase regulation by the telomeric complex. Molecular Cell, 31(2), 153–165.
Zhu, Y. X., Liu, T. Q., Song, K. D., Ning, R., Ma, X. H., & Cui, Z. F. (2009). ADSCs differentiated into cardiomyocytes in cardiac microenvironment. Molecular and Cellular Biochemistry, 324(1–2), 117–129.
Song, K., Wang, H., Wang, H., Wang, L., Qiao, M., Wu, S., & Liu, T. (2011). Investigation of the effective action distance between hematopoietic stem/progenitor cells and human adipose-derived stem cells during their in vitro co-culture. Applied Biochemistry and Biotechnology, 165(3–4), 776–84.
Zhu, Y., Liu, T., Song, K., Jiang, B., Ma, X., & Cui, Z. (2009). Collagen-chitosan polymer as a scaffold for the proliferation of human adipose tissue-derived stem cells. Journal of Materials Science. Materials in Medicine, 20(3), 799–808.
Qu, X., Liu, T., Song, K., Li, X., & Ge, D. (2012). Induced pluripotent stem cells generated from human adipose-derived stem cells using a non-viral polycistronic plasmid in feeder-free conditions. PloS One, 7(10), e48161.
Zhu, Y., Liu, T., Song, K., Fan, X., Ma, X., & Cui, Z. (2009). Ex vivo expansion of adipose tissue-derived stem cells in spinner flasks. Biotechnology Journal, 4(8), 1198–209.
Guilak, F., Lott, K. E., Awad, H. A., Cao, Q., Hicok, K. C., Fermor, B., & Gimble, J. M. (2006). Clonal analysis of the differentiation potential of human adipose-derived adult stem cells. Journal of Cellular Physiology, 206(1), 229–237.
Zhu, Y. X., Liu, T. Q., Song, K. D., Fan, X., Ma, X. H., & Cui, Z. F. (2008). Adipose-derived stem cell: a better stem cell than BMSC. Cell Biochemistry and Function, 26(6), 664–675.
Jiang, L., Liu, T., Song, K. (2012). Growth characteristics of human adipose-derived stem cells during long time culture regulated by cyclin a and cyclin D1. Applied Biochemistry and Biotechnology, 168(8), 2230–44.
Zhu, Y., Liu, T., Ye, H., Song, K., Ma, X., & Cui, Z. (2010). Enhancement of adipose-derived stem cell differentiation in scaffolds with IGF-I gene impregnation under dynamic microenvironment. Stem Cells and Development, 19(10), 1547–56.
Song, K., Li, W., Wang, H., Wang, H., Liu, T., Ning, R., & Wang, L. (2012). Investigation of coculture of human adipose-derived stem cells and mature adipocytes. Applied Biochemistry and Biotechnology, 167(8), 2381–7.
Macieira-Coelho, A. (2011). Cell division and aging of the organism. Biogerontology, 12, 503–515.
Cech, T. R. (2004). Beginning to understand the end of the chromosome. Cell, 116, 273–279.
Weng, N. P. (2008). Telomere and adaptive immunity. Mechanisms of Ageing and Development, 129(1–2), 60–6.
Britt-Compton, B., Capper, R., Rowson, J., & Baird, D. M. (2009). Short telomeres are preferentially elongated by telomerase in human cells. FEBS Letters, 583(18), 3076–80.
Jeon, B. G., Kumar, B. M., Kang, E. J., Ock, S. A., Lee, S. L., Kwack, D. O., Byun, J. H., Park, B. W., & Rho, G. J. (2011). Characterization and comparison of telomere length, telomerase and reverse transcriptase activity and gene expression in human mesenchymal stem cells and cancer cells of various origins. Cell and Tissue Research, 345(1), 149–61.
Acknowledgments
This work was supported by the Fok Ying Tung Education Foundation (132027), National Science Foundation of China (81271719), the State Key Laboratory of Fine Chemicals (KF1111), and the Fundamental Research Funds for the Central Universities (DUT11SM09/DUT12JB09) and SRF for ROCS, SEM.
Conflict of interests
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
L. Wang, K. Song, X. Qu, and H. Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, L., Song, K., Qu, X. et al. hTERT Gene Immortalized Human Adipose-Derived Stem Cells and its Multiple Differentiations: a Preliminary Investigation. Appl Biochem Biotechnol 169, 1546–1556 (2013). https://doi.org/10.1007/s12010-012-0019-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-0019-8