Abstract
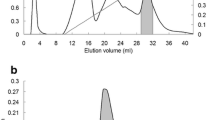
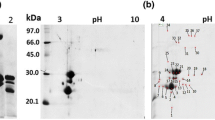
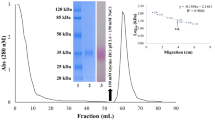
A lectin-like protein from the seeds of Acacia farnesiana was isolated from the albumin fraction, characterized, and sequenced by tandem mass spectrometry. The albumin fraction was extracted with 0.5 M NaCl, and the lectin-like protein of A. farnesiana (AFAL) was purified by ion-exchange chromatography (Mono-Q) followed by chromatofocusing. AFAL agglutinated rabbit erythrocytes and did not agglutinate human ABO erythrocytes either native or treated with proteolytic enzymes. In sodium dodecyl sulfate gel electrophoresis under reducing and nonreducing conditions, AFAL separated into two bands with a subunit molecular mass of 35 and 50 kDa. The homogeneity of purified protein was confirmed by chromatofocusing with a pI = 4.0 ± 0.5. Molecular exclusion chromatography confirmed time-dependent oligomerization in AFAL, in accordance with mass spectrometry analysis, which confers an alteration in AFAL affinity for chitin. The protein sequence was obtained by a liquid chromatography quadrupole time-of-flight experiment and showed that AFAL has 68% and 63% sequence similarity with lectins of Phaseolus vulgaris and Dolichos biflorus, respectively.










Similar content being viewed by others
Abbreviations
- AFAL:
-
Acacia farnesiana lectin-like protein
- VL:
-
vinorama lectin
- GlcNac:
-
N-acetyl-glucosamine
- PPL-1:
-
Parkia platycephala lectin
- PPL-2:
-
Parkia platycephala chimerolectin
- bCFL:
-
Cratylia floribunda lectin
- BSA:
-
bovine serum albumin
- SDS-PAGE:
-
sodium dodecyl sulfate polyacrylamide gel electrophoresis
- HPLC:
-
high-pressure liquid chromatography
- TFA:
-
trifluoroacetic acid
- EDTA:
-
ethylenediaminetetracetic acid
- DTT:
-
dithiothreitol
- PTH:
-
phenylthiohydantoin
- PHAL:
-
Phaseolus vulgaris leukocyte agglutinin
- DBL:
-
Dolichos biflorus lectin
- PDL:
-
Parkia discolor lectin
- PSL:
-
Parkia speciosa lectin
- PJL:
-
Parkia javanica lectin
References
Goldstein, I. J., Hughes, R. C., Monsigny, M., Osawa, T., & Sharon, N. (1980). Nature, 285, 66.
Van Damme, E. J. M., Peumans, W. J., Barre, A., & Rougé, P. (1998). Critical Reviews in Plant Sciences, 17, 575–692.
Guzman-Partida, A. M., Robles-Burgueno, M. R., Ortega-Nieblas, M., & Vazquez-Moreno, I. (2004). Biochimie, 86, 335–342.
Etzler, M. E. (1986). The lectins: Properties, functions and applications in biology and medicine. In I. E. Liener, N. Sharon, & I. J. Goldstein (Eds.) Distribution and function of plant lectins (pp. 371–435). New York: Academic.
Peumans, W. J., & Van Damme, E. J. (1995). Plant Physiology, 109, 347–352.
Helenius, A., Trombetta, E. S., Herbert, D. N., & Simons, J. F. (1997). Trends Cell Biology, 7, 193–200.
Sharon, N., & Lis, H. (2003). Lectins. Dordrecht, The Netherlands: Kluwer.
Imberty, A., Casset, F., Gegg, C. V., Etzler, M. E., & Perez, S. (1994). Glycoconjugate Journal, 11, 400–413.
Hamelryck, T. W., Moore, J. G., Chrispeels, M. J., Loris, R., & Wyns, L. (2000). Journal of Molecular Biology, 299, 875–883.
Loris, R., Hamelryck, T., Bouckaert, J., & Wyns, L. (1998). Biochimica et Biophysica Acta, 1383, 9–36.
Young, N. M., & Oomen, R. P. (1992). Journal of Molecular Biology, 228, 924–934.
Sharma, V., & Surolia, A. J. (1997). Molecular Biology, 267, 433–445.
Bouckaert, J., Loris, R., Poortmans, F., & Wyns, L. (1995). Genetics, 23, 510–540.
Bouckaert, J., Dewallef, Y., Poortmans, F., Wyns, L., & Loris, R. (2000). Journal of Biological Chemistry, 275, 19778–19787.
Lescar, J., Loris, R., Mitchell, E., Gautier, C., Chazalet, V., Cox, V., et al. (2002). Journal of Biological Chemistry, 277, 6608–6614.
Patil, R. S., Ghormade, V. V., & Deshpande, M. V. (2000). Enzyme Microbial Technology, 26, 473–483.
Suvachittanont, W., & Peutpaiboon, A. (1992). Phytochemistry, 319, 4065–4070.
Utarabhand, P., & Akkayanont, P. (1995). Phytochemistry, 38, 281–285.
Cavada, B. S., Santos, C. F., Grangeiro, T. B., Silva, L. I. M. M., Campos, M. J. O., Sousa, F. A. M., & Calvete, J. (1997). Physiology and Molecular Biology of Plants, 3, 109–115.
Cavada, B. S., Madeira, S. V. F., Calvete, J. J., Sousa, L. A. G., Bomfim, L. R., Dantas, A. R., et al. (2000). Preparative Biochemistry & Biotechnology, 30, 271–280.
Cavada, B. S., Castellón, R. E. R., Vasconcelos, G. G., Rocha, B. A. M., Bezerra, G. A., Debray, H., et al. (2005). Acta Crystallographica Section F, 61, 841–843.
Gallego Del Sol, F., Nagano, C., Cavada, B. S., & Calvete, J. J. (2005). Journal of Molecular Biology, 353, 574–583.
Gallego del Sol, F., Gomez, J., Hoos, S., Nagano, C. S., Cavada, B. S., England, P., et al. (2005). Acta Crystallographica Section F, 61, 326–331.
Cavada, B. S., Moreno, F. B., Rocha, B. A. M., Azevedo Jr., W. F. , Castellon, R. E., Goersch, G. V., et al. (2006). FEBS Journal, 273, 3962–3974.
Calvete, J. J., Thole, H. H., Raida, M., Urbanke, C., Romero, A., Grangeiro, T. B., et al. (1999). Biochimica et Biophysica Acta, 1430, 367–375.
Gallego Del Sol, F., Cavada, B. S., & Calvete, J. J. (2006). Journal of Structural Biology, 158, 1–9.
Bradford, M. M. A. (1976). Biochemistry, 72, 248–254.
Vázquez-Moreno, L., Ortega-Nieblas, M., Robles-Burgueño, M. R., & Ramos-Clamont, G. (2000). I.J.B.C, 5, 1–8.
Raghava, G. P. S. (2000). Comparative Asian Studies Programme, 4, 75–76.
Van Damme, E. J. M., Culerrier, R. I., Barre, A., Alvarez, R., Rougè, P., & Peumans, W. J. (2007). Plant Physiology, 144, 662–672.
Hemperly, J. J., & Cunningham, B. A. (1983). Trends in Biochemical Sciences, 5, 100–102.
Hamelryck, T. W., Dao-Thi, M. H., Poortmans, F., Chrispeels, M. J., Wyns, L., & Loris, R. J. (1996). Biological Chemistry, 271, 20479–20485.
Lioi, L., Sparvoli, F., Galasso, I., Lanave, C., & Bollini, R. (2003). Theoretical and Applied Genetics, 107, 814–822.
Hamelryck, T. W., Loris, R., Bouckaert, J., Dao-Thi, M. H., Strecker, G., Imberty, A., et al. (2000). Journal of Biological Chemistry, 286, 1161–1177.
Allen, H. J., & Johnson, A. Z. (1977). Carbohydrate Research, 58, 253–265.
Anantharam, V., Patanjali, S. R., Swamy, M. J., Sanadi, A. R., Goldstein, I. J., & Surolia, A. (1986). Journal of Biological Chemistry, 261, 14621–14627.
Peumans, W. J., Barre, A., Hao, Q., Rougé, P., & Van Damme, E. J. M. (2000). Trends in Glycoscience and Glycotechnology, 12, 83–101.
Wah, D. A., Romero, A., Gallego del Sol, F., Cavada, B. S., Ramos, M. V., Grangeiro, T. B., et al. (2001). Journal of Molecular Biology, 310, 885–894.
Acknowledgments
This work was partly financed by Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Laboratório de Espectrometria de Massas/LNLS, Campinas—Brazil, and FAPESP (SMOLBNet, 01/07532-0). BSC and AHS are senior investigators of CNPq. We also thank Dr. A. Leyva for English language editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santi-Gadelha, T., Rocha, B.A.M., Oliveira, C.C. et al. Purification of a PHA-Like Chitin-binding Protein from Acacia farnesiana Seeds: A Time-dependent Oligomerization Protein. Appl Biochem Biotechnol 150, 97–111 (2008). https://doi.org/10.1007/s12010-008-8144-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8144-0