Abstract
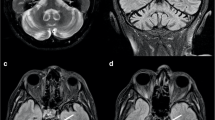
The effectiveness of Resovist-labeled bone marrow stem cells (BMSCs) was evaluated in vivo following their cerebral transplantation in a model of Parkinson’s disease (PD) in rats using MRI, and the MRI findings were further compared with the behavior and histopathological manifestations of these rats. Forty PD rats were randomly assigned into five groups according to the cell doses injected into the rat brain site: control group (normal saline injection) and groups injected with 1 × 105, 1.5 × 105, 2 × 105, and 2.5 × 105 BMSCs. Gradient echo T2-weighted images were obtained immediately after cell transplantation and repeatedly taken 1, 4, 8, and 12 week(s) after cell transplantation. The rotational behavior of the animals was observed before and 1, 4, and 8 week(s) after transplantation. The rats were killed after the last MRI scanning, the brain tissues were analyzed by histopathology techniques, and RNAs were extracted for the expression analysis of selected genes using RT-PCR. One week following cell transplantation, all injected sites showed well-defined hypointense areas on MR images, with the most significant effect observed in rats injected with 2 × 105 BMSCs. These MR findings in PD rats lasted up to 12 weeks. The effectiveness of BMSC transplantation revealed by MRI was well confirmed by the behavioral and histopathological observations as well as indirectly supported by gene expression analyses. With the use of SPIO labeling, MRI techniques provided a dynamic evaluation of the spatial and temporal changes following cell transplantation and allowed the association analysis among the imaging, functions, and gene expression analysis in rats. These data also suggest the therapeutic potential of transplanted BMSCs. It is reasonable to speculate that the use of MRI in in vivo evaluation of the effect and fate of transplanted cells in various disease models will be beneficial to developing new strategies of cell-based gene therapy.






Similar content being viewed by others
Abbreviations
- BMSCs:
-
Bone marrow stem cells
- MRI:
-
Magnetic resonance imaging
- PD:
-
Parkinson’s disease
- SPIO:
-
Superparamagnetic iron oxide
References
Galvan, A., & Wichmann, T. (2008). Pathophysiology of Parkinsonism. Clinical Neurophysiology, 119, 1459–1474.
Sanchez-Ramos, J., Song, S., Cardozo-Pelaez, F., Hazzi, C., Stedeford, T., Willing, A., et al. (2000). Adult bone marrow stromal cells differentiate into neural cells in vitro. Experimental Neurology, 164, 247–256.
Daniela, F., Vescovi, A. L., & Bottai, D. (2007). The stem cells as a potential treatment for neurodegeneration. Methods in Molecular Biology, 399, 199–213.
Woodbury, D., Schwarz, E. J., Prockop, D. J., & Black, I. B. (2000). Adult rat and human bone marrow stromal cells differentiate into neurons. Journal of Neuroscience Research, 61, 364–370.
Li, Y., Chen, J., Wang, L., Zhang, L., Lu, M., & Chopp, M. (2001). Intracerebral transplantation of bone marrow stromal cells in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson’s disease. Neuroscience Letters, 316, 67–70.
Strauer, B. E., & Kornowski, R. (2003). Stem cell therapy in perspective. Circulation, 107, 929–934.
Read, E. J., Keenan, A. M., Carter, C. S., Yolles, P. S., & Davey, R. J. (1990). In vivo traffic of indium-111-oxine labeled human lymphocytes collected by automated apheresis. Journal of Nuclear Medicine, 31, 999–1006.
Corot, C., Robert, P., Idee, J. M., & Port, M. (2006). Recent advances in iron oxide nanocrystal technology for medical imaging. Advanced Drug Delivery Reviews, 58, 1471–1504.
Jendelova, P., Herynek, V., Urdzikova, L., Glogarova, K., Kroupova, J., Andersson, B., et al. (2004). Magnetic resonance tracking of transplanted bone marrow and embryonic stem cells labeled by iron oxide nanoparticles in rat brain and spinal cord. Journal of Neuroscience Research, 76, 232–243.
Wang, Y. X., Hussain, S. M., & Krestin, G. P. (2001). Superparamagnetic iron oxide contrast agents: Physicochemical characteristics and applications in MR imaging. European Radiology, 11, 2319–2331.
Reimer, P., & Tombach, B. (1998). Hepatic MRI with SPIO: Detection and characterization of focal liver lesions. European Radiology, 8, 1198–1204.
Javazon, E. H., Colter, D. C., Schwarz, E. J., & Prockop, D. J. (2001). Rat marrow stromal cells are more sensitive to plating density and expand more rapidly from single-cell-derived colonies than human marrow stromal cells. Stem Cells, 19, 219–225.
Colter, D. C., Sekiya, I., & Prockop, D. J. (2001). Identification of a subpopulation of rapidly self-renewing and multipotential adult stem cells in colonies of human marrow stromal cells. Proceedings of the National Academy of Sciences of the United States of America, 98, 7841–7845.
Fawwaz, R. A., Oluwole, S., Wang, T. S., Kuromoto, N., Iga, C., Hardy, M. A., et al. (1985). Biodistribution of radiolabeled lymphocytes. Radiology, 155, 483–486.
Schafer, R., Kehlbach, R., Wiskirchen, J., Bantleon, R., Pintaske, J., Brehm, B. R., et al. (2007). Transferrin receptor upregulation: In vitro labeling of rat mesenchymal stem cells with superparamagnetic iron oxide. Radiology, 244, 514–523.
Sun, R., Dittrich, J., Le-Huu, M., Mueller, M. M., Bedke, J., Kartenbeck, J., et al. (2005). Physical and biological characterization of superparamagnetic iron oxide- and ultrasmall superparamagnetic iron oxide-labeled cells: A comparison. Investigative Radiology, 40, 504–513.
Metz, S., Bonaterra, G., Rudelius, M., Settles, M., Rummeny, E. J., & Daldrup-Link, H. E. (2004). Capacity of human monocytes to phagocytose approved iron oxide MR contrast agents in vitro. European Radiology, 14, 1851–1858.
Linker, R. A., Kroner, A., Horn, T., Gold, R., Maurer, M., & Bendszus, M. (2006). Iron particle-enhanced visualization of inflammatory central nervous system lesions by high resolution: Preliminary data in an animal model. AJNR. American Journal of Neuroradiology, 27, 1225–1229.
Henning, T. D., Boddington, S., & Daldrup-Link, H. E. (2008). Labeling hESCs and hMSCs with iron oxide nanoparticles for non-invasive in vivo tracking with MR imaging. Journal of Visualized Experiments, 31, 685. http://www.ncbi.nlm.nih.gov/pubmed/19066574.
Hsiao, J. K., Tai, M. F., Chu, H. H., Chen, S. T., Li, H., Lai, D. M., et al. (2007). Magnetic nanoparticle labeling of mesenchymal stem cells without transfection agent: Cellular behavior and capability of detection with clinical 1.5 T magnetic resonance at the single cell level. Magnetic Resonance in Medicine, 58, 717–724.
Wang, L., Deng, J., Wang, J., Xiang, B., Yang, T., Gruwel, M., et al. (2009). Superparamagnetic iron oxide does not affect the viability and function of adipose-derived stem cells, and superparamagnetic iron oxide-enhanced magnetic resonance imaging identifies viable cells. Magnetic Resonance Imaging, 27, 108–119.
Bulte, J. W., Duncan, I. D., & Frank, J. A. (2002). In vivo magnetic resonance tracking of magnetically labeled cells after transplantation. Journal of Cerebral Blood Flow and Metabolism, 22, 899–907.
Guzman, R., Uchida, N., Bliss, T. M., He, D., Christopherson, K. K., Stellwagen, D., et al. (2007). Long-term monitoring of transplanted human neural stem cells in developmental and pathological contexts with MRI. Proceedings of the National Academy of Sciences of the United States of America, 104, 10211–10216.
George, A. J., Bhakoo, K. K., Haskard, D. O., Larkman, D. J., & Reynolds, P. R. (2006). Imaging molecular and cellular events in transplantation. Transplantation, 82, 1124–1129.
Acknowledgments
This research was supported by the Natural science fund for colleges and universities in Jiangsu Province (no. 06KJB320097). Mr. Wenbing Li read this manuscript and gave some comments.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jing Guo and Jun-Kang Shen have contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Guo, J., Shen, JK., Wang, L. et al. In Vivo Evaluation of Cerebral Transplantation of Resovist-Labeled Bone Marrow Stromal Cells in Parkinson’s Disease Rats Using Magnetic Resonance Imaging. Appl Biochem Biotechnol 163, 636–648 (2011). https://doi.org/10.1007/s12010-010-9069-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-010-9069-y