Abstract
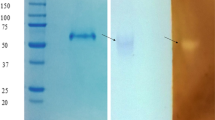
Three mutations, Ser54→Pro, Thr314→Ala, and His415→Tyr, were identified in Aspergillus awamori glucoamylase gene expressed by Saccharomyces cerevisiae. The mutant glucoamylase (GA) was substantially more thermostable than a wild-type GA at 70 °C, with a 3.0 KJ mol−1 increase in the free energy of thermo-inactivation. The effect of starch from different botanical sources on the production of this GA was measured in liquid fermentation using commercial soluble starch, cassava, potato, and corn as the carbon source. The best substrate for GA production was the potato starch showing an enzymatic activity of 6.6 U/mL. The commercial soluble starch was also a good substrate for the enzyme production with 6.3 U/mL, followed by cassava starch and corn starch with 5.9 and 3.0 U/mL, respectively. These results showed a significant difference on GA production related to the carbon source employed. The mutant GA was purified by acarbose–Sepharose affinity chromatography; the estimated molecular mass was 100 kDa. The mutant GA exhibited optimum activity at pH 4.5 and an optimum temperature of 65 °C.




Similar content being viewed by others
References
Roy, I., & Gupta, M. N. (2004). Enzyme and Microbial Technology, 34, 26–32.
Uthumporn, U., Zaidul, I. S. M., & Karim, A. A. (2010). Food and Bioproducts Processing, 88, 47–54.
Mertens, J. A., & Skory, C. D. (2007). Enzyme and Microbial Technology, 40, 874–880.
Michelin, M., Ruller, R., Ward, R. J., Moraes, L. A. B., Jorge, J. A., Terenzi, H. F., et al. (2008). Journal of Industrial Microbiology & Biotechnology, 35, 17–25.
McDaniel, A., Fuchs, E., Liu, Y., & Ford, C. (2008). Microbial Technology, 1(6), 523–531.
Wang, Y., Fuchs, E., da Silva, R., McDaniel, A., Seibel, J., & Ford, C. (2006). Starch/Stärke, 58, 501–508.
James, J. A., & Lee, B. H. (1997). Journal of Food Biochemistry, 21, 1–52.
Ford, C. (1999). Current Opinion in Biotechnology, 10, 353–357.
Liu, H.-L., & Wang, W.-C. (2003). Protein Engineering, 16, 19–25.
Kilonzo, P. M., Margaritis, A., & Bergougnou, M. A. (2009). Journal of Biotechnology, 143, 60–68.
Naessens, M., & Vandamme, E. J. (2003). Biotechnological Letters, 25, 1119–1124.
Swift, R. J., Karandikar, A., Griffen, A. M., Punt, P. J., Hondel, C. A. M. J. J., Robson, G. D., et al. (2000). Fungal Genetics and Biology, 32, 125–133.
Peres, M. F. S., Souza, C. S., Thomaz, D., de Souza, A. R., & Laluce, C. (2006). Process Biochemistry, 41, 77–83.
Pavezzi, F. C., Gomes, E., & da Silva, R. (2008). Brazilian Journal of Microbiology, 39, 108–114.
Lemos, C. M. (2003). M.Sc. Dissertation, UNESP, Rio Claro, Brazil.
Hoffmam, C. S., & Winston, F. (1987). Gene, 57, 267–272.
Bergmeyer, H. U., & Bernt, E. (1974). Methods of enzymatic analysis. Methods, 3, 1205–1215.
Hartree, E. F. (1972). Analytical Biochemistry, 48, 422–427.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Blum, H., Bier, H., & Gross, H. J. (1987). Eletrophoresis, 8(2), 93–99.
Chen, H. M., Bakir, U., Reilly, P. C., & Ford, C. (1994). Biotechnology and Bioengineering, 43, 101–105.
Leite, R. S. R., Alves-Prado, H. F., Cabral, H., Pagnocca, F. C., Gomes, E., & Da Silva, R. (2008). Enzyme and Microbial Technology, 43, 391–395.
Suzuki, Y., Hatagaki, K., & Oda, H. (1991). Applied Microbiology and Biotechnology, 34, 707–714.
Liu, H.-L., Doleyres, Y., Coutinho, P. M., Ford, C., & Reilly, P. J. (2000). Protein Engineering, 13, 655–659.
Li, Y., Reilly, P. J., & Ford, C. (1997). Protein Engineering, 10, 1199–1204.
Allen, M. J., Coutinho, P. M., & Ford, C. (1998). Protein Engineering, 11, 783–788.
Gomes, E., Guez, M. A. U., Martin, N., & da Silva, R. (2007). Química Nova, 30, 136–145.
Li, Y., Coutinho, P. M., & Ford, C. (1998). Protein Engineering, 11, 661–667.
Chen, H. M., Li, Y., Panda, T., Buehler, F. U., Ford, C., & Reilly, P. J. (1996). Protein Engineering, 9, 499–505.
Norouzian, D., Akbarzadeh, A., Scharer, J. M., & Young, M. M. (2006). Research Review Paper, 24, 80–85.
Anto, H., Trivedi, U. B., & Patel, K. C. (2006). Bioresource Technology, 97, 1161–1166.
Hata, Y., Ishida, H., Kojima, Y., Ichikawa, E., Kawato, A., Suginami, K., et al. (1997). Journal of Fermentation Bioengineering, 84, 532–537.
Munch, O., & Tritsch, D. (1990). Biochimica et Biophysica Acta, 1041, 111–116.
Bruins, M. E., Janssen, A. E. M., & Boom, R. M. (2001). Applied Biochemistry and Biotechnology, 90, 155–181.
Flory, N., Gorman, M., Coutinho, P. M., Ford, C., & Reilly, P. J. (1994). Protein Engineering, 7, 1005–1012.
Carrea, G., & Colombo, G. (2000). Trends in Biotechnology, 18, 401–402.
Bakir, U., Coutinho, P. M., Sullivan, P. A., Ford, C., & Reilly, P. J. (1993). Protein Engineering, 6, 939–946.
Withers, J. M., Swift, R. J., Wiebe, M. G., Robson, G. D., Punt, P. J., van den Hondel, C. A. M. J. J., et al. (1998). Biotechnology and Bioengineering, 59, 407–418.
Latorre-Garcia, L., Adam, A. C., & Polaina, J. (2008). World Journal of Microbiology & Biotechnology, 24, 2957–2963.
Vanomi, M., Lotti, M., & Alberghina, L. (1989). Biochimica et Biophysica Acta, 1008, 168–176.
Ghang, D. M., Yu, L., Lim, M. H., Ko, H. M., Im, S. Y., Lee, H. B., et al. (2007). Biotechnological Letters, 29, 1203–1208.
González, C. F., Fariña, J. I., & de Figueroa, L. I. C. (2008). Enzyme and Microbial Technology, 42, 272–277.
Kilonzo, P. M., Margaritis, A., & Bergougnou, M. A. (2008). Journal of Biotechnology, 114(2), 83–95.
Slavik, J., & Kotyk, A. (1984). Biochimica et Biophysica Acta, 766(3), 679–684.
Watanabe, T., Furukawa, S., Kitamoto, K., Takatsuki, A., Hirata, R., Ogihara, H., et al. (2005). International Journal of Food Microbiology, 105, 131–137.
Piper, P., Calderon, C. O., Hatzixanthis, K., & Mollapour, M. (2001). Microbiology, 147, 2635–2642.
Schuller, C., Schüller, C., Mamnun, Y. M., Mollapour, M., Krapf, G., Schuster, M., et al. (2004). Molecular Biology of the Cell, 15, 706–720.
Giannattasioa, S., Guaragnellaa, N., Corte-Realb, M., Passarellac, S., & Marraa, E. (2005). Gen, 354, 93–98.
Brul, S., & Coote, P. (1999). International Journal of Food Microbiology, 50, 1–17.
Halm, M., Hornbaek, T., Arneborg, N., Sefa-Dedeh, S., & Jespersen, L. (2004). International Journal of Food Microbiology, 94, 97–103.
Vihinen, M., & Mantsala, P. (1989). Critical Reviews in Biochemistry and Molecular Biology, 24, 329–418.
Zidehsaraei, A. Z., Moshkelani, M., & Amiri, M. C. (2009). Separation and Purification Technology, 67, 8–13.
Ouyang, A., Benneu, P., Zhang, A., & Yang, S. T. (2007). Process Biochemistry, 42, 561–569.
Acknowledgments
The authors are grateful to the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Conselho Nacional de Pesquisa (CNPq) for their financial support. We are also gratefull to Dr. Clark Ford from Iowa State University for his teachings in this area and for donating our firts mutants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pavezzi, F.C., Carneiro, A.A.J., Bocchini-Martins, D.A. et al. Influence of Different Substrates on the Production of a Mutant Thermostable Glucoamylase in Submerged Fermentation. Appl Biochem Biotechnol 163, 14–24 (2011). https://doi.org/10.1007/s12010-010-8963-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-010-8963-7