Abstract
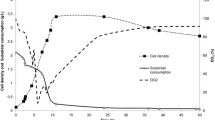
Bacillus subtilis strain TrigoCor 1448 was grown on wheat middlings in 0.5-l solid-state fermentation (SSF) bioreactors for the production of an antifungal biological control agent. Total antifungal activity was quantified using a 96-well microplate bioassay against the plant pathogen Fusarium oxysporum f. sp. melonis. The experimental design for process optimization consisted of a 26−1 fractional factorial design followed by a central composite face-centered design. Initial SSF parameters included in the optimization were aeration, fermentation length, pH buffering, peptone addition, nitrate addition, and incubator temperature. Central composite face-centered design parameters included incubator temperature, aeration rate, and initial moisture content (MC). Optimized fermentation conditions were determined with response surface models fitted for both spore concentration and activity of biological control product extracts. Models showed that activity measurements and spore production were most sensitive to substrate MC with highest levels of each response variable occurring at maximum moisture levels. Whereas maximum antifungal activity was seen in a limited area of the design space, spore production was fairly robust with near maximum levels occurring over a wider range of fermentation conditions. Optimization resulted in a 55% increase in inhibition and a 40% increase in spore production over nonoptimized conditions.






Similar content being viewed by others
References
Yamada, S., Takayama, Y., Yamanaka, M., Ko, K., & Yamaguchi, I. (1990). Journal of Pesticide Science, 15, 95–96.
Vanittanakom, N., Loeffler, W., Koch, U., & Jung, G. (1986). Journal of Antibiotics, 39, 888–901.
Marten, P., Smalla, K., & Berg, G. (2000). Journal of Applied Microbiology, 89, 463–471.
Leenders, F., Stein, T. H., Kablitz, B., Franke, P., & Vater, J. (1999). Rapid Communications in Mass Spectrometry, 13, 943–949.
Ohno, A., Ano, T., & Shoda, M. (1995). Journal of Fermentation and Bioengineering, 80, 517–519.
Toure, Y., Ongena, M., Jacques, P., Guiro, A., & Thonart, P. (2004). Journal of Applied Microbiology, 96, 1151–1160.
Akpa, E., Jacques, P., Wathelet, B., Paquot, M., Fuchs, R., Budzikiewicz, H. et al. (2001). Applied Biochemistry and Biotechnology, 91–93, 551–561.
Davis, D. A., Lynch, H. C., & Varley, J. (1999). Enzyme and Microbial Technology, 25, 322–329.
Jacques, P., Hbid, C., Destain, J., Razafindralambo, H., Paquot, M., De Pauw, E. et al. (1999). Applied Biochemistry and Biotechnology, 77–79, 223–233.
Ohno, A., Ano, T., & Shoda, M. (1993). Journal of Fermentation and Bioengineering, 75, 23–27.
Hbid, C., Jacques, P., Razafindralambo, H., Mpoyo, M. K., Meurice, E., Paquot, M. et al. (1996). Applied Biochemistry and Biotechnology, 57–58, 571–579.
Ohno, A., Ano, T., & Shoda, M. (1995). Biotechnology and Bioengineering, 47, 209–214.
Thimon, L., Peypoux, F., Magetdana, R., Roux, B., & Michel, G. (1992). Biotechnology and Applied Biochemistry, 16, 144–151.
Tsuge, K., Ano, T., & Shoda, M. (1996). Archives of Microbiology, 165, 243–251.
Kluge, B., Vater, J., Salnikow, J., & Eckart, K. (1988). FEBS Letters, 231, 107–110.
Huang, C. C., Liao, Z. M., Hirai, M., Ano, T., & Shoda, M. (1998). Journal of Fermentation and Bioengineering, 86, 605–607.
Ohno, A., Ano, T., & Shoda, M. (1992). Biotechnology Letters, 14, 817–822.
Kuehl, R. O. (2000). Design of experiments: Statistical principles of research design and analysis. Pacific Grove, CA: Duxbury.
Montgomery, D. C. (1991). Design and analysis of experiments. New York: Wiley.
Box, G. E. P., Hunter, W. G., & Hunter, J. S. (1978). Statistics for experimenters: An introduction to design, data analysis, and model building. New York: Wiley.
Myers, R. H., & Montgomery, D. C. (1995). Response surface methodology: Process and product optimization using designed experiments. New York: Wiley.
Pryor, S. W., Gibson, D. M., Krasnoff, S. B., & Walker, L. P. (2006). Transactions of the ASABE, 49, 1643–1649.
Bergstrom, G. C., & da Luz, W. C. (2005). Biocontrol for plants with Bacillus subtilis, Pseudomonas putida, and Sporobolomyces roseus. US Patent, 6, 896–883 B2.
Pryor, S. W., Gibson, D. M., Bergstrom, G. C., & Walker, L. P. (2007). BioTechniques, 42, 168–172.
Acknowledgements
We thank Dr. Gary Bergstrom and Dr. Thomas Zitter in the Department of Plant Pathology, Cornell University, for providing the B. subtilis and F. oxysporum cultures, respectively. This research was supported in part by a USDA MGET (Multi-Disciplinary Graduate Education Traineeship) program grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pryor, S.W., Gibson, D.M., Hay, A.G. et al. Optimization of Spore and Antifungal Lipopeptide Production During the Solid-state Fermentation of Bacillus subtilis . Appl Biochem Biotechnol 143, 63–79 (2007). https://doi.org/10.1007/s12010-007-0036-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-007-0036-1