Abstract
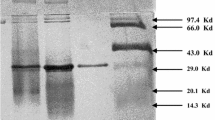
Cyclodextrin glucanotransferase, produced by Bacillus megaterium, was characterized, and the biochemical properties of the purified enzyme were determined. The substrate specificity of the enzyme was tested with different α-1,4-glucans. Cyclodextrin glucanotransferase displayed maximum activity in the case of soluble starch, with a K m value of 3.4 g/L. The optimal pH and temperature values for the cyclization reaction were 7.2 and 60 °C, respectively. The enzyme was stable at pH 6.0–10.5 and 30 °C. The enzyme activity was activated by Sr2+, Mg2+, Co2+, Mn2+, and Cu2+, and it was inhibited by Zn2+and Ag+. The molecular mass of cyclodextrin glucanotransferase was established to be 73,400 Da by sodium dodecyl sulfate–polyacrylamide gel electrophoresis, 68,200 Da by gel chromatography, and 75,000 Da by mass spectrometry. The monomer form of the enzyme was confirmed by the analysis of the N-terminal amino acid sequence. Cyclodextrin glucanotransferase formed all three types of cyclodextrins, but the predominant product was β-cyclodextrin.





Similar content being viewed by others
References
Tonkova, A. (1998). Enzyme and Microbial Technology, 22, 678–686.
Biwer, A., Antranikian, G., & Heinzle, E. (2002). Applied Microbiology and Biotechnology, 59, 609–617.
Singh, M., Sharma, R., & Banerjee, U. (2002). Biotechnology Advances, 20, 341–359.
Valle, E. (2004). Process Biochemistry, 39, 1033–1046.
Choi, J., Lee, J., Choi, K., & Kwon, I. (1996) .US Patent 5492829.
Gawande, B., & Patkar, A. (1999). Biotechnology and Bioengineering, 64, 168–173.
Jemli, S., Messaoud, E., Ayadi-Zouari, D., Naili, B., Khemakhem, B., & Bejar, S. (2007). Biochemical Engineering Journal, 34, 44–50.
Mori, S., Hirose, S., & Oya, T. (1994). Bioscience, Biotechnology, and Biochemistry, 58, 1968–1972.
Tachibana, Y., Kuramura, A., Shirasaka, N., Suzuki, Y., Yamamoto, T., & Fujiwara, S., et al. (1999). Applied and Environmental Microbiology, 65, 1991–1997.
Wind, R., Buitelaar, R., & Dijkuizen, L. (1998). European Journal of Biochemistry, 253, 598–605.
Prowe, S., & Antranikian, G. (2001). International Journal of Systematic and Evolutionary Microbiology, 51, 457–465.
Abelyan, V., Afyan, K., & Manukyan, L. (2000). Applied Biochemistry and Microbiology, 36, 338–343.
Yamamoto, K., Zhang, Z., & Kobayashi, S. (2000). Journal of Agricultural and Food Chemistry, 48, 962–966.
Vassileva, A., Burhan, N., Beschkov, V., Spasova, D., Radoevska, S., & Ivanova, V., et al. (2003). Process Biochemistry, 28, 1585–1591.
Martins, R., & Hatti-Kaul, R. (2002). Enzyme and Microbial Technology, 30, 116–124.
Gawande, B., Goel, A., Patkar, A., & Nene, S. (1999). Applied Microbiology and Biotechnology, 51, 504–509.
Qi, Q., & Zimmermann, W. (2005). Applied Microbiology and Biotechnology, 66, 475–485.
Popova, V., & Pishtiyski, I. (2001). European Food Research and Technology, 213, 67–71.
Cornish-Bowden, A. (1995). Fundamentals of enzyme kinetics. London: Portland Press.
Laemli, U. (1970). Nature, 227, 680–685.
Kestner, A., Vokk, R., Papel, E., & Papeman, A. (1989). Prikladnaja Biochimia i Microbiologia, 25, 425–430 (in Russian).
Gawande, B., & Patkar, A. (2001). Starch/Stärke, 53, 75–83.
Grüll, D., & Stifter, U. (2001).US Patent 6235505.
Shiraishi, K., Kawakami, K., Marushima, H., & Kusunoki, K. (1989). Starch/Stärke, 41, 151–155.
Chang, W., & Koo, Y. (2000). Biotechnology Letters, 22, 651–654.
Tomita, K., Tanaka, T., Fujita, Y., & Nakanishi, K. (1990). Journal of Fermentation and Bioengineering, 70, 190–192.
Kim, T., Lee, Y., & Kim, H. (1993). Biotechnology and Bioengineering, 41, 88–94.
Matioli, G., Zanin, G., & Moraes, F. (2001). Applied Biochemistry and Biotechnology, 91–93, 643–654.
Cao, X., Jin, Z., Wang, X., & Chen, F. (2005). Food Research International, 38, 309–314.
Sian, H., Said, M., Hassan, O., Kamaruddin, K., Ismail, A., & Rahman, R., et al. (2005). Process Biochemistry, 40, 1101–1111.
Larsen, K., Duedahl-Olesen, L., Jørgen, H., Christensen, S., Mathiesen, F., & Pedersen, L., et al. (1998). Carbohydrate Research, 310, 211–219.
Gawande, B., & Patkar, A. (2001). Enzyme and Microbial Technology, 28, 735–743.
Thiemann, V., Dönges, C., Prowe, S., Sterner, R., & Antranikian, G. (2004). Archives of Microbiology, 182, 226–235.
Fujivara, S., Kakhara, H., Sakagichi, K., & Imanaka, T. (1992). Journal of Bacteriology, 174, 7478–7481.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pishtiyski, I., Popova, V. & Zhekova, B. Characterization of Cyclodextrin Glucanotransferase Produced by Bacillus megaterium . Appl Biochem Biotechnol 144, 263–272 (2008). https://doi.org/10.1007/s12010-007-8009-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-007-8009-y