Abstract
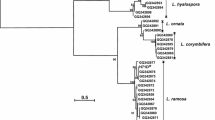
Alternaria sp. ND-16, a bacterium isolated from soil sample, was identified as a strain of Alternaria mali based on the morphology and comparison of internal transcribed spacer rDNA gene sequence studies. Furthermore, it is demonstrated that this strain has xylanase activity, and the activity can be optimized under suitable growing conditions where wheat bran and urea are the primary sources of carbon and nitrogen. Partially purified xylanase from Alternaria sp. ND-16 is shown to have an optimal pH of 6.0 and optimal temperature of 50 °C, making this enzyme potentially suitable for industrial applications. It is also demonstrated that Na+ and Mn2+ show strong inhibition of the xylanase while K+, Li+, Fe2+, Cu2+, and Zn2+ have no significant effect on the activity.






Similar content being viewed by others
References
Polizeli, M. L. T. M., Rizzatti, A. C. S., Monti, R., Terenzi, H. F., Jorge, J. A., & Amorim, D. S. (2005). Xylanase from fungi: Properties and industrial applications. Applied Microbiology and Biotechnology, 67, 577–591.
Vicuna, R., Oyarzum, E., & Osses, M. (1995). Assessment of various commercial enzymes in the bleaching of radiata pine kraft pulps. Journal of Biotechnology, 40, 163–168.
Silversides, F. G., Scott, T. A., Korver, D. R., Afsharmanesh, M., & Hruby, M. (2006). A study on the interaction of xylanase and phytase enzymes in wheat-based diets fed to commercial white and brown egg laying hens. Poultry Science, 85, 297–305.
Maat, J., Roza, M., Verbakel, J., Stam, H., DaSilra, M. J. S., Egmond, M. R., et al. (1992). Xylanases and their application in bakery. In J. Visser, G. Beldman, M. A. K. van Someren, & A. G. J. Voragen (Eds.) Xylans and xylanases (pp. 349–360). Amsterdam: Elsevier.
Jiang, Z. Q., Yang, S. Q., Tan, S. S., Li, L. T., & Li, X. T. (2005). Characterization of a xylanase from the newly isolated thermophilic thermomyces lanuginosus CAU44 and its application in bread making. Letters in Applied Microbiology, 41, 69–76.
Li, Y., Lu, J., Gu, G. X., & Mao, Z. (2005a). Characterization of the enzymatic degradation of arabinoxylans in grist containing wheat malt using response surface methodology. Journal of the American Society of Brewing Chemists, 63, 171–176.
Lu, J., & Li, Y. (2006). Effects of arabinoxylan solubilization on wort viscosity and filtration when mashing with grist containing wheat and wheat malt. Food Chemisty, 98, 164–170.
Lu, J., Li, Y., Gu, G. X., & Mao, Z. (2005). Effects of molecular weight and concentration of arabinoxylans on the membrane plugging. Journal of Agricultural and Food Chemistry, 53, 4996–5002.
Shah, A. R., & Madamwar, D. (2005). Xylanase production by a newly isolated Aspergillus foetidus strain and its characterization. Process Biochemistry, 40, 1763–1771.
Li, Y., Cui, F., Liu, Z., Xu, Y., & Zhao, H. (2007a). Improvement of xylanase production by Penicillium oxalicum ZH-30 using response surface methodology. Enzyme Microbiology Technology, 40, 1381–1388.
Li, Y., Liu, Z., Zhao, H., Xu, Y., & Cui, F. (2007b). Statistical optimization of xylanase production from new isolated Penicillium oxalicum ZH-30 in submerged fermentation. Biochemical Engineering Journal, 34, 82–86.
Azin, M., Moravej, R., & Zareh, D. (2007). Production of xylanase by Trichoderma longibrachiatum on a mixture of wheat bran and wheat straw: Optimization of culture condition by Taguchi method. Enzyme Microbiology Technology, 40, 801–805.
Abrusci, C., Martín-González, A., Del Amo, A., Catalina, F., Collado, J., & Platas, G. (2005). Isolation and identification of bacteria and fungi from cinematographic films. International Biodeterioration and Biodegradation, 56, 58–68.
Li, Y., Liu, Z., Cui, F., Xu, Y., & Zhao, H. (2007c). Production of xylanase from a newly isolated Penicillium sp. ZH-30. World Journal of Microbiology and Biotechnology, 23, 837–843.
Doyle, J. J., & Doyle, J. L. (1987). A rapid DNA isolation procedure for small quantities of fresh leaf tissues. Phytochemical Bulletin, 19, 11–15.
Lee, S. B., & Taylor, J. W. (1990). Isolation of DNA from fungal mycelia and single spores. In M. A. Innis, D. H Gelfand, J. S. Sninsky, & T. J. White (Eds.) PCR protocols: A guide to methods and applications (pp. 282–287). New York: Academic.
Liu, Z. Q., & Sun, Z. H. (2004). Cloning and expression of D-lactonohydrolase cDNA from Fusarium moniliforme in Saccharomyces cerevisiae. Biotech Letters, 26, 1861–1865.
Liu, Z. Q., Li, Y., Xu, Y. Y., Ping, L. F., & Zheng, Y. G. (2007). Cloning, sequencing, and expression of a novel epoxide hydrolase gene from Rhodococcus opacus in Escherichia coli and characterization of enzyme. Applied Microbiology and Biotechnology, 74, 99–106.
Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugars. Analytical Chemistry, 31, 426–428.
Nascimento, R. P., Coelho, R. R. R., Marques, S., Alves, L., Girio, F. M., Bon, E. P. S., et al. (2002). Production and partial characterisation of xylanase from Streptomyces sp. strain AMT-3 isolated from Brazilian cerrado soil. Enzyme Microbiology Technology, 31, 549–555.
Ghosh, M., Das, A., Mishra, A. K., & Nanda, G. (1993). Aspergillus sydowii MG 49 is a strong producer of thermostable xylanolytic enzyme. Enzyme Microbiology Technology, 15, 703–709.
Liu, Z. Q., Li, Y., Ping, L. F., Xu, Y. Y., Cui, F. J., Xue, Y. P., et al. (2007). Isolation and identification of a novel Rhodococcus sp. ML-0004 producing epoxide hydrolase and optimization of enzyme production. Process Biochemistry, 42, 889–894.
Gardes, M., & Bruns, T. D. (1993). ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Molecular Ecology, 2, 113–118.
Liu, Z., Hu, Z., Zheng, G., & Shen, Y. (2008). Optimization of cultivation conditions for the production of 1,3-dihydroxyacetone by Pichia membranifaciens using response surface methodology. Biochemical Engineering Journal, 38, 285–291.
Erland, S., Henrion, B., Martin, F., Glover, L. A., & Alexander, I. J. (1994). Identification of the ectomycorrhizal basidiomycete tylospora-fibrillosa donk by RFLP analysis of the PCR-amplified ITS and IGS regions of ribosomal DNA. New Phytologist, 126, 525–532.
Liu, Z. Q., Zhang, J. F., Zheng, Y. G., & Shen, Y. C. (2008). Production of astaxanthin from a newly isolated phaffia rhodozyma mutant by low-energy ions beam implantation. Journal of Applied Microbiology, 108, 861–872.
Filajdic, N., & Sutton, T. B. (1991). Identification and distribution of Alternaria mali on apples in North Carolina and susceptibility of different varieties of apples to alternaria blotch. Plant Disease, 75, 1045–1048.
Oliveir, L. A., Porto, A. L. F., & Tambourgi, E. B. (2006). Production of xylanase and protease by Penicillium janthinellum CRC 87M-115 from different agricultural wastes. Bioresource Technology, 97, 862–867.
Li, Y., Lin, J., Lu, J., Gu, G., & Mao, Z. (2006). Effect of pH, cultivation time and substrate concentration on the xylanase production by Aspergillus awamori ZH-26 under submerged fermentation using central composite rotary design. Food Technology and Biotechnology, 44, 473–477.
Yang, S. Q., Yan, Q. J., Jiang, Z. Q., Li, L. T., Tian, H. M., & Wang, Y. Z. (2006). High-level of xylanase production by the thermophilic Paecilomyces themophila J18 on wheat straw in solid-state fermentation. Bioresource Technology, 97, 1794–1800.
Kang, S. W., Park, Y. S., Lee, J. S., Hong, S. I., & Kim, S. W. (2004). Production of cellulases and hemicellulases by Aspergillus niger KK2 from lignocellulosic biomass. Bioresource Technology, 91, 153–156.
Sermanni, G. G., Annibale, A., Lena, G. D., Vitale, N. S., Mattia, E. D., & Minelli, V. (1994). The production of exo-enzymes by Lentinus edodes and pleurotus ostreatus and their use for upgrading corn straw. Bioresource Technology, 48, 173–178.
Cai, Q., Yue, X., Niu, T., Ji, C., & Ma, Q. (2004). The screening of culture condition and properties of xylanase by white-rot fungus Pleurotus ostreatus. Process Biochemistry, 39, 1561–1566.
Leathers, T. D., Detroy, R. W., & Bothast, R. J. (1986). Induction and glucose repression of xylanase from a color variant strain of Aureobasidium pullulans. Biotechnology Letters, 8, 867–872.
Singh, A., Kumar, P. K. R., & Schtigerl, K. (1992). Bioconversion of cellulosic materials to ethanol by filamentous fungi. Advances in Biochemical Engineering, Biotechnology, 45, 29–55.
Liu, W., Lu, Y. L., & Ma, G. R. (1999). Induction and glucose repression of endo-β-xylanase in the yeast Trichosporon cutaneum SL409. Process Biochemistry, 34, 67–72.
Ito, K. (1993). Acid stable xylanase from Aspergillus kawachii. J Brew Soc Japan, 88, 920–928.
Kulkarni, N., Shendye, A., & Rao, M. (1999). Molecular and biotechnological aspects of xylanases. FEMS Microbiology Reviews, 23, 411–456.
Haltrich, D., Nidetzky, B., Kulbe, K. D., Steiner, W., & Zupancic, S. (1996). Production of fungal xylanases. Bioresource Technology, 58, 137–161.
Dobrev, G. T., Pishtiyski, I. G., Stanchev, V. S., & Mircheva, R. (2007). Optimization of nutrient medium containing agricultural wastes for xylanase production by Aspergillus niger B03 using optimal composite experimental design. Bioresource Technology, 98, 2671–2678.
Li, Y., Lu, J., & Gu, G. (2005b). Control of arabinoxylan solubilization and hydrolysis in mashing. Food Chemistry, 90, 101–108.
Miyazaki, K., Takenouchi, M., Kondo, H., Noro, N., Suzuki, M., & Tsuda, S. (2006). Thermal stabilization of Bacillus subtilis Family-11 xylanase by directed evolution. Journal of Biological Chemistry, 281, 10236–10242.
Liu, Z. Q., Sun, Z. H., & Leng, Y. (2006). Directed evolution of D-pactonohydrolase from Fusarium moniliforme. Journal of Agricultural and Food Chemistry, 54, 5823–5830.
Acknowledgements
The authors greatly appreciate the help of Dr. Peter Baker of the Department of Chemical and Biological Science, Polytechnic University, USA, for his kindness in editing this manuscript. This work was supported by the Natural Science Foundation of Zhejiang Province (No. Y506136).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, Y., Liu, Z., Cui, F. et al. Isolation and Identification of a Newly Isolated Alternaria sp. ND-16 and Characterization of Xylanase. Appl Biochem Biotechnol 157, 36–49 (2009). https://doi.org/10.1007/s12010-008-8239-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8239-7