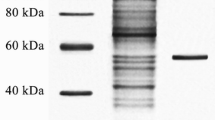
Abstract
The thermostable and organic solvent tolerant whole-cell lipase (WCL) was produced by Burkholderia sp. ZYB002 with broad spectrum organic solvent tolerance. The production medium of the WCL was primarily optimized, which resulted in the maximum activity of 22.8 U/mL and the 5.1-fold increase of the WCL yield. The optimized culture medium was as follows (% w/v or v/v): soybean meal 2, soybean oil 0.5, manganese sulfate 0.1, K2HPO4 0.1, olive oil 0.5, initial pH 6.0, inoculum density 2, liquid volume 35 mL in 250-mL Erlenmeyer flask, and incubation time 24 h. The biochemical characterization of the WCL from Burkholderia sp. ZYB002 was determined, and the results showed that the optimal pH and temperature for lipolytic activity of the WCL was 8.0 and 65°C, respectively. The WCL was stable at temperature up to 70°C for 1 h and retained 79.2% of its original activity. The WCL was highly stable in the pH range from 3.0 to 8.5 for 6 h. Ca2+, K+, Na+, NO −3 , etc. ions stimulated its lipolytic activity, whereas Zn2+ ion caused inhibition effect. The WCL was also relatively stable in n-butanol at a final concentration of 50% (v/v) for 24 h. However, the WCL was strongly inhibited in Triton X-100 at a final concentration of 10% (v/v). The WCL with thermal resistance and organic solvent tolerance showed its great potential in various green industrial chemical processes.







Similar content being viewed by others
References
Gotor-Fernandez, V., Brieva, R., & Gotor, V. (2006). Lipases: Useful biocatalysts for the preparation of pharmaceuticals. Journal of Molecular Catalysis B: Enzymatic, 40, 111–120. doi:10.1016/j.molcatb.2006.02.010.
Hasan, F., Shah, A. A., & Hameed, A. (2006). Industrial applications of microbial lipases. Enzyme and Microbial Technology, 39, 235–251. doi:10.1016/j.enzmictec.2005.10.016.
Sugihara, A., Ueshima, M., Shimada, Y., Tsunasawa, S., & Tominaga, Y. (1992). Purification and characterization of a novel thermostable lipase from Pseudomonas cepacia. Journal of Biochemistry (Tokyo), 112, 598–603.
Tomić, S., Bertoša, B., Kojić-Prodić, B., & Kolosvary, I. (2004). Stereoselectivity of Burkholderia cepacia lipase towards secondary alcohols: Molecular modelling and 3D QSAR approach. Tetrahedron-Asymmetry, 15, 1163–1172. doi:10.1016/j.tetasy.2004.02.016.
Wang, X. Q., Yu, X. W., & Xu, Y. (2009). Homologous expression, purification and characterization of a novel high-alkaline and thermal stable lipase from Burkholderia cepacia ATCC 25416. Enzyme and Microbial Technology, 45, 94–102. doi:10.1016/j.enzmictec.2009.05.004.
Kim, K. K., Song, H. K., Shin, D. H., Hwang, K. Y., & Suh, S. W. (1997). The crystal structure of a triacylglycerol lipase from Pseudomonas cepacia reveals a highly open conformation in the absence of a bound inhibitor. Structure, 5, 173–185. doi:10.1016/S0969-2126(97)00177-9.
Rathi, P., Goswami, V. K., Sahai, V., & Gupta, R. (2002). Statistical medium optimization and production of a hyperthermostable lipase from Burkholderia cepacia in a bioreactor. Journal of Applied Microbiology, 93, 930–936. doi:10.1046/j.1365-2672.2002.01780.x.
Yu, L. J., Xu, Y., Wang, X. Q., & Yu, X. W. (2007). Highly enantioselective hydrolysis of DL-menthyl acetate to L-menthol by whole-cell lipase from Burkholderia cepacia ATCC 25416. Journal of Molecular Catalysis B: Enzymatic, 47, 149–154. doi:10.1016/j.molcatb.2007.04.011.
Schepp, C., Kermasha, S., Michalski, M. C., & Morin, A. (1997). Production, partial purification and characterisation of lipases from Pseudomonas fragi CRDA 037. Process Biochemistry, 32, 225–232. doi:10.1016/S0032-9592(96)00065-9.
Wilhelm, S., Tommassen, J., & Jager, K. E. (1999). A novel lipolytic enzyme located in the outer membrane of Pseudomonas aeruginosa. Journal of Bacteriology, 181, 6977–6986.
Sun, S. Y., & Xu, Y. (2009). Membrane-bound ‘synthetic lipase’ specifically cultured under solid-state fermentation and submerged fermentation by Rhizopus chinensis: A comparative investigation. Bioresource Technology, 100, 1336–1342. doi:10.1016/j.biortech.2008.07.051.
Ni, Y., & Chen, R. R. (2004). Accelerating whole-cell biocatalysis by reducing outer membrane permeability barrier. Biotechnology and Bioengineering, 87, 804–811. doi:10.1002/bit.20202.
Fukuda, H., Hama, S., Tamalampudi, S., & Noda, H. (2008). Whole-cell biocatalysts for biodiesel fuel production. Trends in Biotechnology, 26, 668–673. doi:10.1016/j.tibtech.2008.08.001.
Wang, D., Xu, Y., & Teng, Y. (2007). Synthetic activity enhancement of membrane-bound lipase from Rhizopus chinensis by pretreatment with isooctane. Bioprocess and Biosystems Engineering, 30, 147–155. doi:10.1007/s00449-006-0097-5.
Hlavsova, K., Zarevucka, M., Wimmer, Z., Mackova, M., & Sovova, H. (2009). Geotrichum candidum 4013: Extracellular lipase versus cell-bound lipase from the single strain. Journal of Molecular Catalysis B: Enzymatic, 61, 188–193. doi:10.1016/j.molcatb.2009.06.012.
Sardessai, Y. N., & Bhosle, S. (2004). Industrial potential of organic solvent tolerant bacteria. Biotechnology Progress, 20, 655–660. doi:10.1021/bp0200595.
Shu, Z. Y., Lin, R. F., Jiang, H., Zhang, Y. F., Wang, M. Z., & Huang, J. Z. (2009). A rapid and efficient method for directed screening of lipase-producing Burkholderia cepacia complex strains with organic solvent tolerance from rhizosphere. Journal of Bioscience and Bioengineering, 107, 658–661. doi:10.1016/j.jbiosc.2009.01.011.
Saxena, R. K., Davidson, W. S., Sheoran, A., & Giri, B. (2003). Purification and characterization of an alkaline thermostable lipase from Aspergillus carneus. Process Biochemistry, 39, 239–247. doi:10.1016/S0032-9592(03)00068-2.
Kordel, M., Hofmann, B., Schomburg, D., & Schmid, R. D. (1991). Extracellular lipase of Pseudomonas sp. strain ATCC 21808: Purification, characterization, crystallization, and preliminary X-ray diffraction data. Journal of Bacteriology, 173, 4836–4841.
Tan, T. W., Zhang, M., Xu, J. L., & Zhang, J. (2004). Optimization of culture conditions and properties of lipase from Penicillium camembertii Thom PG-3. Process Biochemistry, 39, 1495–1502. doi:10.1016/S0032-9592(03)00296-6.
Che Omar, I., & Ilias, N. (1996). Characteristics of cell-bound lipase production by a newly isolated strain of Aspergillus flavus. Pertanika Journal of Science & Technology, 4, 1–9.
Gupta, N., Sahai, V., & Gupta, R. (2007). Alkaline lipase from a novel strain Burkholderia multivorans: Statistical medium optimization and production in a bioreactor. Process Biochemistry, 42, 518–526. doi:10.1016/j.procbio.2006.10.006.
Liu, C. H., Lu, W. B., & Chang, J. S. (2006). Optimizing lipase production of Burkholderia sp. by response surface methodology. Process Biochemistry, 41, 1940–1944. doi:10.1016/j.procbio.2006.04.013.
Gerritse, G., Hommes, R. W. J., & Quax, W. J. (1998). Development of a lipase fermentation process that uses a recombinant Pseudomonas alcaligenes strain. Applied and Environmental Microbiology, 64, 2644–2651.
Krzeslak, J., Gerritse, G., Merkerk, R. V., Cool, R. H., & Quax, W. J. (2008). Lipase expression in Pseudomonas alcaligenes is under the control of a two-component regulatory system. Applied and Environmental Microbiology, 74, 1402–1411. doi:10.1128/AEM.01632-07.
Iwai, M., & Tsujisaka, Y. (1984). Fungal lipase. In H. L. Brockman (Ed.), Lipases (pp. 443–469). Amsterdam: Elsevier.
Dandavate, V., Jinjala, J., Keharia, H., & Madamwar, D. (2009). Production, partial purification and characterization of organic solvent tolerant lipase from Burkholderia multivorans V2 and its application for ester synthesis. Bioresource Technology, 100, 3374–3381. doi:10.1016/j.biortech.2009.02.011.
Boekema Bouke, K. H. L., Beselin, A., Breuer, M., Hauer, B., Koster, M., Rosenau, F., et al. (2007). Hexadecane and Tween 80 stimulate lipase production in Burkholderia glumae by different mechanisms. Applied and Environmental Microbiology, 73, 3838–3844. doi:10.1128/AEM.00097-07.
Dalal, S., Singh, P. K., Raghava, S., Rawat, S., & Gupta, M. N. (2008). Purification and properties of the alkaline lipase from Burkholderia cepacia ATCC 25609. Biotechnology and Applied Biochemistry, 51, 23–31. doi:10.1042/BA20070186.
Sharma, A. K., Tiwari, R. P., & Hoondal, G. S. (2001). Properties of a thermostable and solvent stable extracellular lipase from a Pseudomonas sp. AG-8. Journal of Basic Microbiology, 41, 363–366. doi:10.1002/1521-4028(200112).
Sharma, R., Soni, S. K., Vohra, R. M., Gupta, L. K., & Gupta, J. K. (2002). Purification and characterisation of a thermostable alkaline lipase from a new thermophilic Bacillus sp. RSJ-1. Process Biochemistry, 37, 1075–1084. doi:10.1016/S0032-9592(01)00316-8.
Gupta, R., Gupta, N., & Rathi, P. (2004). Bacterial lipases: An overview of production, purification and biochemical properties. Applied Microbiology and Biotechnology, 64, 763–781. doi:10.1007/s00253-004-1568-8.
Acknowledgements
This work was supported by the National High-Technology Project (“863” Project) of P. R. China (No.2007AA100703), the National Natural Science Funds of People’s Republic of China (No. 30870545), and the Natural Science Funds for Distinguished Young Scholar of Fujian Province (No. 2009J06013).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zheng-Yu Shu and Ji-Guang Wu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shu, ZY., Wu, JG., Cheng, LX. et al. Production and Characteristics of the Whole-Cell Lipase from Organic Solvent Tolerant Burkholderia sp. ZYB002. Appl Biochem Biotechnol 166, 536–548 (2012). https://doi.org/10.1007/s12010-011-9446-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-011-9446-1